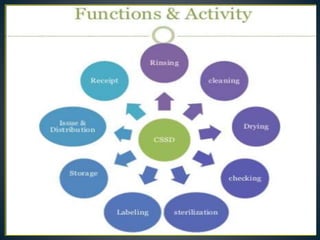
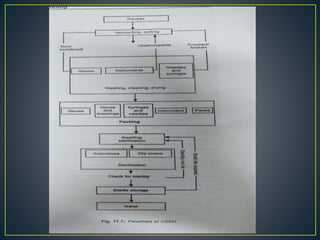
The Central Sterile Supply Department (CSSD) is responsible for receiving, processing, sterilizing, storing and distributing sterile supplies and equipment for hospitals. Key functions of the CSSD include cleaning, packaging, sterilizing items using autoclaves or ethylene oxide, storing sterile supplies, and issuing items to different hospital units. CSSDs aim to provide safe sterile supplies efficiently while standardizing equipment and assisting in infection control.