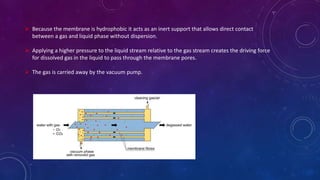
Degassing, or degasification, is the process of removing dissolved gases from liquids, particularly in applications like drug production and painting. Methods include membrane and thermal degassing, with membrane systems utilizing a gas-permeable membrane to remove gases efficiently without chemical consumption. Thermal degassing, on the other hand, leverages high temperatures to reduce gas solubility in water, employing various technologies like spray and tray degassing for effective gas removal.