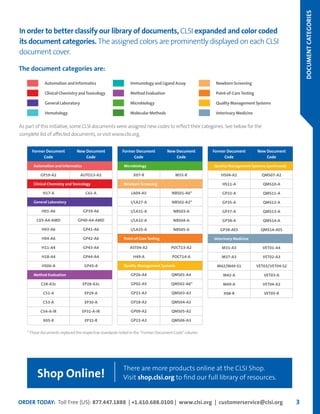
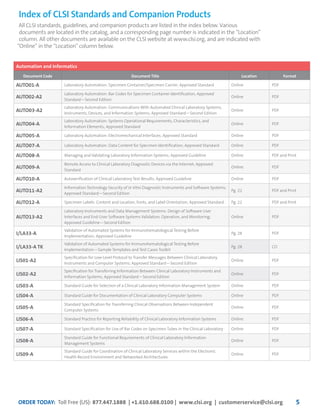
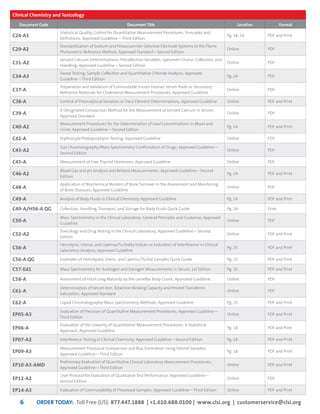
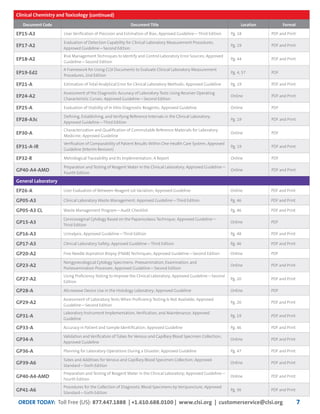
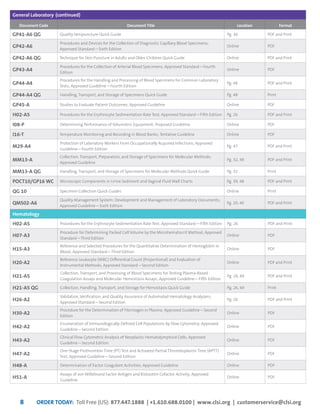
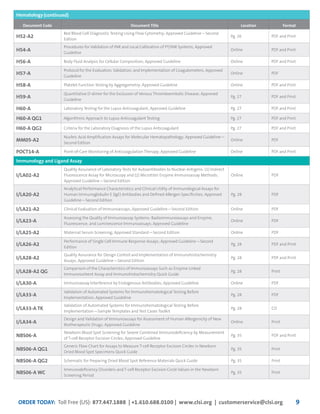
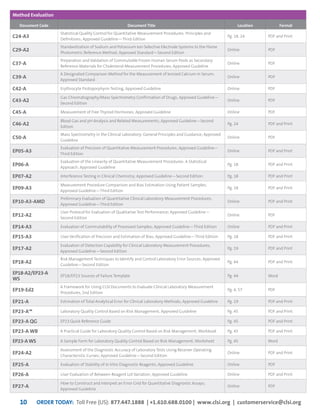
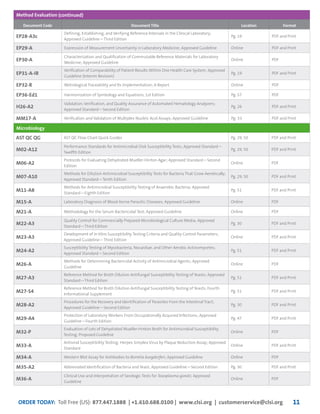
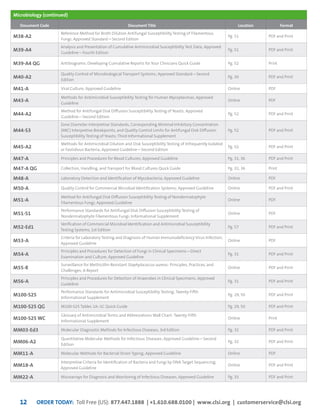
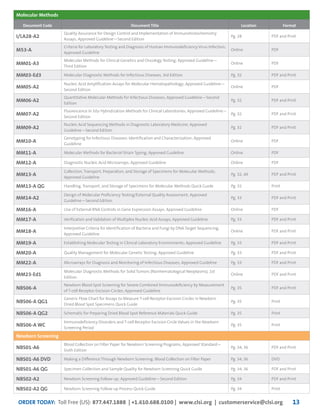
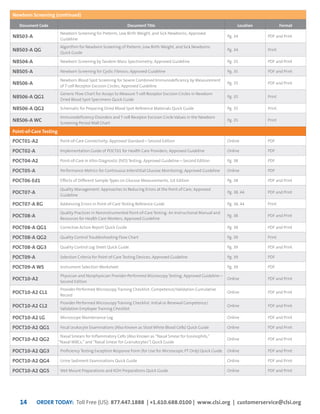
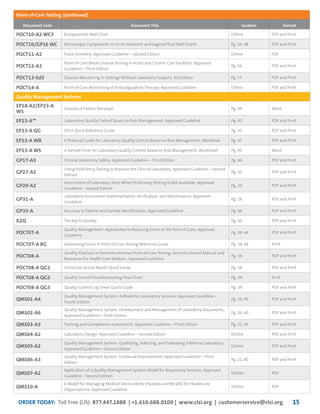
This document provides information about joining CLSI (Clinical and Laboratory Standards Institute) membership. Membership offers benefits like discounts on CLSI standards and programs, opportunities to collaborate on standards development, and continuing education credits. There are different levels of membership for laboratories, health systems, industries, government agencies, and individuals. Membership helps laboratories improve quality through implementing CLSI standards.