DPCO List with effect from April 1 2017
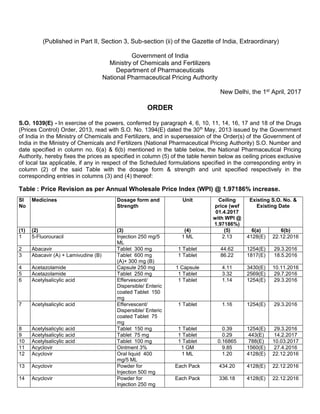
- 1. (Published in Part II, Section 3, Sub-section (ii) of the Gazette of India, Extraordinary) Government of India Ministry of Chemicals and Fertilizers Department of Pharmaceuticals National Pharmaceutical Pricing Authority New Delhi, the 1st April, 2017 ORDER S.O. 1039(E) - In exercise of the powers, conferred by paragraph 4, 6, 10, 11, 14, 16, 17 and 18 of the Drugs (Prices Control) Order, 2013, read with S.O. No. 1394(E) dated the 30th May, 2013 issued by the Government of India in the Ministry of Chemicals and Fertilizers, and in supersession of the Order(s) of the Government of India in the Ministry of Chemicals and Fertilizers (National Pharmaceutical Pricing Authority) S.O. Number and date specified in column no. 6(a) & 6(b) mentioned in the table below, the National Pharmaceutical Pricing Authority, hereby fixes the prices as specified in column (5) of the table herein below as ceiling prices exclusive of local tax applicable, if any in respect of the Scheduled formulations specified in the corresponding entry in column (2) of the said Table with the dosage form & strength and unit specified respectively in the corresponding entries in columns (3) and (4) thereof: Table : Price Revision as per Annual Wholesale Price Index (WPI) @ 1.97186% increase. Sl No Medicines Dosage form and Strength Unit Ceiling price (wef 01.4.2017 with WPI @ 1.97186%) Existing S.O. No. & Existing Date (1) (2) (3) (4) (5) 6(a) 6(b) 1 5-Fluorouracil Injection 250 mg/5 ML 1 ML 2.13 4128(E) 22.12.2016 2 Abacavir Tablet 300 mg 1 Tablet 44.62 1254(E) 29.3.2016 3 Abacavir (A) + Lamivudine (B) Tablet 600 mg (A)+ 300 mg (B) 1 Tablet 86.22 1817(E) 18.5.2016 4 Acetazolamide Capsule 250 mg 1 Capsule 4.11 3430(E) 10.11.2016 5 Acetazolamide Tablet 250 mg 1 Tablet 3.32 2569(E) 29.7.2016 6 Acetylsalicylic acid Effervescent/ Dispersible/ Enteric coated Tablet 150 mg 1 Tablet 1.14 1254(E) 29.3.2016 7 Acetylsalicylic acid Effervescent/ Dispersible/ Enteric coated Tablet 75 mg 1 Tablet 1.16 1254(E) 29.3.2016 8 Acetylsalicylic acid Tablet 150 mg 1 Tablet 0.39 1254(E) 29.3.2016 9 Acetylsalicylic acid Tablet 75 mg 1 Tablet 0.29 443(E) 14.2.2017 10 Acetylsalicylic acid Tablet 100 mg 1 Tablet 0.16865 788(E) 10.03.2017 11 Acyclovir Ointment 3% 1 GM 9.85 1560(E) 27.4.2016 12 Acyclovir Oral liquid 400 mg/5 ML 1 ML 1.20 4128(E) 22.12.2016 13 Acyclovir Powder for Injection 500 mg Each Pack 434.20 4128(E) 22.12.2016 14 Acyclovir Powder for Injection 250 mg Each Pack 336.18 4128(E) 22.12.2016
- 2. 15 Acyclovir Tablet 200 mg 1 Tablet 6.41 2195(E) 23.6.2016 16 Acyclovir Tablet 400 mg 1 Tablet 11.65 2195(E) 23.6.2016 17 Adenosine Injection 3 mg/ML 1 ML 84.38 443(E) 14.2.2017 18 Adrenaline Injection 1 mg/ML 1 ML 12.57 3181(E) 7.10.2016 19 Albendazole Oral Liquid 200mg/5ML 1 ML 1.51 248(E) 24.01.2017 20 Albendazole Tablet 400 mg 1 Tablet 7.29 2195(E) 23.6.2016 21 Allopurinol Tablet 100mg 1 Tablet 1.79 1951(E) 2.6.2016 22 Allopurinol Tablet 300 mg 1 Tablet 7.08 1254(E) 29.3.2016 23 Alprostadil Injection 0.5 mg/ML 1 ML 5,358.16 1560(E) 27.4.2016 24 Amiodarone Tablet 200 mg 1 Tablet 10.72 1819(E) 18.5.2016 25 Amiodarone Injection 50 mg/ML 1 ML 18.55 443(E) 14.2.2017 26 Amiodarone Tablet 100 mg 1 Tablet 5.40 2195(E) 23.6.2016 27 Amitriptyline Tablet 50 mg 1 Tablet 5.50 1404(E) 12.4.2016 28 Amitriptyline Tablet 25 mg 1 Tablet 2.14 2195(E) 23.6.2016 29 Amitriptyline Tablet 75 mg 1 Tablet 5.31 1404(E) 12.4.2016 30 Amitriptyline Tablet 10 mg 1 Tablet 2.17 1404(E) 12.4.2016 31 Amlodipine Tablet 5mg 1 Tablet 2.38 1686(E) 9.5.2016 32 Amlodipine Tablet 10 mg 1 Tablet 4.80 1254(E) 29.3.2016 33 Amlodipine Tablet 2.5 mg 1 Tablet 1.56 2195(E) 23.6.2016 34 Amoxicillin Capsule 500 mg 1 Capsule 5.81 1819(E) 18.5.2016 35 Amoxicillin Capsule 250mg 1 Capsule 2.09 1951(E) 2.6.2016 36 Amoxicillin Oral liquid 250 mg/5 ML 1 ML 1.28 1560(E) 27.4.2016 37 Amoxicillin (A) + Clavulanic acid (B) Powder for injection 1GM (A) +200mg (B) Each Pack 116.16 1819(E) 18.5.2016 38 Amoxicillin (A) + Clavulanic acid (B) Dry Syrup 125 mg (A) + 31.25 (B)/5 ML 1 ML 2.04 1560(E) 27.4.2016 39 Amoxicillin (A) + Clavulanic acid (B) Oral Liquid 200mg (A) + 28.5mg (B)/5ML 1 ML 1.71 1951(E) 2.6.2016 40 Amoxicillin (A) + Clavulanic acid (B) Powder for Injection 500 mg (A) + 100 mg (B) Each Pack 85.18 4128(E) 22.12.2016 41 Amoxicillin (A) + Clavulanic acid (B) Tablet 500 mg (A) + 125 mg (B) 1 Tablet 16.46 1819(E) 18.5.2016 42 Amphotericin B – Conventional Powder for Injection 50 mg Each Pack 281.05 788(E) 10.03.2017 43 Amphotericin B – Lipid/ Liposomal Powder for Injection 50 mg Each Pack 3,394.25 788(E) 10.03.2017 44 Ampicillin Powder for Injection 1 GM Each Pack 20.20 1404(E) 12.4.2016 45 Ampicillin Powder for Injection 500 mg Each Pack 12.44 4128(E) 22.12.2016 46 Artemether (A) + Lumefantrine (B) Tablet 20 mg (A) + 120 mg (B) 1 Tablet 11.85 1254(E) 29.3.2016 47 Artemether (A) + Lumefantrine (B) Tablet 40 mg (A) + 240 mg (B) 1 Tablet 15.27 1254(E) 29.3.2016 48 Artemether (A) + Lumefantrine (B) Tablet 80 mg (A) + 480 mg (B) 1 Tablet 21.92 1254(E) 29.3.2016 49 Artesunate Powder for Injection 120 mg Each Pack 377.59 1404(E) 12.4.2016 50 Artesunate Powder for Injection 60 mg Each Pack 198.33 1404(E) 12.4.2016 51 Artesunate (A) + Sulphadoxine - 1 Tablet 50 mg (A) Combi Pack 20.08 3431(E) 10.11.2016
- 3. Pyrimethamine (B) + 1 Tablet (500 mg + 25 mg) (B) 52 Ascorbic Acid (Vitamin C) Tablet 500mg 1 Tablet 0.86 4129(E) 22.12.2016 53 Atazanavir (A) + Ritonavir (B) Tablet 300 mg (A) + 100 mg (B) 1 Tablet 93.41 1254(E) 29.3.2016 54 Atenolol Tablet 100mg 1 Tablet 3.30 2569(E) 29.7.2016 55 Atenolol Tablet 50 mg 1 Tablet 1.69 2195(E) 23.6.2016 56 Atorvastatin Tablet 40 mg 1 Tablet 18.26 1254(E) 29.3.2016 57 Atorvastatin Tablet 10 mg 1 Tablet 5.19 1254(E) 29.3.2016 58 Atorvastatin Tablet 20 mg 1 Tablet 12.58 1254(E) 29.3.2016 59 Atracurium Injection 10 mg/ML Each Pack 49.59 4128(E) 22.12.2016 60 Atropine Injection 0.6mg/ML 1 ML 3.80 2730(E) 17.8.2016 61 Atropine Ointment 1% 1 GM 3.43 4128(E) 22.12.2016 62 Atropine Drops 1% 1 ML 3.11 3431(E) 10.11.2016 63 Azathioprine Tablet 50mg 1 Tablet 9.20 1951(E) 2.6.2016 64 Azithromycin Capsule 250 mg 1 Capsule 9.94 3180(E) 7.10.2016 65 Azithromycin Capsule 500 mg 1 Capsule 15.68 3180(E) 7.10.2016 66 Azithromycin Oral Liquid 200mg/5ML 1 ML 2.91 