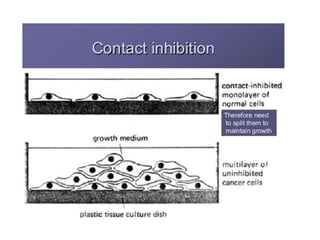
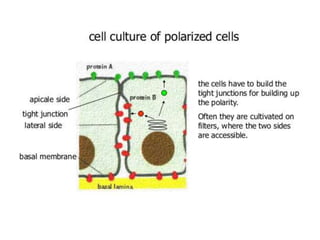
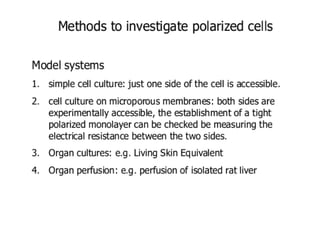
There are two main scientific strategies for studying biological systems: descriptive strategies, which observe the system without altering it, and manipulative strategies, which purposefully alter factors to observe their effects. In vitro techniques allow controlling and manipulating many parameters. Cell culture systems allow studying cells outside the body and include immortalized cell lines, primary cells, co-cultures, and organ-like cultures. Proper conditions and techniques must be used to maintain cell viability in culture.