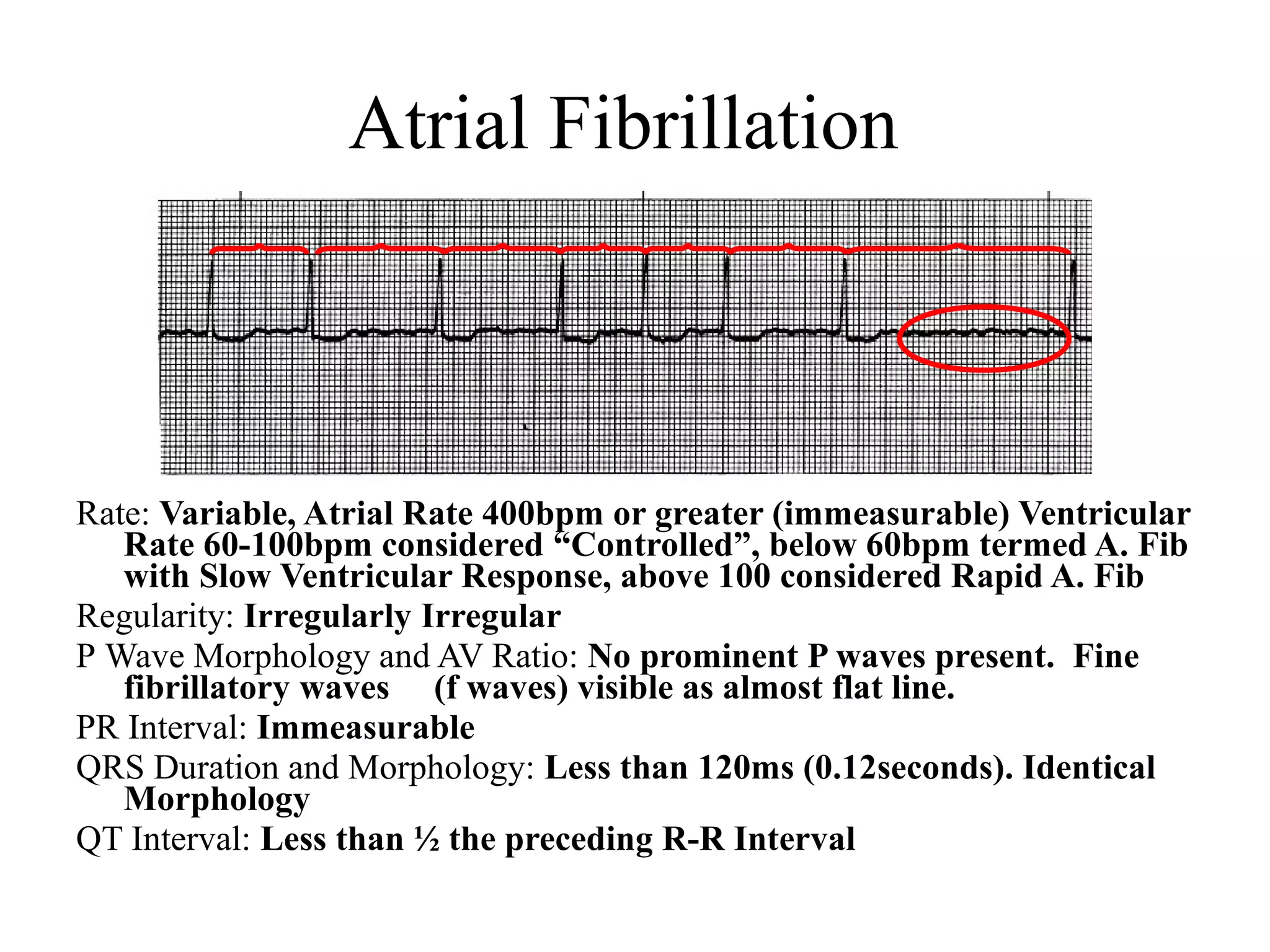
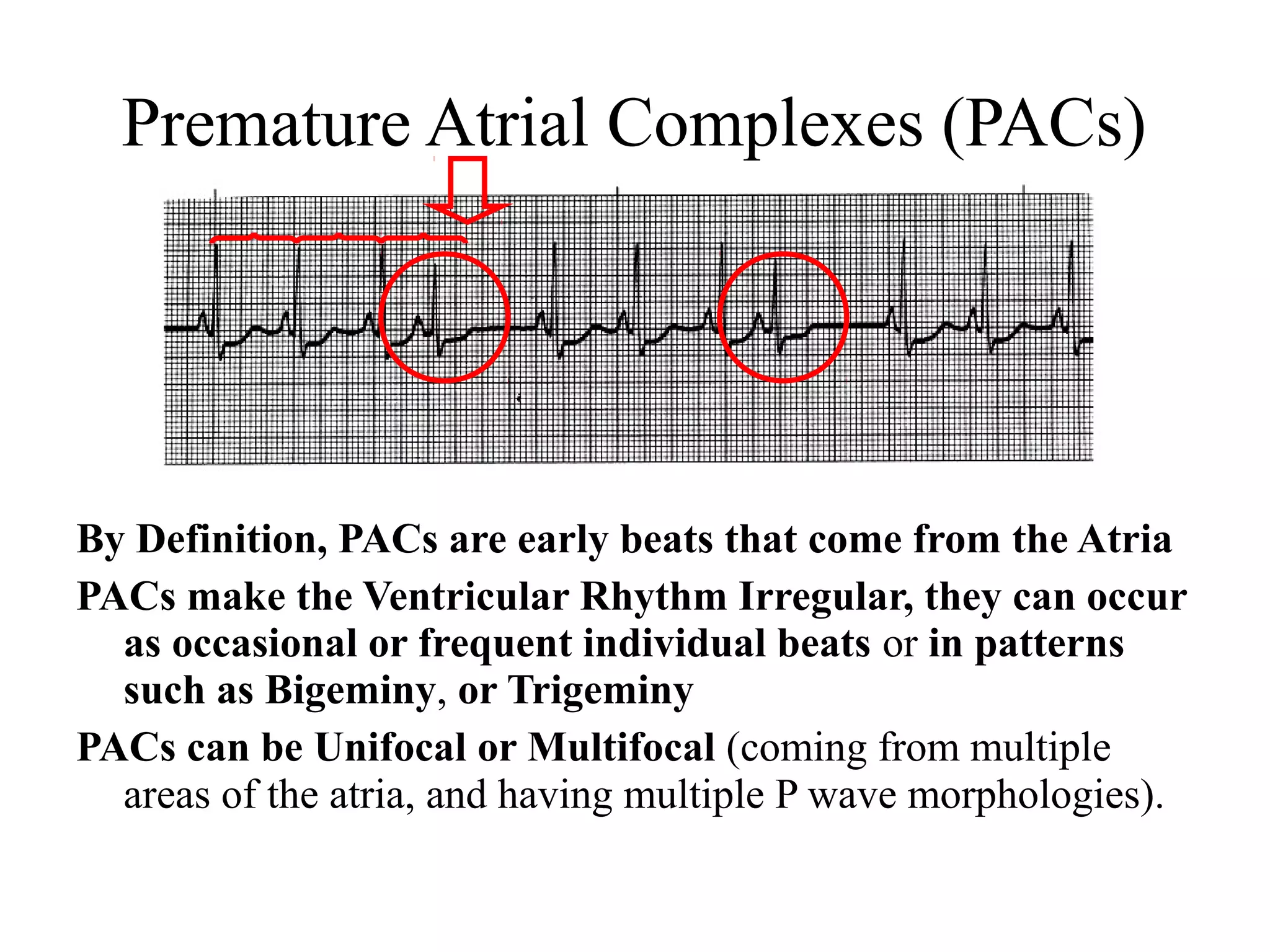
This document provides an overview of basic cardiac dysrhythmias including atrial fibrillation, atrial flutter, premature atrial complexes, wandering atrial pacemaker, junctional escape rhythms, accelerated junctional rhythms, premature junctional complexes, and supraventricular tachycardia. It describes the characteristics, causes, concerns and treatment approaches for each of these arrhythmias.