1687(E) 9.5.2016 67 Azithromycin Powder for Injection 500 mg Each Pack 180.39 4128(E) 22.12.2016 68 Baclofen Tablet 10 mg 1 Tablet 9.75 1254(E) 29.3.2016 69 Baclofen Tablet 20 mg 1 Tablet 13.43 1254(E) 29.3.2016 70 Baclofen Tablet 5 mg 1 Tablet 5.03 1254(E) 29.3.2016 71 Benzathine benzylpenicillin Powder for Injection 12 lac units Each Pack 11.49 4128(E) 22.12.2016 72 Benzathine benzylpenicillin Powder for Injection 6 lac units Each Pack 7.61 4128(E) 22.12.2016 73 Benzoyl Peroxide Cream 2.5% 1 GM 2.22 3430(E) 10.11.2016 74 Benzoyl Peroxide Gel 2.5% 1 GM 3.55 1560(E) 27.4.2016 75 Betamethasone Cream 0.05% 1 GM 0.55 3431(E) 10.11.2016 76 Betamethasone Gel 0.05% 1 GM 0.50 247(E) 24.01.2017 77 Betamethasone Injection 4 mg/ML 1 ML 3.97 3431(E) 10.11.2016 78 Bicalutamide Tablet 50 mg 1 Tablet 64.91 1404(E) 12.4.2016 79 Bisacodyl Suppository 5mg 1 Suppository 7.98 248(E) 24.01.2017 80 Bisacodyl Tablet 5mg 1 Tablet 0.95 1951(E) 2.6.2016 81 Bleomycin Powder for Injection 15 Units Each Pack 563.26 3181(E) 7.10.2016 82 Bortezomib Powder for Injection 2mg Each Pack 11,636.60 1404(E) 12.4.2016 83 Budesonide Inhalation (MDI/DPI)100 mcg/dose 1 Dose 1.24 1560(E) 27.4.2016 84 Budesonide Inhalation (MDI/DPI) 200 mcg/dose 1 Dose 1.53 1560(E) 27.4.2016 85 Budesonide Respiratory Solution for use in Nebulizer 0.5mg/ML 1 ML 10.13 1560(E) 27.4.2016 86 Budesonide Respiratory Solution for use in Nebulizer 1mg/ML 1 ML 12.22 1560(E) 27.4.2016 87 Budesonide (A)+ Formoterol (B) Inhalation (MDI/DPI) 400 mcg (A) + 6 mcg (B) 1 Dose 2.79 1560(E) 27.4.2016
- 4. 88 Budesonide (A)+ Formoterol (B) Inhalation (MDI/DPI) 200 mcg (A) + 6 mcg (B) 1 Dose 2.23 1560(E) 27.4.2016 89 Budesonide (A)+ Formoterol (B) Inhalation (MDI/DPI) 100 mcg (A) + 6 mcg (B) 1 Dose 1.77 1560(E) 27.4.2016 90 Bupivacaine Injection 0.25% 1 ML 2.31 3181(E) 7.10.2016 91 Bupivacaine Injection 0.50% 1 ML 3.71 3181(E) 7.10.2016 92 Bupivacaine Injection 0.5% with 7.5% glucose 1 ML 5.88 788(E) 10.03.2017 93 Caffeine Injection 20 mg/ML 1 ML 237.48 1560(E) 27.4.2016 94 Caffeine Oral Liquid 20mg/ML 1 ML 201.70 1687(E) 9.5.2016 95 Calamine Lotion (As per IP) 1 ML 0.80 788(E) 10.03.2017 96 Calcium folinate Tablet 15 mg 1 Tablet 37.02 1404(E) 12.4.2016 97 Capecitabine Tablet 500 mg 1 Tablet 119.58 1560(E) 27.4.2016 98 Capreomycin Powder for Injection 1 GM Each Pack 327.72 1404(E) 12.4.2016 99 Carbamazepine Tablet 100 mg 1 Tablet 0.65 2963(E) 15.09.2016 100 Carbamazepine CR Tablet 400 mg 1 Tablet 2.89 1254(E) 29.3.2016 101 Carbamazepine CR Tablet 200 mg 1 Tablet 1.47 1254(E) 29.3.2016 102 Carbamazepine Oral Liqiud 100mg/5 ML 1 ML 0.18355 2730(E) 17.8.2016 103 Carbamazepine Tablet 400 mg 1 Tablet 3.13 1254(E) 29.3.2016 104 Carbamazepine Tablet 200 mg 1 Tablet 1.31 2195(E) 23.6.2016 105 Carbimazole Tablet 10mg 1 Tablet 3.49 1819(E) 18.5.2016 106 Carbimazole Tablet 5mg 1 Tablet 1.79 1951(E) 2.6.2016 107 Carboplatin Injection 10 mg/ML 1 ML 50.35 1561 (E) 27.4.2016 108 Carboxymethylcellulose Drops 0.5% 1 ML 12.08 3432(E) 10.11.2016 109 Carboxymethylcellulose Drops 1% 1 ML 16.76 3432(E) 10.11.2016 110 Cefadroxil Tablet 1 GM 1 Tablet 5.94 2962(E) 15.09.2016 111 Cefadroxil Capsule 500mg 1 Capsule 6.77 1687(E) 9.5.2016 112 Cefadroxil Oral liquid 125 mg/5 ML 1 ML 0.59 1560(E) 27.4.2016 113 Cefadroxil Tablet 500 mg 1 Tablet 3.82 1254(E) 29.3.2016 114 Cefazolin Powder for Injection 1 GM Each Pack 23.23 1404(E) 12.4.2016 115 Cefazolin Powder for injection 500 mg Each Pack 14.72 2962(E) 15.09.2016 116 Cefixime Capsule 400 mg 1 Capsule 27.66 4127(E) 22.12.2016 117 Cefixime Capsule 200 mg 1 Capsule 13.65 247(E) 24.01.2017 118 Cefixime Oral liquid 100 mg/5 ML 1 ML 2.03 1687(E) 9.5.2016 119 Cefixime Oral liquid 50 mg/5 ML 1 ML 1.37 1687(E) 9.5.2016 120 Cefixime Tablet 200 mg 1 Tablet 8.65 1819(E) 18.5.2016 121 Cefixime Tablet 400 mg 1 Tablet 20.88 1254(E) 29.3.2016 122 Cefotaxime Powder for Injection 500 mg Each Pack 19.42 443(E) 14.2.2017 123 Cefotaxime Powder for Injection 250 mg Each Pack 14.76 443(E) 14.2.2017 124 Cefotaxime Powder for Injection 1 GM Each Pack 32.48 1817(E) 18.5.2016 125 Ceftazidime Powder for Injection 250 mg Each Pack 61.58 3431(E) 10.11.2016 126 Ceftazidime Powder for Injection 1GM Each Pack 198.02 3431(E) 10.11.2016
- 5. 127 Ceftriaxone Powder for Injection 2 GM Each Pack 121.24 1404(E) 12.4.2016 128 Ceftriaxone Powder for Injection 500 mg Each Pack 42.65 1404(E) 12.4.2016 129 Ceftriaxone Powder for Injection 1GM Each Pack 48.90 1686(E) 9.5.2016 130 Ceftriaxone Powder For Injection 250mg Each Pack 23.35 1951(E) 2.6.2016 131 Cetirizine Tablet 10mg 1 Tablet 1.56 1951(E) 2.6.2016 132 Cetirizine Capsule 10mg 1 Capsule 2.25 247(E) 24.01.2017 133 Cetirizine Oral liquid 5 mg/5 ML 1 ML 0.56 1687(E) 9.5.2016 134 Cetrimide Solution 20% (concentrate for dilution) 1 ML 0.30 3431(E) 10.11.2016 135 Chlorambucil Tablet 5 mg 1 Tablet 75.26 1404(E) 12.4.2016 136 Chlorambucil Tablet 2mg 1 Tablet 33.49 2569(E) 29.7.2016 137 Chloroquine Oral Liquid 50 mg/5 ML 1 ML 0.27 443(E) 14.2.2017 138 Chloroquine Tablet 150mg 1 Tablet 0.60 443(E) 14.2.2017 139 Chlorpheniramine Tablet 4mg 1 Tablet 0.08682 2569(E) 29.7.2016 140 Cholecalciferol Tablet 60000 IU 1 Tablet 22.94 2964(E) 15.9.2016 141 Cholecalciferol Capsule 1000 IU 1 Capsule 4.51 1687(E) 9.5.2016 142 Cholecalciferol Capsule 60000 IU 1 Capsule 27.94 2964(E) 15.9.2016 143 Cholecalciferol Oral liquid 400 IU/ML 1 ML 2.23 1687(E) 9.5.2016 144 Cholecalciferol Tablet 1000 IU 1 Tablet 3.55 1254(E) 29.3.2016 145 Ciprofloxacin Injection 200mg/100ML 1 ML 0.15968 443(E) 14.2.2017 146 Ciprofloxacin Drops 0.30% 1 ML 1.44 443(E) 14.2.2017 147 Ciprofloxacin Tablet 250 mg 1 Tablet 1.65 1686(E) 9.5.2016 148 Ciprofloxacin Tablet 500 mg 1 Tablet 3.37 1686(E) 9.5.2016 149 Ciprofloxacin Ointment 0.3% 1 GM 1.06 3431(E) 10.11.2016 150 Cisplatin Injection 1mg /ML 1 ML 6.52 1561(E) 27.4.2016 151 Clarithromycin Oral liquid 125mg/5 ML 1 ML 4.09 1560(E) 27.4.2016 152 Clarithromycin Tablet 250 mg 1 Tablet 26.49 1254(E) 29.3.2016 153 Clarithromycin Tablet 500 mg 1 Tablet 46.69 1254(E) 29.3.2016 154 Clindamycin Capsule 300 mg 1 Capsule 21.43 1254(E) 29.3.2016 155 Clindamycin Capsule 150 mg 1 Capsule 13.13 1254(E) 29.3.2016 156 Clindamycin Tablet 300mg 1 Tablet 13.83 1686(E) 9.5.2016 157 Clobazam Tablet 5 mg 1 Tablet 4.86 1254(E) 29.3.2016 158 Clobazam Tablet 10 mg 1 Tablet 8.55 1254(E) 29.3.2016 159 Clofazimine Capsule 100mg 1 Capsule 2.24 2569(E) 29.7.2016 160 Clofazimine Capsule 50mg 1 Capsule 1.31 2569(E) 29.7.2016 161 Clomiphene Capsule 100 mg 1 Capsule 52.66 3430(E) 10.11.2016 162 Clomiphene Tablet 50mg 1 Tablet 7.05 443(E) 14.2.2017 163 Clomiphene Tablet 100mg 1 Tablet 11.07 443(E) 14.2.2017 164 Clomipramine Tablet 75mg 1 Tablet 13.58 1687(E) 9.5.2016 165 Clomipramine Tablet 25mg 1 Tablet 5.25 1687(E) 9.5.2016 166 Clomipramine Tablet 10mg 1 Tablet 2.38 1687(E) 9.5.2016 167 Clonazepam Tablet 0.5 mg 1 Tablet 2.94 1404(E) 12.4.2016 168 Clonazepam Tablet 0.25 mg 1 Tablet 1.73 1404(E) 12.4.2016 169 Clonazepam Tablet 1 mg 1 Tablet 4.00 1404(E) 12.4.2016 170 Clopidogrel Tablet 75 mg 1 Tablet 6.57 1686(E) 9.5.2016 171 Clotrimazole Cream 1% 1 GM 2.40 1687(E) 9.5.2016
