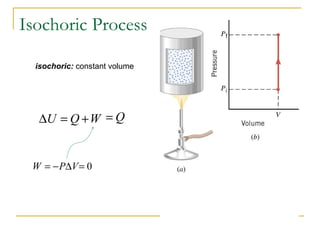
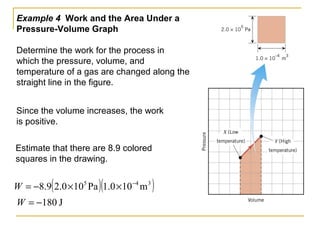
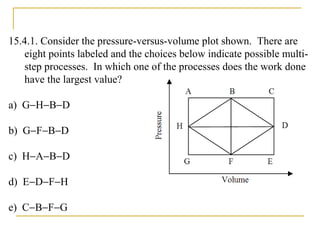
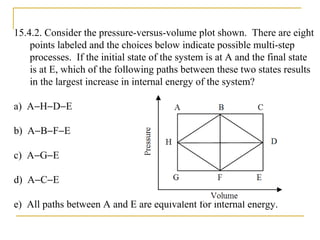
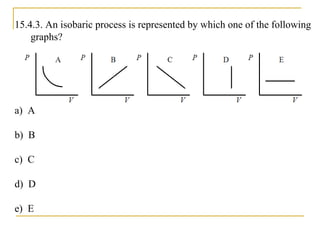
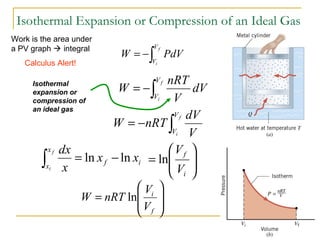
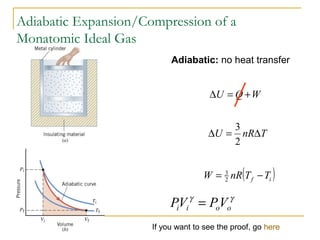
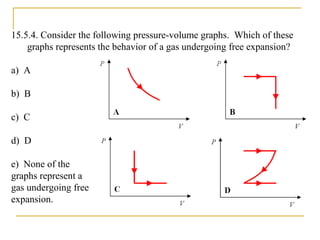
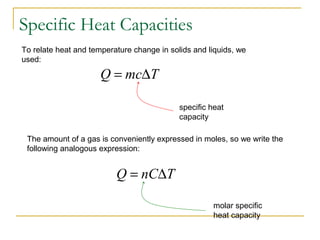
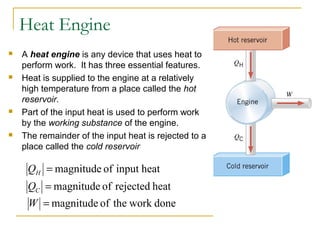
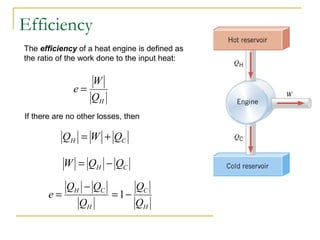
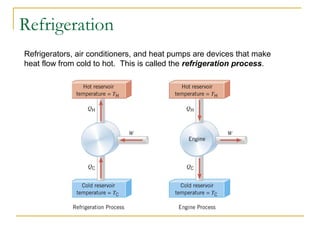
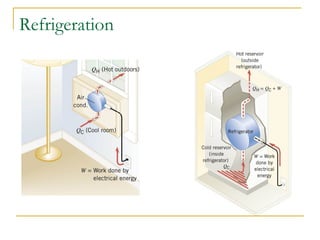
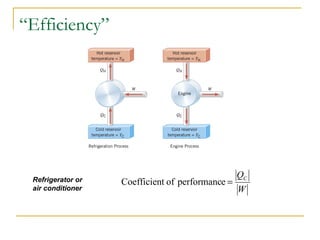
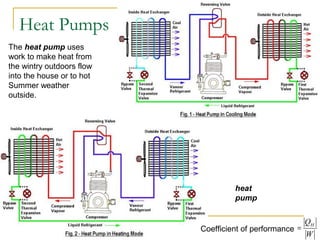
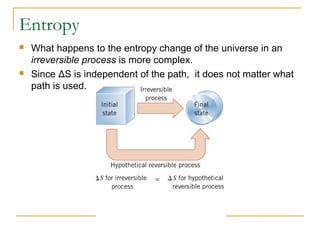
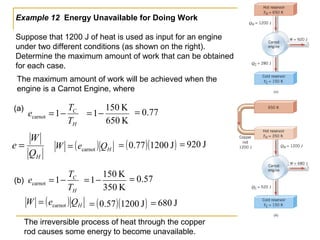
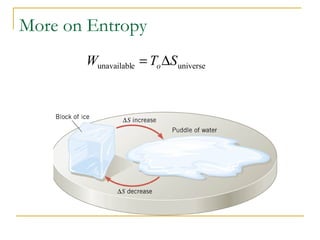
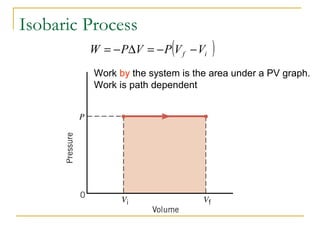
The document provides learning objectives and content for a chapter on thermodynamics. It covers key concepts like the first and second laws of thermodynamics, thermal processes, and using the ideal gas law. For thermal processes using ideal gases, it defines equations for isothermal, adiabatic, isobaric and isochoric processes. Examples are provided for calculating work done during isothermal expansion of an ideal gas and adiabatic compression. The chapter sections will address thermodynamic systems, the laws of thermodynamics, thermal processes, processes for ideal gases, and applications like heat engines and entropy.
























C31CkgJ4186kg0010.0 ⋅= J130=](https://image.slidesharecdn.com/ch15thermodynamicsonline-101101125335-phpapp02/85/Ch-15-Thermodynamics-25-320.jpg)