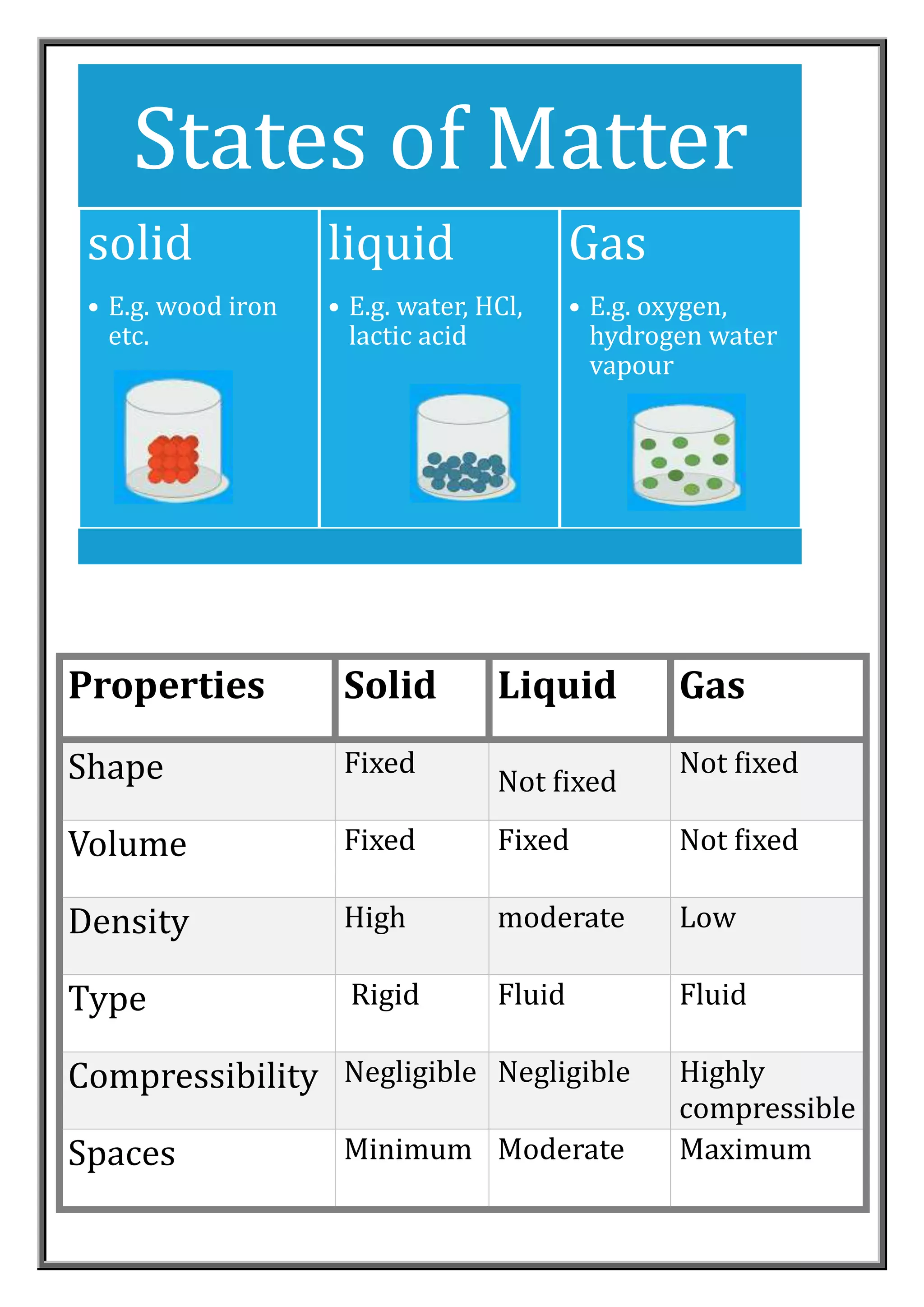
Matter is made up of tiny particles that are too small to be seen, even with a microscope. Experiments show that matter is composed of particles that can move and be transferred. Particles of matter have space between them, are continuously moving, and attract each other. The three common states of matter are solids, liquids, and gases, which have different characteristic properties depending on the strength of attraction between particles and how freely they can move. Changes in temperature or pressure can cause changes in a substance's state, such as melting, boiling, condensation, and sublimation.