- 6. 172 Clotrimazole Drop 1% 1 ML 2.87 1687(E) 9.5.2016 173 Clotrimazole Pessary 100mg 1 Pessary 8.22 248(E) 24.01.2017 174 Clozapine Tablet 100 mg 1 Tablet 7.33 1404(E) 12.4.2016 175 Clozapine Tablet 25 mg 1 Tablet 2.38 1404(E) 12.4.2016 176 Clozapine Tablet 50 mg 1 Tablet 4.55 1404(E) 12.4.2016 177 Coagulation factor VIII Powder for Injection 250 IU Each pack 3,389.14 1404(E) 12.4.2016 178 Colchicine Tablet 0.5 mg 1 Tablet 2.94 2569(E) 29.7.2016 179 Co-trimoxazole (Sulphamethoxazole (A)+Trimethoprim (B)] Tablet 800mg (A)+160mg (B) 1 Tablet 1.00 443(E) 14.2.2017 180 Co-trimoxazole (Sulphamethoxazole (A)+Trimethoprim (B)] Oral Liquid 200mg (A)+40mg (B)/5ML 1 ML 0.20394 443(E) 14.2.2017 181 Co-trimoxazole (Sulphamethoxazole (A)+Trimethoprim (B)] Tablet 400mg (A)+80mg (B) 1 Tablet 0.49 443(E) 14.2.2017 182 Cyclophosphamide Powder for Injection 500 mg Each Pack 72.42 3181(E) 7.10.2016 183 Cyclophosphamide Tablet 50 mg 1 Tablet 3.68 2569(E) 29.7.2016 184 Cycloserine Tablet 250mg 1 Tablet 44.16 1687(E) 9.5.2016 185 Cycloserine Capsule 250 mg 1 Capsule 51.37 1254(E) 29.3.2016 186 Cyclosporine Capsule 50 mg 1 Capsule 47.77 2195(E) 23.6.2016 187 Cyclosporine Capsule 100 mg 1 Capsule 93.01 2195(E) 23.6.2016 188 Cyclosporine Capsule 25 mg 1 Capsule 24.96 2963(E) 15.09.2016 189 Cyclosporine Oral liquid 100 mg/ML 1 ML 85.77 2731(E) 17.8.2016 190 Cytosine arabinoside Injection 100 mg/ ML Each Pack 180.27 4128(E) 22.12.2016 191 Cytosine arabinoside Powder for Injection 500 mg Each Pack 464.71 4128(E) 22.12.2016 192 Cytosine arabinoside Powder for Injection 1000 mg Each Pack 972.31 443(E) 14.2.2017 193 Dacarbazine Powder for Injection 200 mg Each Pack 415.65 1404(E) 12.4.2016 194 Dacarbazine Powder for Injection 500 mg Each Pack 937.67 3181(E) 7.10.2016 195 Darunavir Tablet 600 mg 1 Tablet 157.93 1254(E) 29.3.2016 196 Dexamethasone Injection 4mg/ML Each Pack (10ML) 13.03 248(E) 24.01.2017 197 Dexamethasone Injection 4mg/ML Each Pack (30ML) 33.38 248(E) 24.01.2017 198 Dexamethasone Injection 4mg/ML Each Pack (20ML) 24.97 248(E) 24.01.2017 199 Dexamethasone Injection 4mg/ML Each Pack (2ML) 5.38 248(E) 24.01.2017 200 Dexamethasone Tablet 0.5mg 1 Tablet 0.18355 1819(E) 18.5.2016 201 Diazepam Injection 5mg/ML 1 ML 5.73 3181(E) 7.10.2016 202 Diazepam Oral liquid 2 mg/5 ML 1 ML 2.59 4127(E) 22.12.2016 203 Diazepam suppository 5 mg 1 Suppository 5.65 2730(E) 17.8.2016 204 Diazepam Tablet 5 mg 1 Tablet 1.35 2569(E) 29.7.2016 205 Diazepam Tablet 2mg 1 Tablet 1.42 2569(E) 29.7.2016 206 Diclofenac Injection 25mg/ML 1 ML 1.38 4128(E) 22.12.2016 207 Diclofenac Tablet 50mg 1 Tablet 1.80 1951(E) 2.6.2016 208 Dicyclomine Injection 10 mg/ML 1 ML 2.49 3431(E) 10.11.2016 209 Diethylcarbamazine Oral liquid 120 mg/5 ML 1 ML 0.44 1560(E) 27.4.2016 210 Diethylcarbamazine Tablet 100 mg 1 Tablet 1.28 1254(E) 29.3.2016 211 Diethylcarbamazine Tablet 50 mg 1 Tablet 0.51 2195(E) 23.6.2016 212 Digoxin Tablet 0.25mg 1 Tablet 1.13 1819(E) 18.5.2016
- 7. 213 Diltiazem Capsule 90mg 1 Capsule 9.09 1687(E) 9.5.2016 214 Diltiazem Injection 5 mg/ML 1 ML 2.98 4128(E) 22.12.2016 215 Diltiazem SR Tablet 90 mg 1 Tablet 8.83 1254(E) 29.3.2016 216 Diltiazem Tablet 30 mg 1 Tablet 2.27 2195(E) 23.6.2016 217 Diltiazem Tablet 60 mg 1 Tablet 4.62 2195(E) 23.6.2016 218 Dinoprostone Gel 0.5 mg 1 GM 76.69 1560(E) 27.4.2016 219 Dobutamine Injection 50mg/ML Each Pack 35.48 4128(E) 22.12.2016 220 Docetaxel Powder for Injection 80 mg Each Pack 10,767.88 1404(E) 12.4.2016 221 Docetaxel Powder for Injection 20 mg Each Pack 2,832.12 1404(E) 12.4.2016 222 Domperidone Tablet 10 mg 1 Tablet 2.28 2195(E) 23.6.2016 223 Domperidone Oral Liquid 1mg/ML 1 ML 1.02 4128(E) 22.12.2016 224 Donepezil Tablet 10 mg 1 Tablet 15.18 1254(E) 29.3.2016 225 Donepezil Tablet 5 mg 1 Tablet 10.64 1254(E) 29.3.2016 226 Dopamine Injection 40mg/ML 1 ML 5.12 4128(E) 22.12.2016 227 Doxorubicin Injection 2mg/ML 1 ML 33.97 1686(E) 9.5.2016 228 Doxycycline Capsule 100mg 1 Capsule 2.37 2568(E) 29.7.2016 229 Doxycycline Tablet 100 mg 1 Tablet 0.92 3181(E) 7.10.2016 230 Efavirenz Capsule 200 mg 1 Capsule 21.85 3431(E) 10.11.2016 231 Efavirenz Capsule 600 mg 1 Capsule 56.91 3431(E) 10.11.2016 232 Efavirenz Tablet 200 mg 1 Tablet 20.57 3430(E) 10.11.2016 233 Efavirenz Tablet 600 mg 1 Tablet 64.44 2729(E) 17.8.2016 234 Enalapril Tablet 2.5 mg 1 Tablet 1.82 2195(E) 23.6.2016 235 Enalapril Tablet 5 mg 1 Tablet 3.03 2195(E) 23.6.2016 236 Enoxaparin Injection 40mg/0.4ML 0.1 ML 92.24 2963(E) 15.09.2016 237 Enoxaparin Injection 60mg/0.6ML 0.1 ML 92.24 2963(E) 15.09.2016 238 Entecavir Tablet 0.5 mg 1 Tablet 72.40 1254(E) 29.3.2016 239 Entecavir Tablet 1 mg 1 Tablet 115.03 1254(E) 29.3.2016 240 Erythropoietin Injection 10000 IU/ML Each pack 2,396.09 1404(E) 12.4.2016 241 Erythropoietin Injection 2000 IU/ML Each pack 573.58 1404(E) 12.4.2016 242 Escitalopram Tablet 10 mg 1 Tablet 7.67 1404(E) 12.4.2016 243 Escitalopram Tablet 20 mg 1 Tablet 12.11 1404(E) 12.4.2016 244 Escitalopram Tablet 5 mg 1 Tablet 4.33 1404(E) 12.4.2016 245 Ethambutol Tablet 800 mg 1 Tablet 3.84 2195(E) 23.6.2016 246 Ethambutol Tablet 600mg 1 Tablet 3.21 2569(E) 29.7.2016 247 Ethambutol Tablet 400mg 1 Tablet 2.18 2569(E) 29.7.2016 248 Ethambutol Tablet 200 mg 1 Tablet 0.96 2569(E) 29.7.2016 249 Ethinylestradiol Tablet 0.05 mg 1 Tablet 3.40 4128(E) 22.12.2016 250 Ethinylestradiol Tablet 0.01mg 1 Tablet 2.14 2569(E) 29.7.2016 251 Ethinylestradiol (A) + Levonorgestrel (B) Tablet 0.03mg (A) + 0.15mg (B) 1 Tablet 3.13 1951(E) 2.6.2016 252 Ethionamide Tablet 250 mg 1 Tablet 14.94 1254(E) 29.3.2016 253 Etoposide Injection 20 mg/ML 1 ML 33.92 4128(E) 22.12.2016 254 Fentanyl Injection 50mcg/ML 1 ML 21.48 4128(E) 22.12.2016 255 Filgrastim Injection 300 mcg Each pack 1,298.39 1404(E) 12.4.2016 256 Fluconazole Capsule 100 mg 1 Capsule 23.90 3431(E) 10.11.2016 257 Fluconazole Capsule 200 mg 1 Capsule 34.54 3431(E) 10.11.2016 258 Fluconazole Injection 200 mg /100 ML Each Pack 100.69 1404(E) 12.4.2016 259 Fluconazole Tablet 400 mg 1 Tablet 29.56 1254(E) 29.3.2016
