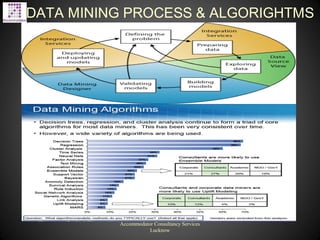
The document proposes software solutions for drug research, including text mining, data warehousing, data mining, database development, and big data analytics. It discusses common challenges in drug research like the high costs and low success rates. It then describes various solutions like text mining patents and research to help identify new research opportunities and reduce duplication of efforts. It provides examples of how various pharmaceutical companies use data mining and warehousing techniques. Overall, the document pitches different IT solutions that can help pharmaceutical and life sciences companies address their research challenges and make their processes more efficient.