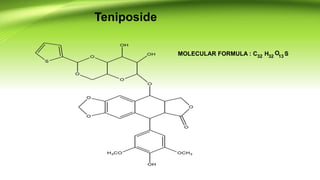
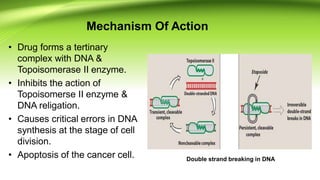
The document discusses the anticancer drugs etoposide and teniposide, including their molecular structures, history, chemistry, mechanisms of action, and medicinal uses. Etoposide and teniposide are effective against various cancers, such as small cell lung cancer and lymphocytic leukemia, by inhibiting DNA topoisomerase II, leading to cell apoptosis. Historical development details and references from medicinal chemistry textbooks are also provided.