- 8. 260 Fluconazole Tablet 200 mg 1 Tablet 17.77 2195(E) 23.6.2016 261 Fluconazole Tablet 150mg 1 Tablet 11.21 1951(E) 2.6.2016 262 Fluconazole Tablet 100mg 1 Tablet 7.55 2569(E) 29.7.2016 263 Flunarizine Tablet 10 mg 1 Tablet 4.47 1254(E) 29.3.2016 264 Flunarizine Tablet 5 mg 1 Tablet 2.61 1254(E) 29.3.2016 265 Fluoxetine Tablet 10mg 1 Tablet 2.36 1687(E) 9.5.2016 266 Fluoxetine Tablet 40mg 1 Tablet 5.05 1687(E) 9.5.2016 267 Fluoxetine Tablet 20 mg 1 Tablet 3.75 3180(E) 7.10.2016 268 Fluoxetine Capsule 10 mg 1 Capsule 2.82 1404(E) 12.4.2016 269 Fluoxetine Capsule 40 mg 1 Capsule 5.24 1404(E) 12.4.2016 270 Fluoxetine Capsule 60 mg 1 Capsule 9.26 1404(E) 12.4.2016 271 Fluoxetine Capsule 20 mg 1 Capsule 3.45 2195(E) 23.6.2016 272 Fluoxetine Tablet 60mg 1 Tablet 7.00 1687(E) 9.5.2016 273 Fluphenazine Depot Injection 25 mg/ML 1 ML 47.34 1560(E) 27.4.2016 274 Folic Acid Capsule 5 mg 1 Capsule 5.33 3430(E) 10.11.2016 275 Folic Acid Tablet 5 mg 1 Tablet 1.31 1819(E) 18.5.2016 276 Furosemide Injection 10 mg/ ML Each Pack 1.03 788(E) 10.03.2017 277 Furosemide Tablet 40 mg 1 Tablet 0.47 788(E) 10.03.2017 278 Fusidic acid Cream 2% 1 GM 8.63 1560(E) 27.4.2016 279 Ganciclovir Powder for Injection 500 mg Each Pack 1,531.30 3180(E) 7.10.2016 280 Gefitinib Tablet 250 mg 1 Tablet 405.52 1404(E) 12.4.2016 281 Gemcitabine Powder for Injection 1 GM Each Pack 5,019.49 1406(E) 12.4.2016 282 Gemcitabine Powder for Injection 200 mg Each Pack 1,090.42 443(E) 14.2.2017 283 Gentamicin Injection 40mg/ML Each Pack (10ML) 17.00 248(E) 24.01.2017 284 Gentamicin Injection 40mg/ML Each Pack (20ML) 25.77 248(E) 24.01.2017 285 Gentamicin Injection 40mg/ML Each Pack (2ML) 4.56 248(E) 24.01.2017 286 Gentamicin Injection 40mg/ML Each Pack (30ML) 28.20 248(E) 24.01.2017 287 Gentamicin Drops 0.3% 1 ML 0.86 3431(E) 10.11.2016 288 Glimepiride Tablet 2 mg 1 Tablet 5.39 1254(E) 29.3.2016 289 Glimepiride Tablet 1 mg 1 Tablet 3.40 1254(E) 29.3.2016 290 Glucose Injection 25% 1 ML 0.16268 4128(E) 22.12.2016 291 Glucose (A) + Sodium Chloride (B) Injection 5% (A) + 0.9% (B) 1000ML Glass 61.15 2209(E) 24.6.2016 292 Glucose (A) + Sodium Chloride (B) Injection 5% (A) + 0.9% (B) 1000ML Non- Glass 49.42 2209(E) 24.6.2016 293 Glucose (A) + Sodium Chloride (B) Injection 5% (A) + 0.9% (B) 100ML Glass 17.11 2209(E) 24.6.2016 294 Glucose (A) + Sodium Chloride (B) Injection 5% (A) + 0.9% (B) 100ML Non-Glass 15.46 2209(E) 24.6.2016 295 Glucose (A) + Sodium Chloride (B) Injection 5% (A) + 0.9% (B) 250ML Glass 23.67 2209(E) 24.6.2016 296 Glucose (A) + Sodium Chloride (B) Injection 5% (A) + 0.9% (B) 250ML Non-Glass 21.38 2209(E) 24.6.2016 297 Glucose (A) + Sodium Chloride (B) Injection 5% (A) + 0.9% (B) 500ML Glass 33.62 2209(E) 24.6.2016 298 Glucose (A) + Sodium Chloride (B) Injection 5% (A) + 0.9% (B) 500ML Non-Glass 29.04 2209(E) 24.6.2016 299 Glucose Injection 5% 1000ML Glass 60.78 2209(E) 24.6.2016 300 Glucose Injection 5% 1000ML Non- Glass 49.04 2209(E) 24.6.2016 301 Glucose Injection 5% 100ML Glass 17.06 2209(E) 24.6.2016 302 Glucose Injection 5% 100ML Non-Glass 15.44 2209(E) 24.6.2016 303 Glucose Injection 5% 250ML Glass 23.57 2209(E) 24.6.2016
- 9. 304 Glucose Injection 5% 250ML Non-Glass 21.29 2209(E) 24.6.2016 305 Glucose Injection 5% 500ML Glass 33.45 2209(E) 24.6.2016 306 Glucose Injection 5% 500ML Non-Glass 28.87 2209(E) 24.6.2016 307 Glyceryl Trinitratre Sublingual Tablet 0.5 mg 1 Tablet 1.71 2569(E) 29.7.2016 308 Glycopyrrolate Injection 0.2 mg/ML 1 ML 11.63 1404(E) 12.4.2016 309 Griseofulvin Tablet 250 mg 1 Tablet 1.51 2195(E) 23.6.2016 310 Griseofulvin Tablet 375 mg 1 Tablet 4.08 787(E) 10.03.2017 311 Haloperidol Injection 5 mg/ML 1 ML 5.52 3181(E) 7.10.2016 312 Haloperidol Oral liquid 2 mg/ 5ML 1 ML 1.95 1687(E) 9.5.2016 313 Haloperidol Tablet 5 mg 1 Tablet 3.25 1404(E) 12.4.2016 314 Haloperidol Tablet 1.5 mg 1 Tablet 1.56 1404(E) 12.4.2016 315 Haloperidol Tablet 10 mg 1 Tablet 4.17 1404(E) 12.4.2016 316 Haloperidol Tablet 20 mg 1 Tablet 4.66 1404(E) 12.4.2016 317 Halothane Inhalation 1 ML 5.55 4128(E) 22.12.2016 318 Homatropine Drops 2% 1 ML 5.64 3431(E) 10.11.2016 319 Human chorionic Gonadotropin Injection 1000 IU Each Pack 192.84 1560(E) 27.4.2016 320 Human chorionic Gonadotropin Injection 5000 IU Each Pack 378.71 1560(E) 27.4.2016 321 Human Normal Immunoglobulin 1 ML 139.98 787(E) 10.03.2017 322 Hydrochlorothiazide Tablet 50 mg 1 Tablet 0.07830 442(E) 14.2.2017 323 Hydrochlorothiazide Tablet 12.5 mg 1 Tablet 0.93 2963(E) 15.09.2016 324 Hydrochlorothiazide Tablet 25 mg 1 Tablet 1.60 2963(E) 15.09.2016 325 Hydrocortisone Injection 200 mg Each Pack 54.44 3431(E) 10.11.2016 326 Hydrocortisone Injection 100mg Each Pack 35.31 1686(E) 9.5.2016 327 Hydrocortisone Powder for Injection 100mg Each Pack 35.31 1687(E) 9.5.2016 328 Hydroxychloroquine Tablet 200 mg 1 Tablet 5.61 1819(E) 18.5.2016 329 Hydroxychloroquine Tablet 400 mg 1 Tablet 11.30 1254(E) 29.3.2016 330 Hydroxypropyl methylcellulose Injection 2% 1 ML 35.48 1560(E) 27.4.2016 331 Hydroxyurea Capsule 500 mg 1 Capsule 11.63 1254(E) 29.3.2016 332 Hyoscine Butylbromide Tablet 10mg 1 Tablet 2.82 1951(E) 2.6.2016 333 Hyoscine butylbromide Injection 20 mg/ML 1 ML 9.86 4128(E) 22.12.2016 334 Ibuprofen Tablet 200mg 1 Tablet 0.36 2730(E) 17.8.2016 335 Ibuprofen Tablet 400mg 1 Tablet 0.66 2730(E) 17.8.2016 336 Ibuprofen Oral liquid 100mg/5ML 1 ML 0.18 4128(E) 22.12.2016 337 Imatinib Capsule 400 mg 1 Capsule 241.66 3430(E) 10.11.2016 338 Imatinib Tablet 400mg 1 Tablet 217.53 1951(E) 2.6.2016 339 Imatinib Capsule 100mg 1 Capsule 73.45 3180(E) 7.10.2016 340 Insulin (Soluble) Injection 40 IU/ML 1 ML 13.66 2193(E) 23.6.2016 341 Intermediate Acting (NPH) Insulin injection 40 IU/ML 1 ML 13.66 2193(E) 23.6.2016 342 Ipratropium Inhalation (MDI/DPI) 20 mcg/dose 1 Dose 0.58 4128(E) 22.12.2016 343 Ipratropium Respirator solution for use in nebulizer 250 mcg/ML 1 ML 2.40 1560(E) 27.4.2016 344 Iron sucrose Injection 20 mg/ML 1 ML 50.85 1404(E) 12.4.2016 345 Isoflurane Inhalation 1 ML 9.48 443(E) 14.2.2017 346 Isosorbide dinitrate Tablet 10 mg 1 Tablet 0.70 1254(E) 29.3.2016 347 Isosorbide dinitrate Tablet 5 mg 1 Tablet 0.72 1254(E) 29.3.2016 348 Isosorbide-5-mononitrate SR Tablet 30 mg 1 Tablet 4.63 1254(E) 29.3.2016 349 Isosorbide-5-mononitrate SR Tablet 60 mg 1 Tablet 6.04 1254(E) 29.3.2016 350 Isosorbide-5-mononitrate Tablet 10 mg 1 Tablet 1.86 443(E) 14.2.2017 351 Isosorbide-5-mononitrate Tablet 20 mg 1 Tablet 2.90 443(E) 14.2.2017
