Computational and Experimental Studies of MTO Catalyzed Olefin Hydrogenation
•
1 like•77 views
The poster that I presented at the 253rd American Chemical Society National Meeting and Exposition in San Francisco, CA. It highlights some of my REU research at North Carolina State University under the mentorship of Dr. Elon Ison.
Report
Share
Report
Share
Download to read offline
Recommended
CcPK2_Biochem_1999

The document summarizes research on the effects of engineering a cation binding site in cytochrome c peroxidase (CCP). Key findings include:
1) Introducing the cation binding site found in ascorbate peroxidase (APX) into CCP (creating the CCPK2 mutant) results in potassium binding at this site.
2) Binding of potassium at the engineered site in CCPK2 leads to a dramatic decrease in enzyme activity and weakening of the characteristic electron paramagnetic resonance (EPR) signal associated with the CCP compound I Trp191 radical.
3) These results indicate that the bound potassium ion destabilizes the Trp191 radical in C
ja5101036

This document summarizes experiments investigating the roles of various organic additives in facilitating coupling reactions between haloarenes and arenes using potassium tert-butoxide (KOtBu). Amino acids like sarcosine and proline were found to initiate these reactions via electron transfer, forming aryl radicals. Secondary amino acids were more effective than primary or tertiary amino acids. Piperazinedione derivatives formed from the condensation of amino acids were also found to initiate reactions by forming electron-rich enolates or dianions that act as electron donors. Alcohols, 1,2-diols, and 1,2-diamines were also investigated for their ability to form electron donors that initiate these coupling reactions.
Chapter 9: Respiration

KEY CONCEPTS
9.1 Catabolic pathways yield energy by oxidizing organic
fuels
9.2 Glycolysis harvests chemical energy by oxidizing glucose to pyruvate
9.3 After pyruvate is oxidized, the citric acid cycle completes the energy-yielding oxidation of organic molecules
9.4 During oxidative phosphorylation, chemiosmosis couples electron transport to ATP synthesis
9.5 Fermentation and anaerobic respiration enable cells to
produce ATP without the use of oxygen
9.6 Glycolysis and the citric acid cycle connect to many other metabolic pathways
H52YCcP_JMB_2003.PDF

This study examines the crystal structure of a cytochrome c peroxidase mutant where the distal catalytic histidine 52 is converted to tyrosine. The crystal structure reveals an unprecedented covalent bond between the indole nitrogen of tryptophan 51 and the phenyl group of tyrosine 52. The authors hypothesize that this novel cross-link results from peroxide activation by the heme iron, followed by oxidation of tryptophan 51 and tyrosine 52. Testing of this hypothesis by incorporating a redox-inactive zinc-protoporphyrin, which showed the absence of the cross-link, supports that the cross-link is a peroxide-dependent process mediated by the heme iron. Additional experiments treating heme-containing
N195P_Biochem_2002

This document summarizes research on engineering a cation-binding site into cytochrome c peroxidase (CcP) in order to study the effects on enzyme activity and structure. A key residue (Asn195) in the engineered cation-binding loop was mutated to proline to stabilize the loop conformation. Crystal structure analysis showed the loop is stabilized in the closed conformation when potassium is bound. While enzyme activity is reduced, it can be titrated based on potassium concentration. The goals were to better understand how cation binding and loop conformation impact electron transfer from cytochrome c and the stability of the tryptophan radical in the enzyme's active site.
1. elimination reaction in brief

Elimination reactions can occur by either E1 or E2 mechanisms. In an E1 reaction, the rate-determining step is the unimolecular formation of a carbocation intermediate. In an E2 reaction, the rate depends on both the substrate and base concentrations, and it involves a single concerted step without an intermediate. The E1 pathway favors more stable carbocation intermediates and products, while the E2 transition state leads directly from starting material to product.
Reductive Elimination

Reductive elimination is an elementary step where the metal's coordination number and oxidation state both decrease as a new covalent bond is formed. It is the reverse of oxidative addition. Reductive elimination is more common for metals in higher oxidation states. For reductive elimination to occur, the eliminating groups must be cis-oriented and there must be a high formal positive charge on the metal. Reductive elimination finds applications in important catalytic reactions like hydrogenation and hydroformylation.
JB REU Report

The document summarizes a study that analyzed 4 genetically engineered strains of Clostridium thermocellum with differing pathways for converting phosphoenolpyruvate (PEP) to pyruvate. The strains were cultured and their metabolite concentrations and reversibility of reactions were measured to calculate free energy changes. Strain 1138 diverted flux through the malate shunt, strain 1163 expressed an exogenous pyruvate kinase, and strain 1251 expressed pyruvate kinase and deleted PPDK and the malate shunt. Strain 1163 had higher GDP and GTP, suggesting its PEP to oxaloacetate conversion was more efficient. Further optimizing the PEP to pyruvate pathway, such as
Recommended
CcPK2_Biochem_1999

The document summarizes research on the effects of engineering a cation binding site in cytochrome c peroxidase (CCP). Key findings include:
1) Introducing the cation binding site found in ascorbate peroxidase (APX) into CCP (creating the CCPK2 mutant) results in potassium binding at this site.
2) Binding of potassium at the engineered site in CCPK2 leads to a dramatic decrease in enzyme activity and weakening of the characteristic electron paramagnetic resonance (EPR) signal associated with the CCP compound I Trp191 radical.
3) These results indicate that the bound potassium ion destabilizes the Trp191 radical in C
ja5101036

This document summarizes experiments investigating the roles of various organic additives in facilitating coupling reactions between haloarenes and arenes using potassium tert-butoxide (KOtBu). Amino acids like sarcosine and proline were found to initiate these reactions via electron transfer, forming aryl radicals. Secondary amino acids were more effective than primary or tertiary amino acids. Piperazinedione derivatives formed from the condensation of amino acids were also found to initiate reactions by forming electron-rich enolates or dianions that act as electron donors. Alcohols, 1,2-diols, and 1,2-diamines were also investigated for their ability to form electron donors that initiate these coupling reactions.
Chapter 9: Respiration

KEY CONCEPTS
9.1 Catabolic pathways yield energy by oxidizing organic
fuels
9.2 Glycolysis harvests chemical energy by oxidizing glucose to pyruvate
9.3 After pyruvate is oxidized, the citric acid cycle completes the energy-yielding oxidation of organic molecules
9.4 During oxidative phosphorylation, chemiosmosis couples electron transport to ATP synthesis
9.5 Fermentation and anaerobic respiration enable cells to
produce ATP without the use of oxygen
9.6 Glycolysis and the citric acid cycle connect to many other metabolic pathways
H52YCcP_JMB_2003.PDF

This study examines the crystal structure of a cytochrome c peroxidase mutant where the distal catalytic histidine 52 is converted to tyrosine. The crystal structure reveals an unprecedented covalent bond between the indole nitrogen of tryptophan 51 and the phenyl group of tyrosine 52. The authors hypothesize that this novel cross-link results from peroxide activation by the heme iron, followed by oxidation of tryptophan 51 and tyrosine 52. Testing of this hypothesis by incorporating a redox-inactive zinc-protoporphyrin, which showed the absence of the cross-link, supports that the cross-link is a peroxide-dependent process mediated by the heme iron. Additional experiments treating heme-containing
N195P_Biochem_2002

