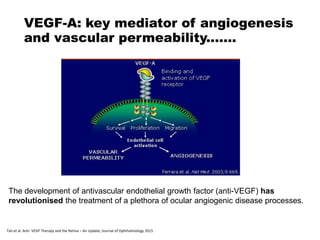
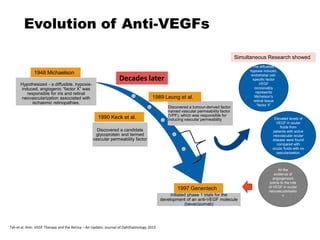
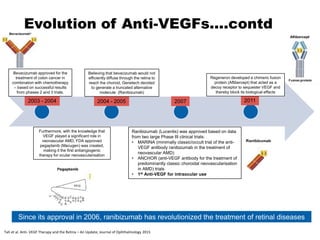
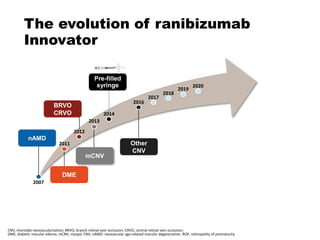
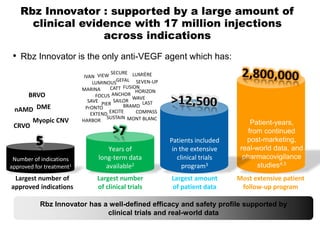
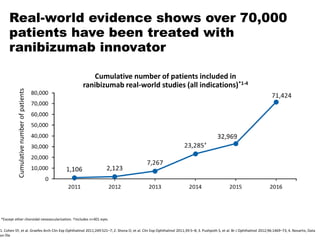
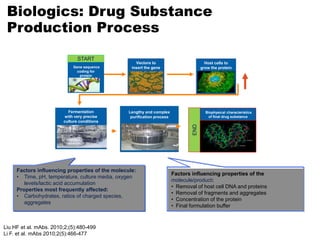
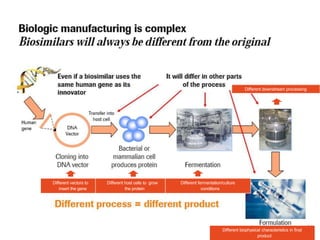
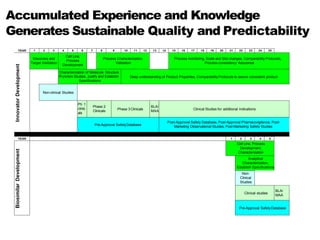
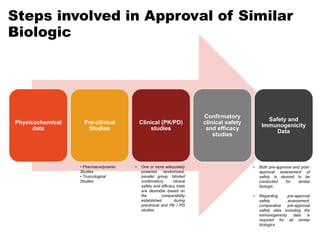
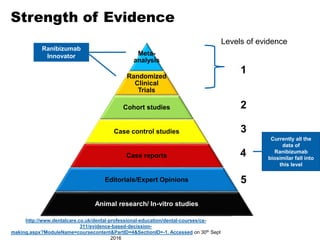
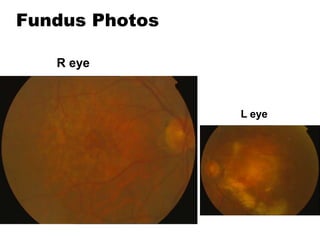
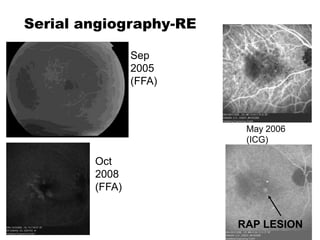
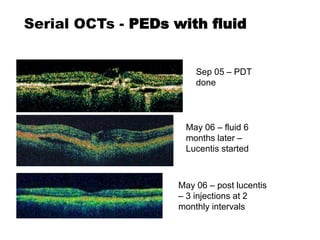
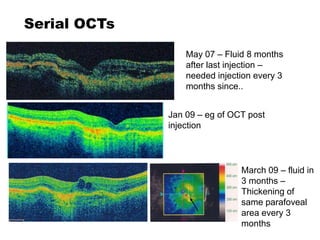
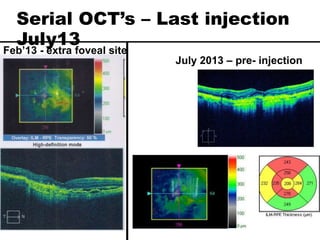
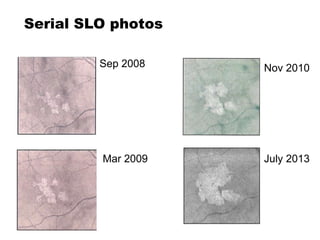
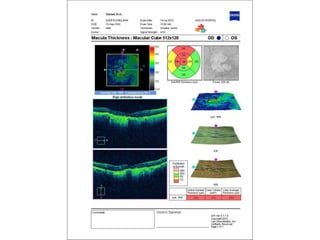
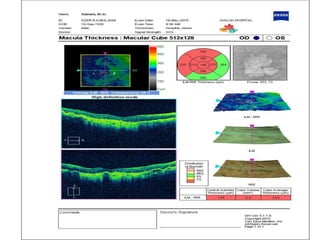
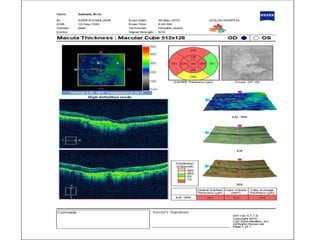
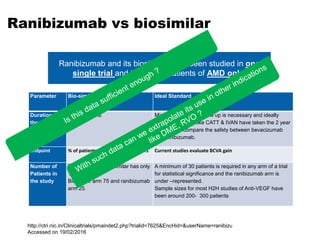
This document discusses the treatment of retinal conditions with a focus on anti-VEGF therapies. It provides a history of the development of anti-VEGF treatments from the discovery of VEGF in the 1980s and 1990s to the approval of ranibizumab and aflibercept in the 2000s. It then discusses the evolution of ranibizumab and the evidence from clinical trials supporting its use. Finally, it addresses biosimilars that are attempting to enter the anti-VEGF market and highlights some of the differences between biosimilars and innovator biologics.