- 10. 352 Ispaghula Granules/ Husk/ Powder 1 GM 0.74 443(E) 14.2.2017 353 Kanamycin Powder for injection 500 mg Each Pack 20.19 2962(E) 15.09.2016 354 Kanamycin Powder for Injection 1 GM Each Pack 36.99 1404(E) 12.4.2016 355 Kanamycin Powder for Injection 750 mg Each Pack 34.48 1404(E) 12.4.2016 356 Ketamine Injection 50 mg/ML 1 ML 9.88 3181(E) 7.10.2016 357 Ketamine Injection 10 mg/ML 1 ML 10.02 3181(E) 7.10.2016 358 Labetalol Injection 5 mg/ML 1 ML 47.96 1560(E) 27.4.2016 359 Lactulose Oral liquid 10 g/15 ML 1 ML 1.00 1560(E) 27.4.2016 360 Lamivudine (A) + Nevirapine (B) + Stavudine (C) Tablet 150 mg (A) + 200 mg (B) + 30 mg(C) 1 Tablet 14.76 4128(E) 22.12.2016 361 Lamivudine (A) + Zidovudine (B) Tablet 150mg (A) + 300mg (B) 1 Tablet 18.98 2569(E) 29.7.2016 362 L-Asparaginase Powder for Injection 10000 KU Each Pack 1,518.86 1404(E) 12.4.2016 363 Leflunomide Tablet 20 mg 1 Tablet 18.49 2195(E) 23.6.2016 364 Leflunomide Tablet 10 mg 1 Tablet 9.48 2195(E) 23.6.2016 365 Letrozole Tablet 2.5 mg 1 Tablet 37.00 1404(E) 12.4.2016 366 Levetiracetam ER Tablet 750 mg 1 Tablet 16.45 1254(E) 29.3.2016 367 Levetiracetam Injection 100mg/ML 1 ML 20.65 1687(E) 9.5.2016 368 Levetiracetam Tablet 250 mg 1 Tablet 5.54 1254(E) 29.3.2016 369 Levetiracetam Tablet 500 mg 1 Tablet 11.19 1254(E) 29.3.2016 370 Levetiracetam Tablet 750 mg 1 Tablet 17.19 1254(E) 29.3.2016 371 Levetiracetam Oral Liquid 100mg/ML 1 ML 3.45 2964(E) 15.9.2016 372 Levodopa (A) + Carbidopa (B) Tablet 100mg (A) + 10mg (B) 1 Tablet 1.47 1819(E) 18.5.2016 373 Levodopa (A) + Carbidopa (B) Tablet 250mg (A) + 25 mg (B) 1 Tablet 3.53 2963(E) 15.09.2016 374 Levodopa (A) + Carbidopa (B) CR Tablet 100 mg (A) + 25 mg (B) 1 Tablet 3.16 1254(E) 29.3.2016 375 Levodopa (A) + Carbidopa (B) Tablet 100mg (A) + 25mg (B) 1 Tablet 2.14 1819(E) 18.5.2016 376 Levofloxacin Tablet 750 mg 1 Tablet 10.34 1254(E) 29.3.2016 377 Levofloxacin Tablet 250 mg 1 Tablet 4.17 1254(E) 29.3.2016 378 Levofloxacin Tablet 500 mg 1 Tablet 6.35 1254(E) 29.3.2016 379 Levothyroxine Tablet 100 mcg 1 Tablet 1.13 1254(E) 29.3.2016 380 Levothyroxine Tablet 12.5 mcg 1 Tablet 1.28 1254(E) 29.3.2016 381 Levothyroxine Tablet 125 mcg 1 Tablet 1.45 1254(E) 29.3.2016 382 Levothyroxine Tablet 150mcg 1 Tablet 1.46 1254(E) 29.3.2016 383 Levothyroxine Tablet 25 mcg 1 Tablet 1.24 1254(E) 29.3.2016 384 Levothyroxine Tablet 50 mcg 1 Tablet 0.93 1254(E) 29.3.2016 385 Levothyroxine Tablet 75 mcg 1 Tablet 1.23 1254(E) 29.3.2016 386 Levothyroxine Tablet 88 mcg 1 Tablet 1.45 1254(E) 29.3.2016 387 Lignocaine Injection 2% 1 ML 0.93 3181(E) 7.10.2016 388 Lignocaine Injection 2%(Preservative free for IV use) 1 ML 0.92 443(E) 14.2.2017 389 Lignocaine Injection 1% Each Pack 7.51 3181(E) 7.10.2016 390 Lignocaine Topical forms Injection 2-5% 1 GM or 1 ML 0.99 788(E) 10.03.2017 391 Lignocaine (A) + Adrenaline (B) Injection 2% (A) + 1 ML 0.87 443(E) 14.2.2017
- 11. 1:200000 (5mcg/ML) (B) 392 Linezolid Tablet 600 mg 1 Tablet 31.26 1254(E) 29.3.2016 393 Lithium Tablet 300 mg 1 Tablet 1.38 3181(E) 7.10.2016 394 Loperamide Capsule 2mg 1 Capsule 3.39 1687(E) 9.5.2016 395 Loperamide Tablet 2 mg 1 Tablet 1.87 1404(E) 12.4.2016 396 Lopinavir (A) + Ritonavir (B) Tablet 200 mg (A) + 50 mg (B) 1 Tablet 43.12 1254(E) 29.3.2016 397 Lorazepam injection 2 mg/ML 1 ML 7.36 3431(E) 10.11.2016 398 Lorazepam Tablet 1 mg 1 Tablet 1.96 1254(E) 29.3.2016 399 Lorazepam Tablet 2 mg 1 Tablet 2.38 1254(E) 29.3.2016 400 Mannitol Injection 20% 1 ML 0.28 3431(E) 10.11.2016 401 Mebendazole Oral liquid 100 mg/5 ML 1 ML 0.74 1560(E) 27.4.2016 402 Mebendazole Tablet 100 mg 1 Tablet 2.77 1254(E) 29.3.2016 403 Medroxyprogesteroneacetate Tablet 10 mg 1 Tablet 5.09 2195(E) 23.6.2016 404 Mefenamic acid Oral liquid 100 mg/5 ML 1 ML 0.49 1560(E) 27.4.2016 405 Mefenamic acid Tablet 500 mg 1 Tablet 2.31 4127(E) 22.12.2016 406 Mefenamic acid Tablet 250 mg 1 Tablet 1.82 4127(E) 22.12.2016 407 Mefloquine Tablet 250 mg 1 Tablet 48.24 2195(E) 23.6.2016 408 Melphalan Tablet 2mg 1 Tablet 89.02 2569(E) 29.7.2016 409 Melphalan Tablet 5mg 1 Tablet 152.68 2569(E) 29.7.2016 410 Mesna Injection 100 mg/ML 1 ML 15.22 1560(E) 27.4.2016 411 Metformin Immediate Release Tablet 1000 mg 1 Tablet 3.41 1254(E) 29.3.2016 412 Metformin Controlled Release Tablet 750 mg 1 Tablet 2.27 4127(E) 22.12.2016 413 Metformin Immediate Release Tablet 750 mg 1 Tablet 2.88 4127(E) 22.12.2016 414 Metformin Controlled ReleaseTablet 1000 mg 1 Tablet 3.45 1254(E) 29.3.2016 415 Metformin Controlled ReleaseTablet 500 mg 1 Tablet 1.80 1254(E) 29.3.2016 416 Metformin Tablet (Immediate Release)500mg 1 Tablet 1.42 1951(E) 2.6.2016 417 Methotrexate Injection 25 mg/ ML 1 ML 43.64 1404(E) 12.4.2016 418 Methotrexate Injection 50 mg/ML 1 ML 36.35 4128(E) 22.12.2016 419 Methotrexate Tablet 2.5 mg 1 Tablet 4.34 2195(E) 23.6.2016 420 Methotrexate Tablet 10 mg 1 Tablet 11.02 2195(E) 23.6.2016 421 Methotrexate Tablet 7.5 mg 1 Tablet 10.52 2195(E) 23.6.2016 422 Methotrexate Tablet 5 mg 1 Tablet 7.60 2963(E) 15.09.2016 423 Methyldopa Tablet 250 mg 1 Tablet 2.30 788(E) 10.03.2017 424 Methyldopa Tablet 500 mg 1 Tablet 4.42 787(E) 10.03.2017 425 Methylergometrine Injection 0.2 mg/ML 1 ML 13.50 4128(E) 22.12.2016 426 Methylergometrine Tablet 0.125 mg 1 Tablet 7.49 248(E) 24.01.2017 427 Methylprednisolone Injection 40 mg/ML 1 ML 45.41 248(E) 24.01.2017 428 Methylprednisolone Tablet 16 mg 1 Tablet 8.44 1254(E) 29.3.2016 429 Methylprednisolone Tablet 8 mg 1 Tablet 4.83 1254(E) 29.3.2016 430 Metoclopramide Oral Liquid 5 mg/5ML 1 ML 0.42 3431(E) 10.11.2016 431 Metoclopramide Tablet 10mg 1 Tablet 1.10 2569(E) 29.7.2016 432 Metoprolol Capsule 25mg 1 Capsule 4.00 1687(E) 9.5.2016