This document summarizes research on engineering a cation-binding site into cytochrome c peroxidase (CcP) in order to study the effects on enzyme activity and structure. A key residue (Asn195) in the engineered cation-binding loop was mutated to proline to stabilize the loop conformation. Crystal structure analysis showed the loop is stabilized in the closed conformation when potassium is bound. While enzyme activity is reduced, it can be titrated based on potassium concentration. The goals were to better understand how cation binding and loop conformation impact electron transfer from cytochrome c and the stability of the tryptophan radical in the enzyme's active site.
1. elimination reaction in brief

Elimination reactions can occur by either E1 or E2 mechanisms. In an E1 reaction, the rate-determining step is the unimolecular formation of a carbocation intermediate. In an E2 reaction, the rate depends on both the substrate and base concentrations, and it involves a single concerted step without an intermediate. The E1 pathway favors more stable carbocation intermediates and products, while the E2 transition state leads directly from starting material to product.
Reductive Elimination

Reductive elimination is an elementary step where the metal's coordination number and oxidation state both decrease as a new covalent bond is formed. It is the reverse of oxidative addition. Reductive elimination is more common for metals in higher oxidation states. For reductive elimination to occur, the eliminating groups must be cis-oriented and there must be a high formal positive charge on the metal. Reductive elimination finds applications in important catalytic reactions like hydrogenation and hydroformylation.
JB REU Report

The document summarizes a study that analyzed 4 genetically engineered strains of Clostridium thermocellum with differing pathways for converting phosphoenolpyruvate (PEP) to pyruvate. The strains were cultured and their metabolite concentrations and reversibility of reactions were measured to calculate free energy changes. Strain 1138 diverted flux through the malate shunt, strain 1163 expressed an exogenous pyruvate kinase, and strain 1251 expressed pyruvate kinase and deleted PPDK and the malate shunt. Strain 1163 had higher GDP and GTP, suggesting its PEP to oxaloacetate conversion was more efficient. Further optimizing the PEP to pyruvate pathway, such as
EAS

Electrophilic aromatic substitution is a reaction where an atom attached to an aromatic system is replaced by an electrophile. The aromatic ring attacks the electrophile, forming a carbocation intermediate. This intermediate is stabilized by resonance. A Lewis base then donates electrons back to the ring, restoring aromaticity. Substituents can activate or deactivate the ring by donating or withdrawing electron density. Activating groups make the reaction more likely and direct substitution to the ortho- and para- positions, while deactivating groups have the opposite effects.
Organometallic compounds

This document summarizes key concepts in organometallic chemistry. It discusses the definition of organometallic compounds as those containing metal-carbon bonds. It outlines different types of ligands that can bind to metals, including carbonyl, carbene, and cyclic π systems. It also describes principles for understanding bonding interactions between ligands and metals, such as the 18-electron rule and molecular orbital theory. Spectroscopic techniques for analyzing organometallic compounds are also summarized.
Aza Cope Rearrangement of Propargyl Enammonium Cations Catalyzed by a Self-As...

Dr. Kenneth Raymond is a renowned chemist who has received numerous awards and honors over his career. His research focuses on catalyzing the Aza Cope rearrangement of propargyl enammonium cations using a self-assembled "nanozyme" made of gallium ions and ligands. The nanozyme is able to accelerate the rearrangement reaction rate by facilitating a more negative transition state entropy compared to the uncatalyzed reaction. Analysis of reaction kinetics shows the nanozyme displays Michaelis-Menten behavior.
Amine rich nitrogen doped carbon dots

This document summarizes research on using amine-rich nitrogen-doped carbon nanodots (NCNDs) as a co-reactant platform for electrochemiluminescence (ECL). The NCNDs were found to enhance the ECL signal of ruthenium tris(bipyridine) through their primary and tertiary amino groups acting as co-reactants in the ECL process. Methylated NCNDs, with tertiary amino groups, showed an even higher ECL signal than unmodified NCNDs. Additionally, a covalently linked hybrid of NCNDs and ruthenium tris(bipyridine) exhibited self-enhanced ECL, with the NCND
Daltons DYME Presentation - Beyond Al: Group 13 Salphen catalysts for efficie...

This was the presentation given at the RSC Dalton's DYME meeting on 29/06/2021. The background of the presentation is using Group 13 Salphen catalysts for the cycloaddition between terminal epoxides and CO2 to form cyclic carbonates.
Oxidation and reduction

1. The document discusses oxidation-reduction reactions and key concepts like oxidation, reduction, and oxidation numbers. It also defines important coenzymes like NAD, FAD, and ATP that are involved in cellular respiration.
2. Key details are provided about the structures and roles of NAD, FAD, and ATP in metabolism. NAD and FAD act as electron carriers in redox reactions and the citric acid cycle. ATP is discussed as the main energy currency molecule in cells.
3. The pathways of glycolysis, the Krebs cycle, the electron transport chain, and oxidative phosphorylation are summarized in relation to the roles of NAD, FAD, and ATP production.
Elimination reaction exercises v3.2

A fully interactive version of this presentation with functioning navigation buttons can be found by clicking on the link below:
https://connect.csupomona.edu/eliminations
Organometallic Reactions and Catalysis

Organometallic compounds undergo a rich variety of reactions (oxidative addition, reductive elimination, cyclometalization, migratory insertion, carbonylation, hydrometallation hydrate elimination, etc ) that can sometimes be combined into useful homogeneous catalytic cycles. In this presentation, I have discussed organometallic reactions of particular importance for synthetic and catalytic processes like the oxo process (hydroformylation), heck coupling reaction, Wilkinson’s Catalyst
(Hydrogenation) etc.
Postdoctoral Research Summary

Adam B. Powell developed a heterogeneous catalyst composed of palladium, bismuth nitrate, and tellurium metal that promotes the aerobic oxidative esterification of aliphatic alcohols with high yields. The addition of bismuth and tellurium additives significantly increased the rate of product formation and overall yield compared to the catalyst without additives. The catalyst was shown to esterify a variety of activated and aliphatic alcohols, expanding the scope of this transformation. Future work includes adapting the catalyst for other oxidative reactions and developing a robust Pd-Bi-Te catalyst for flow applications.
Reductive elimination reactions

this file may contain
1. Definition
2. Mechanism
3. Examples
4. Application
5. References
Organic mechanisms

The document summarizes various organic reaction mechanisms including:
1) Free radical substitution, electrophilic addition, nucleophilic substitution, elimination, addition-elimination, electrophilic substitution, esterification, alkaline hydrolysis, nucleophilic addition.
2) Specific mechanisms are described for hydration of alkenes, addition polymerization, bromination of alkenes, nucleophilic substitution, elimination, dehydration, esterification.
3) The formation of polymers like polyamides, polyesters through reactions of dibasic acids and diamines or diols are summarized.
Advanced organic chemistry

This document discusses elimination reactions, specifically E1 and E2 reactions. It explains that E1 reactions proceed through a carbocation intermediate and involve a two-step mechanism, while E2 reactions are concerted and involve both the alkyl halide and base in a single step. It also describes factors that influence the reactivity and selectivity of elimination reactions, such as substrate structure, the nature of the leaving group and base, and conformational effects.
Biochemistry 304 2014 student edition enzymes and enzyme kinetics

