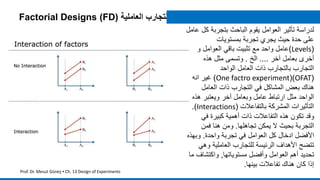
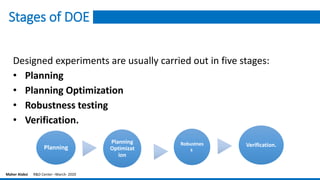
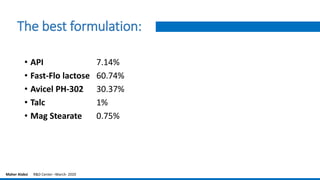
The document provides an overview of the design of experiments (DOE) methodology, covering its history, principles, and applications in product development and quality improvement. It discusses the advantages and disadvantages of DOE compared to traditional methods, as well as terminology and key concepts such as factors, interaction effects, randomization, and replication. Furthermore, it outlines how to select appropriate experimental designs based on objectives and the number of factors involved.


































![The Response surface design (RSD) and factorial designs (FD) are the
most commonly employed designs in pharmaceutical industry. [1]
- The Box-behnken design (BBD) is the most popular among all response surface
methodology (RSM) because it requires fewer runs in 3 factor experimental design than all
other RSM designs [1]
Which design used in pharmaceutical industry?
1.Wang F, Chen L, Jiang S, He J, Zhang X, Peng J, J Liposome Res, 2014, 24, 171.
Optimization Strategies of Experimental Designs [8]
There are various designs and plots are available in DoE to obtain an optimized formulation. The most widely
used designs in pharmaceutical applications are RSM and FD, both of which serve different purposes. The best
criteria to select a design is that which can give an optimized formulation in fewer runs that in turn saves time as
well as money.](https://image.slidesharecdn.com/designofexperimentsformulationdevelopmentexploringthebestpracticesdoe-200302065615/85/Design-of-experiments-formulation-development-exploring-the-best-practices-doe-40-320.jpg)