- 12. 433 Metoprolol Tablet 25mg 1 Tablet 2.94 1951(E) 2.6.2016 434 Metoprolol Capsule 50mg 1 Capsule 6.10 1687(E) 9.5.2016 435 Metoprolol SR Tablet 25 mg 1 Tablet 3.82 1254(E) 29.3.2016 436 Metoprolol SR Tablet 50 mg 1 Tablet 5.46 1254(E) 29.3.2016 437 Metoprolol Tablet 50mg 1 Tablet 4.45 1951(E) 2.6.2016 438 Metronidazole Injection 500 mg/100 ML 1 ML 0.12276 443(E) 14.2.2017 439 Metronidazole Oral liquid 200 mg/5 ML 1 ML 0.28 3182(E) 7.10.2016 440 Metronidazole Tablet 400 mg 1 Tablet 0.76 1819(E) 18.5.2016 441 Metronidazole Tablet 200 mg 1 Tablet 0.41 2195(E) 23.6.2016 442 Midazolam Injection 5mg/ML 1 ML 13.73 4128(E) 22.12.2016 443 Midazolam Injection 1mg/ML 1 ML 5.59 4128(E) 22.12.2016 444 Mifepristone Tablet 200mg 1 Tablet 304.38 1819(E) 18.5.2016 445 Misoprostol Tablet 100 mcg 1 Tablet 8.24 2730(E) 17.8.2016 446 Misoprostol Tablet 200 mcg 1 Tablet 15.80 1254(E) 29.3.2016 447 Moxifloxacin Tablet 400 mg 1 Tablet 23.97 1254(E) 29.3.2016 448 Mycophenolate mofetil Tablet 250 mg 1 Tablet 37.67 1404(E) 12.4.2016 449 Mycophenolate mofetil Tablet 500 mg 1 Tablet 74.53 1404(E) 12.4.2016 450 N-acetylcysteine Injection 200 mg/ML 1 ML 21.20 4128(E) 22.12.2016 451 N-acetylcysteine Sachet 200 mg 1 GM 7.13 4127(E) 22.12.2016 452 Naloxone injection 0.4 mg/ML 1 ML 82.08 3431(E) 10.11.2016 453 Natamycin Drops 5% 1 ML 21.58 1560(E) 27.4.2016 454 Neostigmine injection 0.5 mg/ML 1 ML 4.12 2730(E) 17.8.2016 455 Neostigmine Tablet 15 mg 1 Tablet 4.41 4128(E) 22.12.2016 456 Nevirapine Oral Liquid 50mg/5ML 1 ML 0.79 4128(E) 22.12.2016 457 Nevirapine Tablet 200 mg 1 Tablet 13.54 1254(E) 29.3.2016 458 Nifedipine Capsule 10 mg 1 Capsule 0.81 4128(E) 22.12.2016 459 Nifedipine Tablet 10 mg 1 Tablet 1.17 2195(E) 23.6.2016 460 Nitrofurantoin Capsule 100 mg 1 Capsule 6.64 3180(E) 7.10.2016 461 Nitrofurantoin Oral liquid 25 mg/5 ML 1 ML 0.77 1560(E) 27.4.2016 462 Nitrofurantoin Tablet 100 mg 1 Tablet 6.84 2730(E) 17.8.2016 463 Norethisterone Tablet 5mg 1 Tablet 4.84 1951(E) 2.6.2016 464 Omeprazole Capsule 10mg 1 Capsule 2.00 248(E) 24.01.2017 465 Omeprazole Tablet 10mg 1 Tablet 4.19 247(E) 24.01.2017 466 Omeprazole Tablet 40 mg 1 Tablet 7.89 3180(E) 7.10.2016 467 Omeprazole Capsule 20mg 1 Capsule 2.36 1819(E) 18.5.2016 468 Omeprazole Capsule 40mg 1 Capsule 4.67 4128(E) 22.12.2016 469 Omeprazole Powder for oral liquid 20mg 1 GM 1.16 247(E) 24.01.2017 470 Omeprazole Tablet 20 mg 1 Tablet 4.66 4127(E) 22.12.2016 471 Ondansetron Injection 2mg/ML 1 ML 5.54 1951(E) 2.6.2016 472 Ondansetron Tablet 4mg 1 Tablet 4.44 1951(E) 2.6.2016 473 Ondansetron Oral liquid 2 mg/5ML 1 ML 1.06 3181(E) 7.10.2016 474 Ondansetron Tablet 8 mg 1 Tablet 9.24 2195(E) 23.6.2016 475 Oral Rehydration Salts As Licensed 1 GM 0.72 1819(E) 18.5.2016 476 Oral Rehydration Salts As Licensed 1 ML 0.12897 1818(E) 18.5.2016 477 Oxytocin Injection 5IU/ML 1 ML 16.69 4128(E) 22.12.2016 478 Paclitaxel Injection 30mg / 5ML 1 ML 211.16 1561 (E) 27.4.2016 479 Paclitaxel Injection 100 mg/16.7 ML 1 ML 211.16 1560(E) 27.4.2016
- 13. 480 Pantoprazole Injection 40mg Each Pack 42.13 1951(E) 2.6.2016 481 Paracetamol injection 150mg/ML Each Pack (2ML) 6.10 248(E) 24.01.2017 482 Paracetamol Oral Liquid 250mg/5ML 1 ML 0.57 2962(E) 15.09.2016 483 Paracetamol Oral Liquid 120 mg/5ML 1 ML 0.50 3430(E) 10.11.2016 484 Paracetamol Oral Liquid 125 mg/5ML 1 ML 0.32 3431(E) 10.11.2016 485 Paracetamol Suppository 80mg Each Suppository 6.16 3431(E) 10.11.2016 486 Paracetamol Suppository 170mg Each Suppository 7.28 3431(E) 10.11.2016 487 Paracetamol Tablet 500mg 1 Tablet 0.85 1951(E) 2.6.2016 488 Paracetamol Tablet 650 mg 1 Tablet 1.74 1254(E) 29.3.2016 489 Paracetamol Oral Liquid 150 mg/5ML 1 ML 0.55 2962(E) 15.09.2016 490 Paracetamol Oral Liquid 100 mg/5ML 1 ML 0.48 2962(E) 15.09.2016 491 Paracetamol Oral Liquid 650 mg/5ML 1 ML 0.49 2962(E) 15.09.2016 492 Paracetamol Oral Liquid 500 mg/5ML 1 ML 0.60 2962(E) 15.09.2016 493 Pegylated interferon alfa 2b Injection 100mcg Each Pack 14,802.08 1687(E) 9.5.2016 494 Pegylated interferon alfa 2b Injection 80mcg Each Pack 11,833.84 1687(E) 9.5.2016 495 Penicillamine Capsule 250mg 1 Capsule 13.49 1686(E) 9.5.2016 496 Permethrin Cream 5% 1 GM 1.65 3181(E) 7.10.2016 497 Permethrin Lotion 1% 1 GM 0.86 248(E) 24.01.2017 498 Pheniramine Injection 22.75 mg/ML 1 ML 1.05 3431(E) 10.11.2016 499 Phenobarbitone Oral Liquid 20mg/5ML 1 ML 0.36 443(E) 14.2.2017 500 Phenobarbitone Injection 200mg/ML 1 ML 18.13 2730(E) 17.8.2016 501 Phenobarbitone Tablet 60mg 1 Tablet 1.67 1951(E) 2.6.2016 502 Phenobarbitone Tablet 30 mg 1 Tablet 1.14 2195(E) 23.6.2016 503 Phenytoin Capsule 300mg 1 Capsule 3.61 247(E) 24.01.2017 504 Phenytoin Capsule 100 mg 1 Capsule 1.44 248(E) 24.01.2017 505 Phenytoin ER Capsule 300mg 1 Capsule 3.61 247(E) 24.01.2017 506 Phenytoin ER Tablet 300mg 1 Tablet 5.50 1254(E) 29.3.2016 507 Phenytoin injection 25 mg/ML 1 ML 2.38 3431(E) 10.11.2016 508 Phenytoin Oral liquid 125 mg/5 ML 1 ML 0.84 247(E) 24.01.2017 509 Phenytoin Oral liquid 30 mg/5 ML 1 ML 0.28 442(E) 14.2.2017 510 Phenytoin Tablet 50mg 1 Tablet 0.71 1686(E) 9.5.2016 511 Phenytoin Tablet 300mg 1 Tablet 4.48 1254(E) 29.3.2016 512 Phenytoin Injection 50 mg/ML 1 ML 5.08 3181(E) 7.10.2016 513 Pilocarpine Drops 2% 1 ML 9.56 3431(E) 10.11.2016 514 Piperacillin (A) + Tazobactam (B) Powder for Injection 1 g (A) + 125 mg (B) Each Pack 83.64 1404(E) 12.4.2016 515 Piperacillin (A) + Tazobactam (B) Powder for Injection 2 g (A) + 250 mg (B) Each Pack 197.86 1404(E) 12.4.2016 516 Piperacillin (A) + Tazobactam (B) Powder for Injection 4 g (A) + 500 mg (B) Each Pack 415.63 1404(E) 12.4.2016 517 Potassium chloride Injection 150 mg/ML 1 ML 2.29 1560(E) 27.4.2016