Enzyme kinetics and the mechanisms of enzyme catalysis are described. Key points include:
1) Enzymes lower the activation energy of biochemical reactions, increasing rates up to billions of times faster than uncatalyzed reactions. This is achieved through various catalytic mechanisms including acid-base, covalent, and metal ion catalysis.
2) Michaelis-Menten kinetics describe enzyme-catalyzed reactions, relating reaction velocity to substrate concentration. The Michaelis constant Km and maximum velocity Vmax are important parameters.
3) Different kinetic approaches like rapid equilibrium and steady state are used to derive rate equations depending on if reaction steps are at equilibrium. Rate equations can be plotted and analyzed to determine
Molecular rearrangements

Deals with definitions, types and mechanistic aspects of rearrangements.
Carbocations, Nitreens, Migration of groups and migratory apptitude
isotope effect and fragmantation

Isotopes are two atoms of the same element that have the same number of protons but different numbers of neutrons. Isotopes are specified by the mass number.
Chapter 7 Alkenes and Alkyne 

1) Alkenes are hydrocarbons that contain a carbon-carbon double bond. They include many naturally occurring compounds and important industrial materials.
2) The degree of unsaturation relates the molecular formula to possible structures by counting the number of multiple bonds or rings. Each double bond or ring replaces two hydrogens.
3) Alkenes react through electrophilic addition reactions, often involving a carbocation intermediate. The stability of the carbocation predicts the orientation of addition.
Electrophilic aromatic substitution reactions

This is Power Point Presentation on Topic "Electrophilic Aromatic Substitution Reactions" as per syllabus of "University of Mumbai" for S.Y. B. Pharmacy (Sem.: IV) students.
Aromatic electrophilic substitution

This document provides an overview of aromatic electrophilic substitution reactions (AES). It defines important terms like arenium ions, electrophiles, nucleophiles and discusses the effects of substituents on reactivity. The mechanisms of common AES reactions like nitration, sulfonation, Friedel-Crafts alkylation and acylation are covered. The document also discusses topics like the mesomeric and inductive effects of substituents, the synthesis of tribromobenzene, and the relative reactivities of benzene and substituted benzenes in bromination. Examples of AES on phenols, xylenes, cresols and other aromatic compounds are provided.
Thesis-abstract

This doctoral thesis uses computational methods like density functional theory and molecular dynamics simulations to study the structural and functional role of cytochrome P450 enzymes. It investigates the metabolism of various substrates by CYP3A4 and CYP450 enzymes to understand reaction pathways and influence of substrate structure on reactivity. Specific reactions studied include hydroxylation of phenyl rings, morpholine rings, and camphor. Flexibility studies using the RIGIX program also examined how the protein environment modulates electronic structure and reactivity. The research provides new insights into CYP450 catalysis at the molecular level and could aid in drug design.
Michael Ludden L3Report2016

This document describes Michael Ludden's synthesis and characterization of various molybdenum complexes. Three complexes were synthesized - [CpMo(CO)3Me], [CpMo(CO)3Et], and [CpMo(CO)2(COMe)(PPh3)]. They were characterized using NMR and IR spectroscopy. The results confirmed the structures of the complexes and showed how changing ligands affects properties. Kinetic measurements of migratory insertion reactions will be taken using these complexes to understand reaction rate dependence on factors like solvent, temperature and ligand type.
OBC epoxidations paper - Queen Mary University LONDON UK - Thomas Follier

This document reports on a study of the catalytic activity of manganese complexes with two similar polyamine ligands (7 and 8) that differ by the presence of a secondary or tertiary nitrogen, in the epoxidation of styrene. Ligand 7 showed the highest activity with MnSO4 and H2O2, while ligand 8 was most effective with Mn(OTf)2, MnCl2, and Mn(ClO4)2 using peracetic acid. Kinetic analysis indicated the structural differences in the ligands lead to differences in the nature of the active species formed. Ligand 7 with MnSO4 produced the epoxide in 78% yield, while ligand 8 with Mn(OT
Thesis-PerLindecrantz-2009

This document describes research into direct aldol reactions mediated by dimethylzinc. Three aromatic aldol products were synthesized and their NMR data reported. Racemic aldol products were produced using either dialkylzinc/dialcohol or magnesium bromide/DIPEA. Successful asymmetric and autocatalytic aldol reactions were also achieved using chiral catalysts. The products of some aldol reactions were found to facilitate their own formation and the reaction between other substrates, demonstrating autocatalytic properties. Further study of this autocatalytic process is warranted.
More Related Content
What's hot
EAS

Electrophilic aromatic substitution is a reaction where an atom attached to an aromatic system is replaced by an electrophile. The aromatic ring attacks the electrophile, forming a carbocation intermediate. This intermediate is stabilized by resonance. A Lewis base then donates electrons back to the ring, restoring aromaticity. Substituents can activate or deactivate the ring by donating or withdrawing electron density. Activating groups make the reaction more likely and direct substitution to the ortho- and para- positions, while deactivating groups have the opposite effects.
Organometallic compounds

This document summarizes key concepts in organometallic chemistry. It discusses the definition of organometallic compounds as those containing metal-carbon bonds. It outlines different types of ligands that can bind to metals, including carbonyl, carbene, and cyclic π systems. It also describes principles for understanding bonding interactions between ligands and metals, such as the 18-electron rule and molecular orbital theory. Spectroscopic techniques for analyzing organometallic compounds are also summarized.
Aza Cope Rearrangement of Propargyl Enammonium Cations Catalyzed by a Self-As...

Dr. Kenneth Raymond is a renowned chemist who has received numerous awards and honors over his career. His research focuses on catalyzing the Aza Cope rearrangement of propargyl enammonium cations using a self-assembled "nanozyme" made of gallium ions and ligands. The nanozyme is able to accelerate the rearrangement reaction rate by facilitating a more negative transition state entropy compared to the uncatalyzed reaction. Analysis of reaction kinetics shows the nanozyme displays Michaelis-Menten behavior.
Amine rich nitrogen doped carbon dots

This document summarizes research on using amine-rich nitrogen-doped carbon nanodots (NCNDs) as a co-reactant platform for electrochemiluminescence (ECL). The NCNDs were found to enhance the ECL signal of ruthenium tris(bipyridine) through their primary and tertiary amino groups acting as co-reactants in the ECL process. Methylated NCNDs, with tertiary amino groups, showed an even higher ECL signal than unmodified NCNDs. Additionally, a covalently linked hybrid of NCNDs and ruthenium tris(bipyridine) exhibited self-enhanced ECL, with the NCND
Daltons DYME Presentation - Beyond Al: Group 13 Salphen catalysts for efficie...

