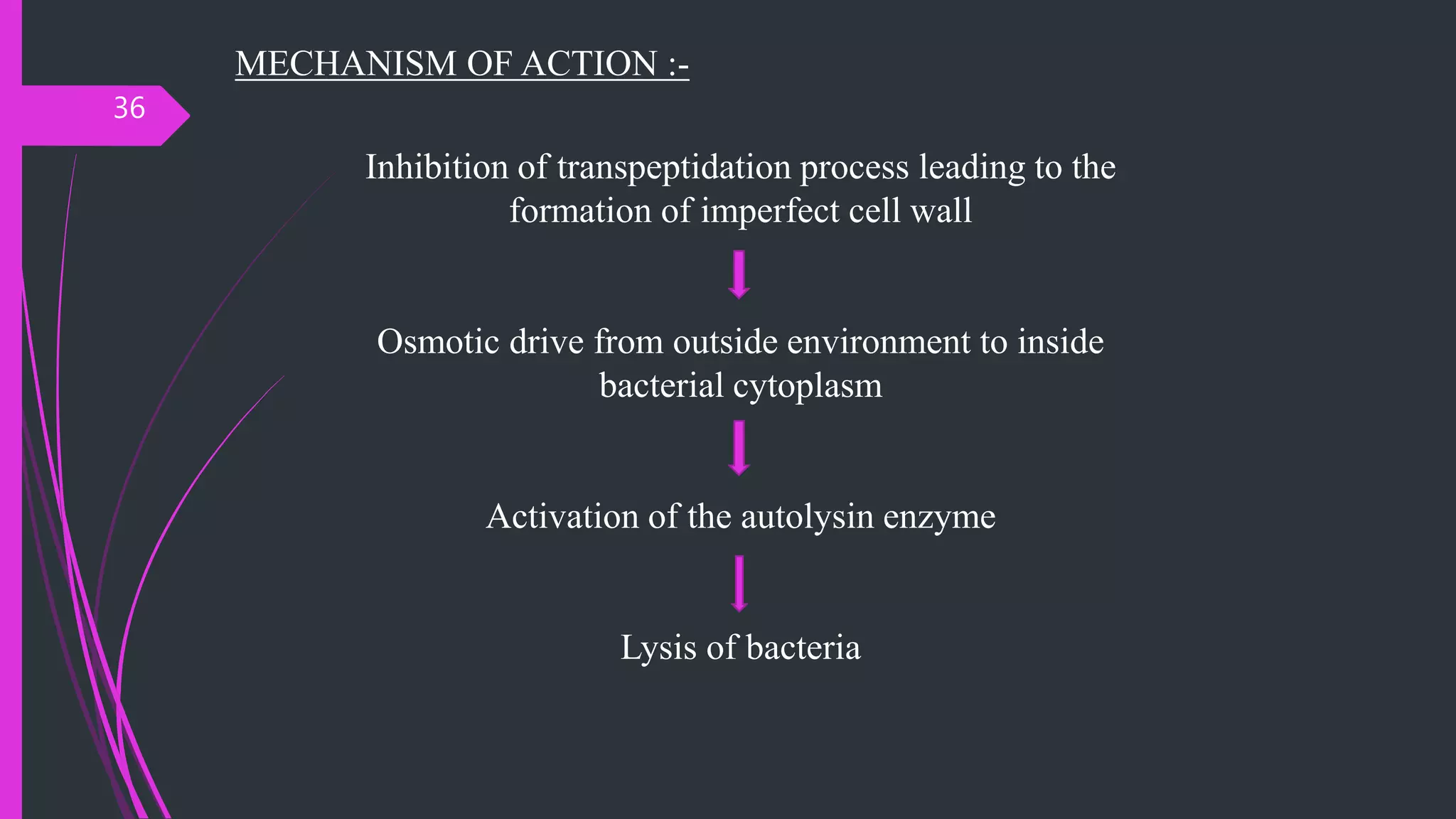
This document provides an overview of inhibitors of bacterial cell wall synthesis, with a focus on beta-lactam antibiotics such as penicillins, cephalosporins, carbapenems, and glycopeptides. It begins with an introduction to bacterial cell wall synthesis and the classes of antibiotics that inhibit this process. The bulk of the document then discusses specific antibiotic classes in more detail, including their mechanisms of action, classifications, pharmacokinetics, uses, and adverse effects. Key points covered include the structures, spectra and uses of various penicillins; classifications and characteristics of cephalosporins; and brief descriptions of other cell wall synthesis inhibitors like carbapenems and glycopeptides.