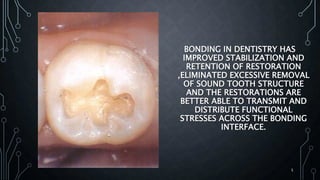
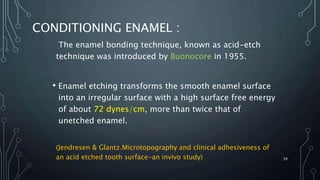
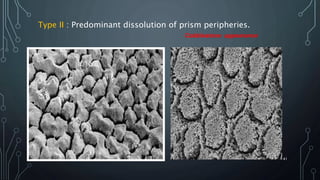
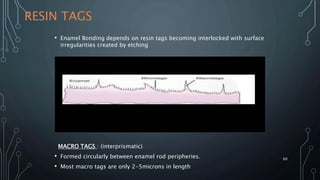
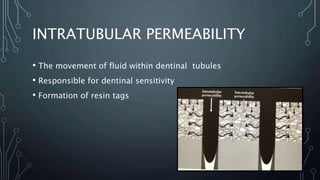
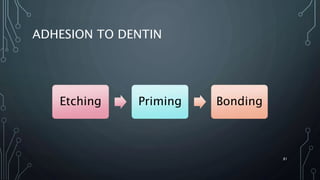
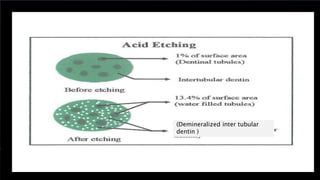
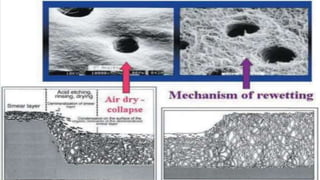
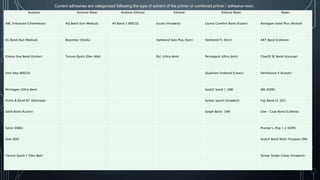
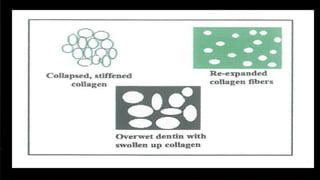
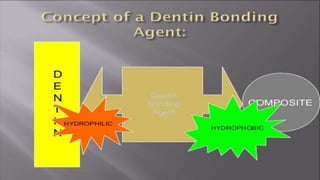
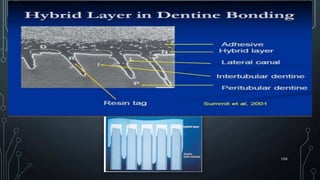
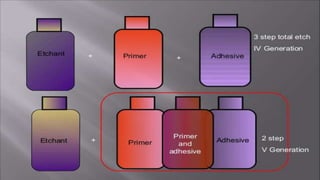
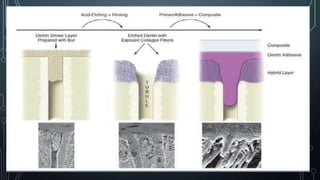
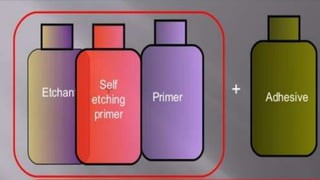
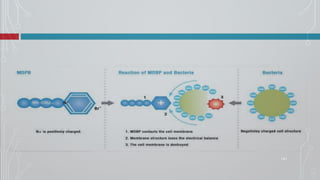
The document discusses dentin bonding agents and adhesion to tooth structures. It begins with an introduction on the history and advantages of adhesive dentistry. It then discusses the composition and structure of enamel and dentin, and how adhesion occurs to each. For enamel, acid etching is used to condition the surface and increase bonding. For dentin, multiple generations of dentin bonding agents have been developed to facilitate adhesion. The document covers topics like mechanisms of bonding, classifications of agents, and challenges of the bonding procedure.