This was the presentation given at the RSC Dalton's DYME meeting on 29/06/2021. The background of the presentation is using Group 13 Salphen catalysts for the cycloaddition between terminal epoxides and CO2 to form cyclic carbonates.
Oxidation and reduction

1. The document discusses oxidation-reduction reactions and key concepts like oxidation, reduction, and oxidation numbers. It also defines important coenzymes like NAD, FAD, and ATP that are involved in cellular respiration.
2. Key details are provided about the structures and roles of NAD, FAD, and ATP in metabolism. NAD and FAD act as electron carriers in redox reactions and the citric acid cycle. ATP is discussed as the main energy currency molecule in cells.
3. The pathways of glycolysis, the Krebs cycle, the electron transport chain, and oxidative phosphorylation are summarized in relation to the roles of NAD, FAD, and ATP production.
Elimination reaction exercises v3.2

A fully interactive version of this presentation with functioning navigation buttons can be found by clicking on the link below:
https://connect.csupomona.edu/eliminations
Organometallic Reactions and Catalysis

Organometallic compounds undergo a rich variety of reactions (oxidative addition, reductive elimination, cyclometalization, migratory insertion, carbonylation, hydrometallation hydrate elimination, etc ) that can sometimes be combined into useful homogeneous catalytic cycles. In this presentation, I have discussed organometallic reactions of particular importance for synthetic and catalytic processes like the oxo process (hydroformylation), heck coupling reaction, Wilkinson’s Catalyst
(Hydrogenation) etc.
Postdoctoral Research Summary

Adam B. Powell developed a heterogeneous catalyst composed of palladium, bismuth nitrate, and tellurium metal that promotes the aerobic oxidative esterification of aliphatic alcohols with high yields. The addition of bismuth and tellurium additives significantly increased the rate of product formation and overall yield compared to the catalyst without additives. The catalyst was shown to esterify a variety of activated and aliphatic alcohols, expanding the scope of this transformation. Future work includes adapting the catalyst for other oxidative reactions and developing a robust Pd-Bi-Te catalyst for flow applications.
Reductive elimination reactions

this file may contain
1. Definition
2. Mechanism
3. Examples
4. Application
5. References
Organic mechanisms

The document summarizes various organic reaction mechanisms including:
1) Free radical substitution, electrophilic addition, nucleophilic substitution, elimination, addition-elimination, electrophilic substitution, esterification, alkaline hydrolysis, nucleophilic addition.
2) Specific mechanisms are described for hydration of alkenes, addition polymerization, bromination of alkenes, nucleophilic substitution, elimination, dehydration, esterification.
3) The formation of polymers like polyamides, polyesters through reactions of dibasic acids and diamines or diols are summarized.
Advanced organic chemistry

This document discusses elimination reactions, specifically E1 and E2 reactions. It explains that E1 reactions proceed through a carbocation intermediate and involve a two-step mechanism, while E2 reactions are concerted and involve both the alkyl halide and base in a single step. It also describes factors that influence the reactivity and selectivity of elimination reactions, such as substrate structure, the nature of the leaving group and base, and conformational effects.
Biochemistry 304 2014 student edition enzymes and enzyme kinetics

Enzyme kinetics and the mechanisms of enzyme catalysis are described. Key points include:
1) Enzymes lower the activation energy of biochemical reactions, increasing rates up to billions of times faster than uncatalyzed reactions. This is achieved through various catalytic mechanisms including acid-base, covalent, and metal ion catalysis.
2) Michaelis-Menten kinetics describe enzyme-catalyzed reactions, relating reaction velocity to substrate concentration. The Michaelis constant Km and maximum velocity Vmax are important parameters.
3) Different kinetic approaches like rapid equilibrium and steady state are used to derive rate equations depending on if reaction steps are at equilibrium. Rate equations can be plotted and analyzed to determine
Molecular rearrangements

Deals with definitions, types and mechanistic aspects of rearrangements.
Carbocations, Nitreens, Migration of groups and migratory apptitude
isotope effect and fragmantation

Isotopes are two atoms of the same element that have the same number of protons but different numbers of neutrons. Isotopes are specified by the mass number.
Chapter 7 Alkenes and Alkyne 

1) Alkenes are hydrocarbons that contain a carbon-carbon double bond. They include many naturally occurring compounds and important industrial materials.
2) The degree of unsaturation relates the molecular formula to possible structures by counting the number of multiple bonds or rings. Each double bond or ring replaces two hydrogens.
3) Alkenes react through electrophilic addition reactions, often involving a carbocation intermediate. The stability of the carbocation predicts the orientation of addition.
Electrophilic aromatic substitution reactions

This is Power Point Presentation on Topic "Electrophilic Aromatic Substitution Reactions" as per syllabus of "University of Mumbai" for S.Y. B. Pharmacy (Sem.: IV) students.
Aromatic electrophilic substitution

This document provides an overview of aromatic electrophilic substitution reactions (AES). It defines important terms like arenium ions, electrophiles, nucleophiles and discusses the effects of substituents on reactivity. The mechanisms of common AES reactions like nitration, sulfonation, Friedel-Crafts alkylation and acylation are covered. The document also discusses topics like the mesomeric and inductive effects of substituents, the synthesis of tribromobenzene, and the relative reactivities of benzene and substituted benzenes in bromination. Examples of AES on phenols, xylenes, cresols and other aromatic compounds are provided.
What's hot (18)
Aza Cope Rearrangement of Propargyl Enammonium Cations Catalyzed by a Self-As...

Aza Cope Rearrangement of Propargyl Enammonium Cations Catalyzed by a Self-As...
Daltons DYME Presentation - Beyond Al: Group 13 Salphen catalysts for efficie...

Daltons DYME Presentation - Beyond Al: Group 13 Salphen catalysts for efficie...
Biochemistry 304 2014 student edition enzymes and enzyme kinetics

Biochemistry 304 2014 student edition enzymes and enzyme kinetics
Similar to Computational and Experimental Studies of MTO Catalyzed Olefin Hydrogenation
Thesis-abstract

This doctoral thesis uses computational methods like density functional theory and molecular dynamics simulations to study the structural and functional role of cytochrome P450 enzymes. It investigates the metabolism of various substrates by CYP3A4 and CYP450 enzymes to understand reaction pathways and influence of substrate structure on reactivity. Specific reactions studied include hydroxylation of phenyl rings, morpholine rings, and camphor. Flexibility studies using the RIGIX program also examined how the protein environment modulates electronic structure and reactivity. The research provides new insights into CYP450 catalysis at the molecular level and could aid in drug design.
Michael Ludden L3Report2016

This document describes Michael Ludden's synthesis and characterization of various molybdenum complexes. Three complexes were synthesized - [CpMo(CO)3Me], [CpMo(CO)3Et], and [CpMo(CO)2(COMe)(PPh3)]. They were characterized using NMR and IR spectroscopy. The results confirmed the structures of the complexes and showed how changing ligands affects properties. Kinetic measurements of migratory insertion reactions will be taken using these complexes to understand reaction rate dependence on factors like solvent, temperature and ligand type.
OBC epoxidations paper - Queen Mary University LONDON UK - Thomas Follier

This document reports on a study of the catalytic activity of manganese complexes with two similar polyamine ligands (7 and 8) that differ by the presence of a secondary or tertiary nitrogen, in the epoxidation of styrene. Ligand 7 showed the highest activity with MnSO4 and H2O2, while ligand 8 was most effective with Mn(OTf)2, MnCl2, and Mn(ClO4)2 using peracetic acid. Kinetic analysis indicated the structural differences in the ligands lead to differences in the nature of the active species formed. Ligand 7 with MnSO4 produced the epoxide in 78% yield, while ligand 8 with Mn(OT
Thesis-PerLindecrantz-2009