- 14. 518 Potassium chloride Oral liquid 500 mg/5 ML 1 ML 0.27 1687(E) 9.5.2016 519 Povidone iodine Solution 7.5% 1 ML 0.74 1687(E) 9.5.2016 520 Povidone iodine Solution 4% 1 ML 1.65 247(E) 24.01.2017 521 Povidone iodine Solution 5% 1 ML 0.34 3431(E) 10.11.2016 522 Povidone iodine Solution 10% 1 ML 0.67 443(E) 14.2.2017 523 Pralidoxime chloride (2-PAM) Injection 25 mg/ML 1 ML 3.52 3430(E) 10.11.2016 524 Prednisolone Drops 1% 1 ML 3.18 1561 (E) 27.4.2016 525 Prednisolone Oral liquid 5mg / 5ML 1 ML 0.41 1560(E) 27.4.2016 526 Prednisolone Tablet 5 mg 1 Tablet 0.53 1819(E) 18.5.2016 527 Prednisolone Tablet 10 mg 1 Tablet 0.92 1819(E) 18.5.2016 528 Prednisolone Tablet 20 mg 1 Tablet 1.85 2195(E) 23.6.2016 529 Prednisolone Oral liquid 15mg / 5ML 1 ML 0.72 1560(E) 27.4.2016 530 Premix Insulin 30:70 Injection (Regular:NPH) Injection 40 IU /ML 1 ML 13.66 2193(E) 23.6.2016 531 Prilocaine (A) + Lignocaine (B) Cream 2.5% (A) + 2.5% (B ) 1 GM 19.34 1687(E) 9.5.2016 532 Primaquine Tablet 7.5 mg 1 Tablet 2.00 2730(E) 17.8.2016 533 Primaquine Tablet 15 mg 1 Tablet 4.34 2729(E) 17.8.2016 534 Primaquine Tablet 2.5 mg 1 Tablet 1.47 2963(E) 15.09.2016 535 Procarbazine Tablet 50 mg 1 Tablet 42.74 247(E) 24.01.2017 536 Procarbazine Capsule 50 mg 1 Capsule 31.37 2730(E) 17.8.2016 537 Proparacaine Drops 0.5% 1 ML 9.44 1560(E) 27.4.2016 538 Propofol Injection 10 mg/ML 1 ML 6.83 3181(E) 7.10.2016 539 Propranolol Capsule 80mg 1 Capsule 5.46 1687(E) 9.5.2016 540 Propranolol Capsule 40 mg 1 Capsule 3.60 3180(E) 7.10.2016 541 Propranolol Tablet 40mg 1 Tablet 2.61 444(E) 14.2.2017 542 Propranolol Tablet 80 mg 1 Tablet 4.72 1254(E) 29.3.2016 543 Propranolol Tablet 10 mg 1 Tablet 1.09 2195(E) 23.6.2016 544 Protamine Injection 10mg/ML 1 ML 8.39 3181(E) 7.10.2016 545 Pyrazinamide Tablet 500 mg 1 Tablet 3.84 2195(E) 23.6.2016 546 Pyrazinamide Tablet 750 mg 1 Tablet 5.84 2195(E) 23.6.2016 547 Pyrazinamide Tablet 1000 mg 1 Tablet 8.56 2195(E) 23.6.2016 548 Pyrazinamide Tablet 1500mg 1 Tablet 9.47 2569(E) 29.7.2016 549 Pyridoxine Tablet 100 mg 1 Tablet 5.30 1254(E) 29.3.2016 550 Quinine Injection 300 mg/ML 1 ML 9.22 4128(E) 22.12.2016 551 Quinine Tablet 300 mg 1 Tablet 5.19 2195(E) 23.6.2016 552 Rabies Vaccine Each Pack 310.08 1686(E) 9.5.2016 553 Raltegravir Tablet 400 mg 1 Tablet 142.00 1254(E) 29.3.2016 554 Ramipril Capsule 2.5mg 1 Capsule 4.47 1687(E) 9.5.2016 555 Ramipril Capsule 5mg 1 Capsule 6.99 1687(E) 9.5.2016 556 Ramipril Tablet 2.5 mg 1 Tablet 4.70 1254(E) 29.3.2016 557 Ramipril Tablet 5 mg 1 Tablet 7.42 1254(E) 29.3.2016 558 Ranitidine Injection 25 mg/ML 1 ML 1.51 3431(E) 10.11.2016 559 Ranitidine Oral liquid 75 mg/5 ML 1 ML 0.55 1560(E) 27.4.2016 560 Ranitidine Tablet 150 mg 1 Tablet 0.70 1254(E) 29.3.2016 561 Ribavirin Capsule 200 mg 1 Capsule 75.90 1254(E) 29.3.2016 562 Rifampicin Capsule 600 mg 1 Capsule 10.98 1254(E) 29.3.2016 563 Rifampicin Tablet 300 mg 1 Tablet 2.77 3431(E) 10.11.2016 564 Risperidone Oral liquid 1 mg/ML 1 ML 1.82 1560(E) 27.4.2016
- 15. 565 Risperidone Tablet 2 mg 1 Tablet 4.66 1404(E) 12.4.2016 566 Risperidone Tablet 4 mg 1 Tablet 9.47 1404(E) 12.4.2016 567 Risperidone Tablet 1 mg 1 Tablet 2.91 1404(E) 12.4.2016 568 Ritonavir Tablet 100 mg 1 Tablet 27.45 2729(E) 17.8.2016 569 Ritonavir Capsule 100 mg 1 Capsule 26.70 3431(E) 10.11.2016 570 Rituximab Injection 10 mg/ML 1 ML 717.63 1404(E) 12.4.2016 571 Salbutamol Tablet 2 mg 1 Tablet 0.15131 2730(E) 17.8.2016 572 Salbutamol Capsule 4 mg 1 Capsule 0.57 3430(E) 10.11.2016 573 Salbutamol Inhalation (MDI/DPI) 100 mcg/dose 1 Dose 0.38 443(E) 14.2.2017 574 Salbutamol Oral liquid 2 mg/5 ML 1 ML 0.15 4128(E) 22.12.2016 575 Salbutamol Respirator solution for use in nebulizer 5mg/ML 1 ML 0.67 1687(E) 9.5.2016 576 Salbutamol Tablet 4 mg 1 Tablet 0.17335 2195(E) 23.6.2016 577 Salicylic acid Ointment 6% 1 GM 1.70 1560(E) 27.4.2016 578 Sodium Bicarbonate Injection 8.4% 1 ML 1.22 443(E) 14.2.2017 579 Sodium Bicarbonate Injection 7.5% 1 ML 1.32 443(E) 14.2.2017 580 Sodium Chloride Injection 0.9% 1000ML Glass 56.15 2209(E) 24.6.2016 581 Sodium Chloride Injection 0.9% 1000ML Non- Glass 44.29 2209(E) 24.6.2016 582 Sodium Chloride Injection 0.9% 100ML Glass 16.61 2209(E) 24.6.2016 583 Sodium Chloride Injection 0.9% 100ML Non-Glass 14.96 2209(E) 24.6.2016 584 Sodium Chloride Injection 0.9% 250ML Glass 22.39 2209(E) 24.6.2016 585 Sodium Chloride Injection 0.9% 250ML Non-Glass 20.11 2209(E) 24.6.2016 586 Sodium Chloride Injection 0.9% 500ML Glass 31.12 2209(E) 24.6.2016 587 Sodium Chloride Injection 0.9% 500ML Non-Glass 26.51 2209(E) 24.6.2016 588 Sodium Nitroprusside Injection 10 mg/ML 1 ML 24.42 1819(E) 18.5.2016 589 Sodium valproate Tablet 300 mg 1 Tablet 3.76 1254(E) 29.3.2016 590 Sodium valproate Tablet 200 mg 1 Tablet 2.80 2195(E) 23.6.2016 591 Sodium valproate Tablet 500 mg 1 Tablet 6.56 2195(E) 23.6.2016 592 Sodium Valproate Injection 100 mg/ML 1 ML 5.45 2963(E) 15.09.2016 593 Sodium Valproate Oral Liquid 200mg/5ML 1 ML 0.55 4128(E) 22.12.2016 594 Sofosbuvir Tablet 400 mg 1 Tablet 631.52 1254(E) 29.3.2016 595 Somatostatin Powder for Injection 3 mg Each pack 1,438.04 1404(E) 12.4.2016 596 Spironolactone Tablet 25 mg 1 Tablet 1.80 788(E) 10.03.2017 597 Stavudine (A) + Lamivudine (B) Tablet 30mg (A) +150mg (B) 1 Tablet 9.03 2569(E) 29.7.2016 598 Streptokinase Injection 7,50,000 IU Each Pack 1,191.15 3431(E) 10.11.2016 599 Streptokinase Injection 15,00,000 IU Each Pack 1,693.59 3431(E) 10.11.2016 600 Streptomycin Powder for Injection 750 mg Each Pack 7.97 3181(E) 7.10.2016 601 Streptomycin Powder for Injection 1GM Each Pack 9.46 248(E) 24.01.2017 602 Sucralfate Oral liquid 1 GM 1 ML 0.67 1560(E) 27.4.2016 603 Sulfasalazine Tablet 500 mg 1 Tablet 3.77 1819(E) 18.5.2016 604 Tacrolimus Capsule 2 mg 1 Capsule 72.40 1404(E) 12.4.2016 605 Tamoxifen Tablet 10 mg 1 Tablet 2.44 2730(E) 17.8.2016 606 Tamoxifen 1 Tablet 2.75 2730(E) 17.8.2016 607 Telmisartan Tablet 80 mg 1 Tablet 9.51 1254(E) 29.3.2016
- 16. 608 Telmisartan Tablet 20 mg 1 Tablet 3.45 1254(E) 29.3.2016 609 Telmisartan Tablet 40 mg 1 Tablet 6.24 1254(E) 29.3.2016 610 Temozolomide Capsule 250 mg 1 Capsule 4,434.02 1404(E) 12.4.2016 611 Temozolomide Capsule 100 mg 1 Capsule 1,971.40 1404(E) 12.4.2016 612 Temozolomide Capsule 20 mg 1 Capsule 564.30 1404(E) 12.4.2016 613 Temozolomide Tablet 20mg 1 Tablet 354.54 1687(E) 9.5.2016 614 Temozolomide Tablet 100mg 1 Tablet 1,463.33 1687(E) 9.5.2016 615 Temozolomide Tablet 250mg 1 Tablet 3,621.90 1687(E) 9.5.2016 616 Tenofovir Tablet 300 mg 1 Tablet 44.65 1254(E) 29.3.2016 617 Tenofovir (A) + Lamivudine (B) Tablet 300 mg (A) +300 mg (B) 1 Tablet 40.73 1254(E) 29.3.2016 618 Tenofovir (A) + Lamivudine (B) + Efavirenz (C) Tablet 300 mg (A) + 300 mg (B) + 600 mg (C) 1 Tablet 93.52 1254(E) 29.3.2016 619 Tetanus Toxoid Injection Each Pack (0.5ML) 5.64 248(E) 24.01.2017 620 Tetanus Toxoid Injection Each Pack (5ML) 24.89 248(E) 24.01.2017 621 Thalidomide Capsule 50 mg 1 Capsule 33.40 1404(E) 12.4.2016 622 Thalidomide Capsule 100 mg 1 Capsule 57.80 1404(E) 12.4.2016 623 Thiamine Injection 100 mg/ML 1 ML 23.47 1560(E) 27.4.2016 624 Thiamine Tablet 100 mg 1 Tablet 3.54 4128(E) 22.12.2016 625 Thiopentone Powder for Injection 1 GM Each Pack 46.76 3181(E) 7.10.2016 626 Thiopentone Powder for Injection 0.5 GM Each Pack 38.54 4128(E) 22.12.2016 627 Timolol Drops 