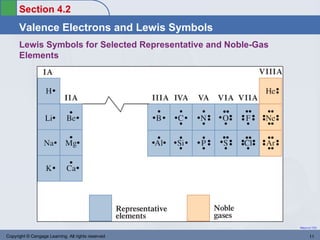
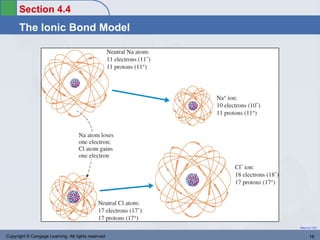
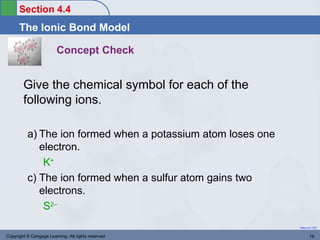
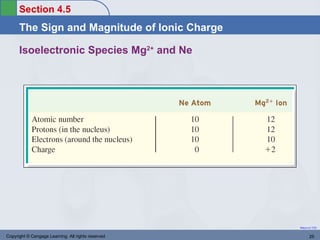
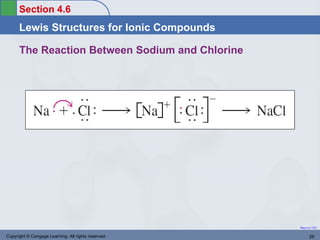
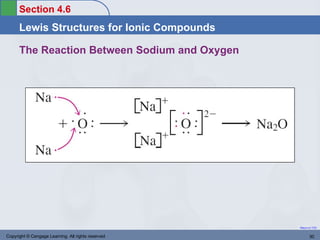
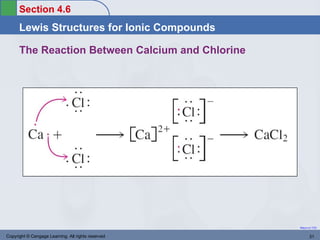
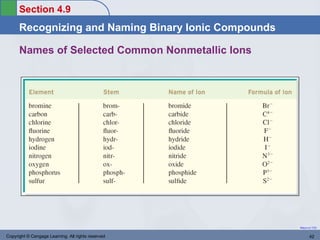
This document provides an overview of chemical bonding and ionic compounds. It discusses the ionic bond model, where ions are formed through the transfer of electrons between atoms. Metals tend to lose electrons to form positively charged cations, while nonmetals gain electrons to form negatively charged anions. Ionic compounds are formed when cations and anions combine in ratios that result in an electrically neutral compound. The structures and names of ionic compounds are also covered.