This document describes research into direct aldol reactions mediated by dimethylzinc. Three aromatic aldol products were synthesized and their NMR data reported. Racemic aldol products were produced using either dialkylzinc/dialcohol or magnesium bromide/DIPEA. Successful asymmetric and autocatalytic aldol reactions were also achieved using chiral catalysts. The products of some aldol reactions were found to facilitate their own formation and the reaction between other substrates, demonstrating autocatalytic properties. Further study of this autocatalytic process is warranted.
Liu2010

This document summarizes research on immobilizing palladium nanoparticles with a functional ionic liquid grafted onto a cross-linked polymer for solvent-free Heck reactions. Specifically:
1) A functional ionic liquid containing an amine group was synthesized and copolymerized with divinylbenzene to produce a cross-linked polymer support.
2) Palladium nanoparticles were immobilized on this polymer using aqueous palladium chloride, then reduced with sodium borohydride.
3) Characterization with FTIR, TGA, TEM, XPS showed the palladium nanoparticles were successfully supported on the polymer through amine binding.
4) This catalyst was tested for Heck
Molbiol 2011-04-metabolism

The document discusses metabolism and metabolic pathways. It summarizes that catabolism provides energy and building blocks for anabolism through metabolic pathways. Metabolic pathways involve enzymatically catalyzed reactions, with enzymes determining the pathways. Reaction rates are influenced by factors like temperature, pH, substrate concentration, and inhibitors. The document then discusses specific metabolic pathways like glycolysis, the Krebs cycle, and the electron transport chain which are involved in breaking down carbohydrates to release energy through cellular respiration.
MolBiol #3.2

The document discusses metabolism and metabolic pathways. It explains that catabolism releases energy through breakdown of molecules while anabolism uses this energy to build molecules. Metabolic pathways involve a series of enzyme-catalyzed reactions. Enzymes are proteins that catalyze specific reactions and influence reaction rates without being consumed. ATP is generated through substrate-level phosphorylation and oxidative phosphorylation during cellular respiration. Aerobic respiration using oxygen yields more ATP than anaerobic respiration with other electron acceptors.
Molbiol 2011-04-metabolism

The document discusses metabolism and metabolic pathways. It summarizes that catabolism provides energy and building blocks for anabolism through metabolic pathways. Metabolic pathways involve enzymatically catalyzed reactions, with enzymes determining the pathways. Reaction rates are influenced by factors like temperature, pH, substrate concentration, and inhibitors. The document then discusses specific metabolic pathways like glycolysis, the Krebs cycle, and the electron transport chain which are involved in breaking down carbohydrates to release energy through cellular respiration.
Mechanistic Aspects of Oxidation of P-Bromoacetophen one by Hexacyanoferrate ...

The kinetics of oxidation of p-bromoacetophenone by hexacyanoferrate (III) has been studied in alkaline
medium. The order of reaction with respect of both acetophenone and hexacynoferrate (III) has been found to be
unity. The rate of reaction increases with increase in the concentration of sodium hydroxide.On addition of
neutral KCl, reaction rate increases. The effects of solvent and temperature have been also studied. The product
p-bromophenyl glyoxal have been characterized by IR studies.
Biological oxidation L5 (oxidative phosphorylation) pdf

The document discusses oxidative phosphorylation and chemiosmotic theory. It explains that oxidative phosphorylation is the process of synthesizing ATP from ADP and Pi coupled to the electron transport chain. The chemiosmotic theory proposes that the electron transport chain pumps protons across the inner mitochondrial membrane, creating an electrochemical gradient. ATP synthase then uses this proton gradient to drive the phosphorylation of ADP to ATP. The theory is supported by evidence that ATP synthase acts as a proton-driven rotary motor to generate ATP through conformational changes.
poster template 2015 4501

This document summarizes an experiment analyzing the diatom Thalassiosira pseudonana and the protein Tp34211. Tp34211 is a putative monoheme cytochrome c that may act as a catalyst in the reaction between carbon dioxide and Rubisco, an important carbon fixation enzyme. The experiment transformed Tp34211 into E. coli cells and purified the protein. Testing showed that Tp34211 increased the rate of a peroxidase reaction as hydrogen peroxide concentration increased, indicating it may act as a catalyst, although not a strong one. Further analysis of Tp34211's role in carbon dioxide and Rubisco interconversion in T. pseudonana is needed.
7.27.10 enzymes coloso

Enzymes are proteins that catalyze biochemical reactions in cells. They increase the rate of reactions by lowering activation energy. Most enzymes are named based on their substrate or the reaction they catalyze. Studying enzyme kinetics and regulation provides insight into metabolic pathways and cellular functions.
Gonzalez et al., JACS 2011, 133, 5500-5507

This document summarizes research on using phosphoramidite gold(I) catalysts to catalyze the diastereo- and enantioselective synthesis of 3,4-substituted pyrrolidines through cycloadditions of allenenes. Key findings include:
1) The phosphoramidite ligand (R,R,R)-9a enabled highly enantioselective cycloadditions, providing up to 99% ee for various allenenes.
2) Computational studies supported a stepwise mechanism involving carbocation intermediates that explain the regio- and stereoselectivity of the reactions.
3) Both cis and trans reaction pathways were found to be
CPsarakis REU Final Paper

This study investigated the polymerization of lactic acid as a model for prebiotic peptide formation via ester-amide exchange. Lactic acid was polymerized in a closed system at 85°C over various time points. HPLC and 1H-NMR were used to analyze the polymers and determine degree of polymerization (DP) and total lactic acid units. DP was found to increase with time while total units decreased, showing polymer regeneration. Methods showed consistent results within 10-15% error. Further studies will compare kinetics to a computer simulation to determine rate constants and model polymerization from various monomers.
acs%2Eorganomet%2E6b00671

This summary provides the key details from the document in 3 sentences:
The document reports on a study of chromium-catalyzed ethylene oligomerization using bis(benzimidazolemethyl)amine (BIMA) ligands. It finds that N-alkyl-substituted BIMA ligands produce extremely high catalyst activities (>100,000 g mmol-1 h-1 bar-1) and yield alternating distributions of linear α-olefins. The alternating distributions can be explained by a metallacyclic mechanism in which both single and double ethylene insertions into the metallacycle are possible.
Learning new reactions by solving total syntheses merged

Total synthesis of Sterpurenone New, Total Synthesis of (훽)-Cyperolone, Protecting Group-Free Total Synthesis of (−)-Lannotinidine B, Enantiospecific Total Synthesis of the (−)-Presilphiperfolan-8-ol, Enantioselective Total Synthesis of (−)-Pavidolide B, total synthesis of Eupalinilide E
5th international olympiad metropolises

This document presents 5 theoretical chemistry problems. Problem 1 involves identifying compounds containing element X based on provided information about mass percentages and oxidation states. Key compounds identified are:
A is ClF. B and Z1 contain Cl, F, and O. Z1 is ClOF3.
The document also involves writing chemical equations, identifying possible isomers, explaining conductivity, and writing hydrolysis equations.
Undergraduate Laboratory Development: Finding Cost-Effective Catalysts for th...