0.5% 1 ML 11.28 4128(E) 22.12.2016 628 Timolol Drops 0.25% 1 ML 4.88 3431(E) 10.11.2016 629 Tiotropium Inhalation (DPI) 18 mcg/dose 1 Dose 2.34 1687(E) 9.5.2016 630 Tiotropium Inhalation (MDI) 9 mcg/dose 1 Dose 2.20 1687(E) 9.5.2016 631 Tramadol Tablet 50mg 1 Tablet 7.63 4127(E) 22.12.2016 632 Tramadol Capsule 50 mg 1 Capsule 4.28 2195(E) 23.6.2016 633 Tramadol Tablet 100mg 1 Tablet 18.61 1687(E) 9.5.2016 634 Tranexamic acid Injection 100 mg/ML 1 ML 13.55 1404(E) 12.4.2016 635 Tranexamic acid Tablet 500 mg 1 Tablet 15.76 1254(E) 29.3.2016 636 Trastuzumab Injection 440 mg/50 ML Each Pack 56,912.83 1687(E) 9.5.2016 637 Trihexyphenidyl Tablet 2mg 1 Tablet 1.15 1951(E) 2.6.2016 638 Tropicamide Eye Drop 1% 1 ML 9.52 3431(E) 10.11.2016 639 Vancomycin Powder for Injection 1 GM Each Pack 431.83 4128(E) 22.12.2016 640 Vancomycin Powder for Injection 500 mg Each Pack 259.74 4128(E) 22.12.2016 641 Vecuronium Powder for Injection 10 mg Each Pack 171.56 1560(E) 27.4.2016 642 Vecuronium Powder for Injection 4 mg Each Pack 82.80 1560(E) 27.4.2016 643 Verapamil Injection 2.5 mg/ML 1 ML 1.10 788(E) 10.03.2017 644 Verapamil Tablet 40 mg 1 Tablet 0.71 788(E) 10.03.2017 645 Verapamil Tablet 80 mg 1 Tablet 1.31 788(E) 10.03.2017 646 Vincristine Injection 1 mg/ML 1 ML 47.28 3181(E) 7.10.2016 647 Vitamin A Injection 50000 IU/ML 1 ML 1.80 788(E) 10.03.2017 648 Vitamin A Tablet 50000 IU 1 Tablet 0.67 787(E) 10.03.2017
- 17. 649 Warfarin Tablet 5 mg 1 Tablet 2.24 2195(E) 23.6.2016 650 Warfarin Tablet 1mg 1 Tablet 2.28 1254(E) 29.3.2016 651 Water for Injection Injection Each Pack (10 ML) 2.21 443(E) 14.2.2017 652 Water for Injection Injection Each Pack (5 ML) 2.13 443(E) 14.2.2017 653 Xylometazoline Nasal Drops 0.1% 1 ML 4.92 1560(E) 27.4.2016 654 Xylometazoline Nasal Drops 0.05% 1 ML 3.72 1560(E) 27.4.2016 655 Zidovudine Tablet 300 mg 1 Tablet 12.94 2569(E) 29.7.2016 656 Zidovudine Capsule 300 mg 1 Capsule 1.88 247(E) 24.01.2017 657 Zidovudine (A) + Lamivudine (B) + Nevirapine (C) Tablet 300 mg (A) + 150 mg (B) + 200 mg(C) 1 Tablet 19.06 248(E) 24.01.2017 658 Zolpidem Capsule 5mg 1 Capsule 6.96 442(E) 14.2.2017 659 Zolpidem Tablet 10 mg 1 Tablet 7.99 1404(E) 12.4.2016 660 Zolpidem Tablet 5 mg 1 Tablet 5.09 1404(E) 12.4.2016 Notes:- (a) The ceiling prices are applicable with effect from 01.4.2017 (ceiling prices are inclusive of Wholesale Price Index (WPI) @1.97186% for the year 2016 over 2015). (b) In respect of Gentamycin injection 40mg/ml, Dexamethasone injection 4mg/ml, Paracetamol injection 150mg/ml, Glucose Injection 5%, Sodium chloride Injection 0.9%, Glucose + Sodium chloride Injection 5%+0.9%, Water for Injection and Tetanus Toxoid injection, for any other pack size manufactured, the manufacturer shall approach NPPA for specific price approval for its formulations. (c) In respect of any other scheduled formulation, for which ceiling price is not mentioned above or in S.O. 1008, 1009, 1010 dated 30.3.2017 or in S.O 412(E) dated 13.2.2017, the manufacturer shall approach the NPPA for specific price approval for its formulations. (d) All manufacturers of scheduled formulations, selling branded or generic or both the versions of scheduled formulations at price higher than the ceiling price (plus local taxes as applicable) so fixed and notified by the Government, shall revise the prices of all such formulations downward not exceeding the ceiling price specified in column (5) in the above table plus local taxes as applicable, if any. (e) All the existing manufacturers of above mentioned scheduled formulations having MRP lower than the ceiling price specified in column (5) in the above table (plus local taxes as applicable, if any), may revise the existing M.R.P. of their formulations, on the basis of WPI @ 1.97186% for year 2016 in accordance with paragraph 16(2) of DPCO, 2013, read with para 13(2) of DPCO, 2013. (f) The manufacturers may add local taxes only if they have paid actually or if it is payable to the Government on the ceiling price mentioned in column (5) of the above said table. (g) The ceiling price for a pack of the scheduled formulation shall be arrived at by the concerned manufacturer in accordance with the ceiling price specified in column (5) of the above table as per provisions contained in paragraph 11 of the Drugs (Prices Control) Order, 2013. The manufacturer shall issue a price list in Form–V from date of Notification as per paragraph 24 of the DPCO, 2013 to NPPA through IPDMS and submit a copy to State Drug Controller and dealers. (h) As per para 24(4) of DPCO 2013, every retailer and dealer shall display price list and the supplementary price list, if any, as furnished by the manufacturer, on a conspicuous part of the premises where he carries on business in a manner so as to be easily accessible to any person wishing to consult the same. (i) Where an existing manufacturer of scheduled formulation with dosage or strength or both as specified in the above table launches a new drug as per paragraph 2 (u) of the DPCO, 2013 such existing manufacturer shall apply for prior price approval of such new drug to the NPPA in Form I as specified under Schedule-II of the DPCO, 2013.
- 18. (j) The manufacturers of above said scheduled formulations shall furnish quarterly return to the NPPA, in respect of production / import and sale of scheduled formulations in Form-III of Schedule-II of the DPCO, 2013 through IPDMS. Any manufacturer intending to discontinue production of above said scheduled formulation shall furnish information to the NPPA, in respect of discontinuation of production and / or import of scheduled formulation in Form-IV of Schedule-II of the DPCO, 2013 at least six months prior to the intended date of discontinuation. (k) The manufacturers not complying with the ceiling price and notes specified hereinabove shall be liable to deposit the overcharged amount along with interest thereon under the provisions of the Drugs (Prices Control) Order, 2013 read with Essential Commodities Act, 1955. (l) Consequent to the issue of ceiling prices of such formulations as specified in column (2) of the above table in this notification, the price order(s) fixing ceiling or retail price, if any, issued prior to the above said date of notification, stand(s) superseded. PN/175/43/2017/F F. No. 8(43)/2017/D.P./NPPA-Div.-II (BALJIT SINGH) Assistant Director