-Developed an undergraduate organic or inorganic chemistry laboratory experiment to showcase the catalytic coupling reaction between waste carbon dioxide and epoxides to yield value-added cyclic carbonate materials
-Focused on maximizing the reaction yield and reducing experiment costs by employing bimetallic salen complexes and zinc octoate in the catalytic system
-Aided in the optimization of system conditions of coupling reaction in simple polypropylene centrifuge tubes and synthesis of bimetallic aluminum salen catalyst
-Presented poster named "Undergraduate Laboratory Development: Finding Cost-Effective Catalysts for the Coupling of Epoxides and CO2" at the 257th American Chemical Society (ACS) National Meeting & Exposition in Orlando, FL
Dsc peptide

This document summarizes a study that measured the enthalpy change (ΔH) associated with the α-helix to random coil transition of an alanine peptide in water using calorimetry. The researchers synthesized a 50-residue peptide containing primarily alanine residues and determined its ΔH to be between 0.9-1.3 kcal/mol per residue, providing a basic parameter for predicting thermal unfolding of peptide helices. Circular dichroism spectra and melting curves confirmed the peptide adopted an α-helical structure at low temperatures and underwent a reversible helix-coil transition. The ΔH value suggests the peptide backbone, rather than side chains, makes the dominant contribution to helix stability.
Kinetic And Mechanism of Oxidation of Cobalt Metal Complex By Acidic Potassiu...

ABSTRACT: The Kinetic of oxidation of Cobalt derived from 8-hydroxy quinolone and salicylaldehyde by potassium permanganate has been studied in the presence of acidic medium. The overall reaction is first order with respect to KMnO4, Substrate acid and temperature. No effect of salts has been found suitable mechanism is given.
Similar to Computational and Experimental Studies of MTO Catalyzed Olefin Hydrogenation (20)
OBC epoxidations paper - Queen Mary University LONDON UK - Thomas Follier

OBC epoxidations paper - Queen Mary University LONDON UK - Thomas Follier
Mechanistic Aspects of Oxidation of P-Bromoacetophen one by Hexacyanoferrate ...

Mechanistic Aspects of Oxidation of P-Bromoacetophen one by Hexacyanoferrate ...
Biological oxidation L5 (oxidative phosphorylation) pdf

Biological oxidation L5 (oxidative phosphorylation) pdf
Learning new reactions by solving total syntheses merged

Learning new reactions by solving total syntheses merged
Undergraduate Laboratory Development: Finding Cost-Effective Catalysts for th...

Undergraduate Laboratory Development: Finding Cost-Effective Catalysts for th...
Kinetic And Mechanism of Oxidation of Cobalt Metal Complex By Acidic Potassiu...

Kinetic And Mechanism of Oxidation of Cobalt Metal Complex By Acidic Potassiu...
Recently uploaded
Shallowest Oil Discovery of Turkiye.pptx

The Petroleum System of the Çukurova Field - the Shallowest Oil Discovery of Türkiye, Adana
Applied Science: Thermodynamics, Laws & Methodology.pdf

When I was asked to give a companion lecture in support of ‘The Philosophy of Science’ (https://shorturl.at/4pUXz) I decided not to walk through the detail of the many methodologies in order of use. Instead, I chose to employ a long standing, and ongoing, scientific development as an exemplar. And so, I chose the ever evolving story of Thermodynamics as a scientific investigation at its best.
Conducted over a period of >200 years, Thermodynamics R&D, and application, benefitted from the highest levels of professionalism, collaboration, and technical thoroughness. New layers of application, methodology, and practice were made possible by the progressive advance of technology. In turn, this has seen measurement and modelling accuracy continually improved at a micro and macro level.
Perhaps most importantly, Thermodynamics rapidly became a primary tool in the advance of applied science/engineering/technology, spanning micro-tech, to aerospace and cosmology. I can think of no better a story to illustrate the breadth of scientific methodologies and applications at their best.
Equivariant neural networks and representation theory

Or: Beyond linear.
Abstract: Equivariant neural networks are neural networks that incorporate symmetries. The nonlinear activation functions in these networks result in interesting nonlinear equivariant maps between simple representations, and motivate the key player of this talk: piecewise linear representation theory.
Disclaimer: No one is perfect, so please mind that there might be mistakes and typos.
dtubbenhauer@gmail.com
Corrected slides: dtubbenhauer.com/talks.html
Randomised Optimisation Algorithms in DAPHNE

Slides from talk:
Aleš Zamuda: Randomised Optimisation Algorithms in DAPHNE .
Austrian-Slovenian HPC Meeting 2024 – ASHPC24, Seeblickhotel Grundlsee in Austria, 10–13 June 2024
https://ashpc.eu/
Bob Reedy - Nitrate in Texas Groundwater.pdf

Presented at June 6-7 Texas Alliance of Groundwater Districts Business Meeting
SAR of Medicinal Chemistry 1st by dk.pdf

In this presentation include the prototype drug SAR on thus or with their examples .
Syllabus of Second Year B. Pharmacy
2019 PATTERN.
ANAMOLOUS SECONDARY GROWTH IN DICOT ROOTS.pptx

Abnormal or anomalous secondary growth in plants. It defines secondary growth as an increase in plant girth due to vascular cambium or cork cambium. Anomalous secondary growth does not follow the normal pattern of a single vascular cambium producing xylem internally and phloem externally.
bordetella pertussis.................................ppt

Bordettela is a gram negative cocobacilli spread by air born drop let
The debris of the ‘last major merger’ is dynamically young

The Milky Way’s (MW) inner stellar halo contains an [Fe/H]-rich component with highly eccentric orbits, often referred to as the
‘last major merger.’ Hypotheses for the origin of this component include Gaia-Sausage/Enceladus (GSE), where the progenitor
collided with the MW proto-disc 8–11 Gyr ago, and the Virgo Radial Merger (VRM), where the progenitor collided with the
MW disc within the last 3 Gyr. These two scenarios make different predictions about observable structure in local phase space,
because the morphology of debris depends on how long it has had to phase mix. The recently identified phase-space folds in Gaia
DR3 have positive caustic velocities, making them fundamentally different than the phase-mixed chevrons found in simulations
at late times. Roughly 20 per cent of the stars in the prograde local stellar halo are associated with the observed caustics. Based
on a simple phase-mixing model, the observed number of caustics are consistent with a merger that occurred 1–2 Gyr ago.
We also compare the observed phase-space distribution to FIRE-2 Latte simulations of GSE-like mergers, using a quantitative
measurement of phase mixing (2D causticality). The observed local phase-space distribution best matches the simulated data
1–2 Gyr after collision, and certainly not later than 3 Gyr. This is further evidence that the progenitor of the ‘last major merger’
did not collide with the MW proto-disc at early times, as is thought for the GSE, but instead collided with the MW disc within
the last few Gyr, consistent with the body of work surrounding the VRM.
8.Isolation of pure cultures and preservation of cultures.pdf

Isolation of pure culture, its various method.
BREEDING METHODS FOR DISEASE RESISTANCE.pptx

Plant breeding for disease resistance is a strategy to reduce crop losses caused by disease. Plants have an innate immune system that allows them to recognize pathogens and provide resistance. However, breeding for long-lasting resistance often involves combining multiple resistance genes
The use of Nauplii and metanauplii artemia in aquaculture (brine shrimp).pptx

Although Artemia has been known to man for centuries, its use as a food for the culture of larval organisms apparently began only in the 1930s, when several investigators found that it made an excellent food for newly hatched fish larvae (Litvinenko et al., 2023). As aquaculture developed in the 1960s and ‘70s, the use of Artemia also became more widespread, due both to its convenience and to its nutritional value for larval organisms (Arenas-Pardo et al., 2024). The fact that Artemia dormant cysts can be stored for long periods in cans, and then used as an off-the-shelf food requiring only 24 h of incubation makes them the most convenient, least labor-intensive, live food available for aquaculture (Sorgeloos & Roubach, 2021). The nutritional value of Artemia, especially for marine organisms, is not constant, but varies both geographically and temporally. During the last decade, however, both the causes of Artemia nutritional variability and methods to improve poorquality Artemia have been identified (Loufi et al., 2024).
Brine shrimp (Artemia spp.) are used in marine aquaculture worldwide. Annually, more than 2,000 metric tons of dry cysts are used for cultivation of fish, crustacean, and shellfish larva. Brine shrimp are important to aquaculture because newly hatched brine shrimp nauplii (larvae) provide a food source for many fish fry (Mozanzadeh et al., 2021). Culture and harvesting of brine shrimp eggs represents another aspect of the aquaculture industry. Nauplii and metanauplii of Artemia, commonly known as brine shrimp, play a crucial role in aquaculture due to their nutritional value and suitability as live feed for many aquatic species, particularly in larval stages (Sorgeloos & Roubach, 2021).
What is greenhouse gasses and how many gasses are there to affect the Earth.

What are greenhouse gasses how they affect the earth and its environment what is the future of the environment and earth how the weather and the climate effects.
Deep Software Variability and Frictionless Reproducibility

Deep Software Variability and Frictionless ReproducibilityUniversity of Rennes, INSA Rennes, Inria/IRISA, CNRS
The ability to recreate computational results with minimal effort and actionable metrics provides a solid foundation for scientific research and software development. When people can replicate an analysis at the touch of a button using open-source software, open data, and methods to assess and compare proposals, it significantly eases verification of results, engagement with a diverse range of contributors, and progress. However, we have yet to fully achieve this; there are still many sociotechnical frictions.
Inspired by David Donoho's vision, this talk aims to revisit the three crucial pillars of frictionless reproducibility (data sharing, code sharing, and competitive challenges) with the perspective of deep software variability.
Our observation is that multiple layers — hardware, operating systems, third-party libraries, software versions, input data, compile-time options, and parameters — are subject to variability that exacerbates frictions but is also essential for achieving robust, generalizable results and fostering innovation. I will first review the literature, providing evidence of how the complex variability interactions across these layers affect qualitative and quantitative software properties, thereby complicating the reproduction and replication of scientific studies in various fields.
I will then present some software engineering and AI techniques that can support the strategic exploration of variability spaces. These include the use of abstractions and models (e.g., feature models), sampling strategies (e.g., uniform, random), cost-effective measurements (e.g., incremental build of software configurations), and dimensionality reduction methods (e.g., transfer learning, feature selection, software debloating).
I will finally argue that deep variability is both the problem and solution of frictionless reproducibility, calling the software science community to develop new methods and tools to manage variability and foster reproducibility in software systems.
Exposé invité Journées Nationales du GDR GPL 2024
Thornton ESPP slides UK WW Network 4_6_24.pdf

ESPP presentation to EU Waste Water Network, 4th June 2024 “EU policies driving nutrient removal and recycling
and the revised UWWTD (Urban Waste Water Treatment Directive)”
Deep Behavioral Phenotyping in Systems Neuroscience for Functional Atlasing a...

Functional Magnetic Resonance Imaging (fMRI) provides means to characterize brain activations in response to behavior. However, cognitive neuroscience has been limited to group-level effects referring to the performance of specific tasks. To obtain the functional profile of elementary cognitive mechanisms, the combination of brain responses to many tasks is required. Yet, to date, both structural atlases and parcellation-based activations do not fully account for cognitive function and still present several limitations. Further, they do not adapt overall to individual characteristics. In this talk, I will give an account of deep-behavioral phenotyping strategies, namely data-driven methods in large task-fMRI datasets, to optimize functional brain-data collection and improve inference of effects-of-interest related to mental processes. Key to this approach is the employment of fast multi-functional paradigms rich on features that can be well parametrized and, consequently, facilitate the creation of psycho-physiological constructs to be modelled with imaging data. Particular emphasis will be given to music stimuli when studying high-order cognitive mechanisms, due to their ecological nature and quality to enable complex behavior compounded by discrete entities. I will also discuss how deep-behavioral phenotyping and individualized models applied to neuroimaging data can better account for the subject-specific organization of domain-general cognitive systems in the human brain. Finally, the accumulation of functional brain signatures brings the possibility to clarify relationships among tasks and create a univocal link between brain systems and mental functions through: (1) the development of ontologies proposing an organization of cognitive processes; and (2) brain-network taxonomies describing functional specialization. To this end, tools to improve commensurability in cognitive science are necessary, such as public repositories, ontology-based platforms and automated meta-analysis tools. I will thus discuss some brain-atlasing resources currently under development, and their applicability in cognitive as well as clinical neuroscience.
如何办理(uvic毕业证书)维多利亚大学毕业证本科学位证书原版一模一样

原版纸张【微信:741003700 】【(uvic毕业证书)维多利亚大学毕业证】【微信:741003700 】学位证,留信认证(真实可查,永久存档)offer、雅思、外壳等材料/诚信可靠,可直接看成品样本,帮您解决无法毕业带来的各种难题!外壳,原版制作,诚信可靠,可直接看成品样本。行业标杆!精益求精,诚心合作,真诚制作!多年品质 ,按需精细制作,24小时接单,全套进口原装设备。十五年致力于帮助留学生解决难题,包您满意。
本公司拥有海外各大学样板无数,能完美还原海外各大学 Bachelor Diploma degree, Master Degree Diploma
1:1完美还原海外各大学毕业材料上的工艺:水印,阴影底纹,钢印LOGO烫金烫银,LOGO烫金烫银复合重叠。文字图案浮雕、激光镭射、紫外荧光、温感、复印防伪等防伪工艺。材料咨询办理、认证咨询办理请加学历顾问Q/微741003700
留信网认证的作用:
1:该专业认证可证明留学生真实身份
2:同时对留学生所学专业登记给予评定
3:国家专业人才认证中心颁发入库证书
4:这个认证书并且可以归档倒地方
5:凡事获得留信网入网的信息将会逐步更新到个人身份内,将在公安局网内查询个人身份证信息后,同步读取人才网入库信息
6:个人职称评审加20分
7:个人信誉贷款加10分
8:在国家人才网主办的国家网络招聘大会中纳入资料,供国家高端企业选择人才
Recently uploaded (20)
Applied Science: Thermodynamics, Laws & Methodology.pdf

Applied Science: Thermodynamics, Laws & Methodology.pdf
Equivariant neural networks and representation theory

Equivariant neural networks and representation theory
bordetella pertussis.................................ppt

bordetella pertussis.................................ppt
The debris of the ‘last major merger’ is dynamically young

The debris of the ‘last major merger’ is dynamically young
8.Isolation of pure cultures and preservation of cultures.pdf

8.Isolation of pure cultures and preservation of cultures.pdf
The use of Nauplii and metanauplii artemia in aquaculture (brine shrimp).pptx

The use of Nauplii and metanauplii artemia in aquaculture (brine shrimp).pptx
What is greenhouse gasses and how many gasses are there to affect the Earth.

What is greenhouse gasses and how many gasses are there to affect the Earth.
Deep Software Variability and Frictionless Reproducibility

Deep Software Variability and Frictionless Reproducibility
Deep Behavioral Phenotyping in Systems Neuroscience for Functional Atlasing a...

Deep Behavioral Phenotyping in Systems Neuroscience for Functional Atlasing a...
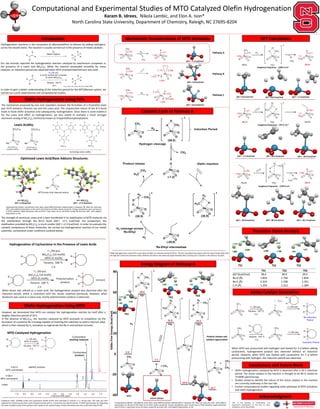
Computational and Experimental Studies of MTO Catalyzed Olefin Hydrogenation
- 1. The strength of aluminum Lewis acid is best manifested in its stabilization of MTO molecule via the coordination through the Re=O bond (∆Go= -17.5 kcal/mol). For comparison, the stabilization provided by B(C6F5)3 is much smaller (∆Go= +2.3 kcal/mol). In order to evaluate the catalytic competency of these molecules, we carried out hydrogenation reaction of our model substrate, cyclooctene under conditions outlined below: Computational and Experimental Studies of MTO Catalyzed Olefin Hydrogenation Karam B. Idrees, Nikola Lambic, and Elon A. Ison* North Carolina State University, Department of Chemistry, Raleigh, NC 27695-8204 Hydrogenation reactions is the conversion of alkenes(olefins) to alkanes by adding hydrogens across the double bond. The reaction is usually carried out in the presence of metal catalysts: However, we discovered that MTO can catalyze the hydrogenation reaction by itself after a lengthy induction period of 20 h. In the absence of B(C6F5)3, the reaction catalyzed by MTO proceeds to completion via the formation of a putative Re-H analog capable of inserting the substrate to yield a rhenium alkyl, which is then cleaved by H2 activation to regenerate the Re-H and achieve turnover. TS1 TS2 Introduction Olefin Hydrogenation Using FLPs Mechanistic Considerations of MTO Activation Catalytic Cycle of Pathway A Energy Diagram of Pathway A Transition States Analysis Conclusions and Future Work -REU at the Interface of Computations and Experiments coordinators: Dr. Elon Ison, Dr. Elena Jakubikova, and Dr. Reza Ghiladi. DFT Calculations B(C6F5)3 Al(C6F5)3 Increasing Lewis acidity LA= Al(C6F5)3 ∆Go= -17.5 kcal/mol Optimized Lewis Acid/Base Adducts Structures: MTO/ B(C6F5)3 Acid/Base adduct MTO/ Al(C6F5)3 Acid/Base adduct Lewis Acidity: -40 -20 0 20 40 60 80 GibbsFreeEnergy(kcal/mol) ReactionCoordinate ∆Go= 0.0 kcal/mol ∆G‡= 58.6 kcal/mol ∆Go= -7.2 kcal/mol Imaginary frequency: -1284.4 cm-1 Imaginary frequency: -547.6 cm-1 ∆Go= -7.2 kcal/mol ∆G‡= 30.9 kcal/mol ∆Go= -29.6 kcal/mol ∆Go= -29.6 kcal/mol ∆G‡= 30.5 kcal/mol ∆Go= -34.7 kcal/mol Imaginary frequency: -1259.6 cm-1 Olefin Hydrogenation Using MTO TS1 TS2 TS3 ∆G‡ (kcal/mol) 58.6 30.9 30.5 Re-H (Å) 1.853 1.756 1.854 Re-C (Å) 2.323 2.339 2.351 C-H (Å) 1.354 1.512 1.364 Conditions: MTO (0.0046 mmol) and cyclooctene (0.092 mmol) were dissolved In toluene in a J-Young tube. The tube was then subjected to 3 freeze pump thaw cycles and pressurized with H2. Conversion was determined by 1H NMR spectroscopy by integrating the ratios of olefinic peak of the product with respect to the reactant peak. Product formation was also confirmed using GC-MS. Olefin hydrogenation using MTO is only observed after an induction period of 20 hrs. The first calculated step agrees with the experimental data with the high ∆G≠ of the first transition state. Methane formation was observed experimentally when carrying out a reaction in the absence of olefin. • Olefin hydrogenation catalyzed by MTO is observed after a 20 h induction period. The active catalyst in this reaction is thought to be Re-H, based on 1H NMR spectroscopy. • Studies aimed to identify the nature of the active catalyst in the reaction are currently underway in the Ison lab. • Further computational studies regarding other pathways of MTO activation and olefin hydrogenation. Acknowledgment Computational details: Calculations were done using M06 functional implemented in Gaussian 09. Basis set used was SDD with added f polarization on Re and 6-31G (d,p) on all other atoms except Re. Energy calculations were carried out in PCM solvation model (benzene) with 6-311G++ (d,p) basis set on all atoms except Re and was SDD with added f polarization on Re. ∆Go= 4.8 kcal/mol ∆Go= 13.0 kcal/mol ∆Go= -7.2 kcal/mol Pathway A Pathway B Pathway C Computational details: Calculations were done using M06 functional implemented in Gaussian 09. Basis set used was SDD with added f polarization on Re and 6-31G (d,p) on all other atoms except Re. Energy calculations were carried out in PCM solvation model (benzene) with 6-311G++ (d,p) basis set on all atoms except Re and was SDD with added f polarization on Re. Active Catalyst Generation TS1 TS2 TS3 Our lab recently reported the hydrogenation reaction catalyzed by oxorhenium complexes in the presence of a Lewis acid B(C6F5)3. While the reaction proceeded smoothly for many catalysts, an induction period was observed when MTO (methyltrioxorhenium) was used . J. Am. Chem. Soc., 2016, 138 (14), pp 4832–4842 In order to gain a better understanding of the induction period for the MTO/Borane system, we carried out a joint experimental and computational studies. The mechanism proposed by Ison and coworkers involves the formation of a frustrated Lewis pair (FLP) between rhenium oxo and the Lewis acid. The unquenched nature of the B-O bond leads to facile olefin activation and subsequently, hydrogenation. Since there is some evidence for the Lewis acid effect on hydrogenation, we also aimed to evaluate a much stronger aluminum analog of B(C6F5)3 commonly known as tris(pentafluorophenyl)alane. LA= B(C6F5)3 ∆Go= 2.3kcal/mol Hydrogenation of Cyclooctene in the Presence of Lewis Acids When boron was utilized as a Lewis acid, the hydrogenation product was observed after the induction period, which is consistent with the results reported previously. However, when aluminum was used as a Lewis acid, mostly polymerization product is observed. MTO Catalyzed Hydrogenation MTO Active catalyst Product release and catalyst regeneration TS3 When MTO was pressurized with hydrogen and heated for 3 d before adding cyclooctene, hydrogenated product was observed without an induction period. However, when MTO was heated with cyclooctene for 3 d before pressurizing with hydrogen, the induction period was observed.