Benzene can be prepared through several methods including:
1) Cyclic polymerization of ethyne which was first done by passing ethyne through a red hot iron tube.
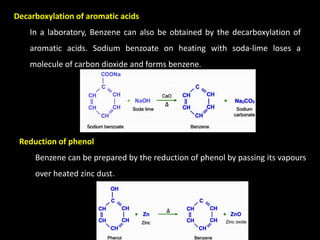
2) Decarboxylation of aromatic acids such as sodium benzoate.
3) Reduction of phenol by passing its vapors over heated zinc dust.
4) Hydrolysis of benzene sulphonic acid with superheated steam.
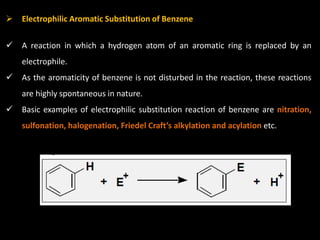
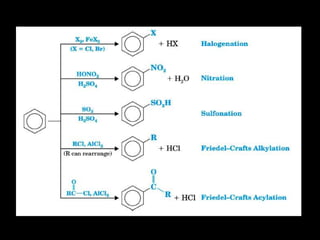
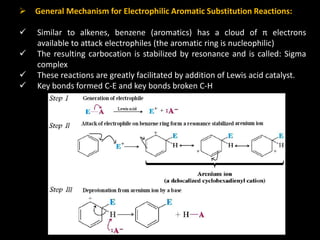
Benzene is a colorless, highly inflammable liquid with a characteristic smell and boiling point of 80°C. It undergoes electrophilic aromatic substitution reactions where a hydrogen is replaced by an electrophile such as halogen or nitrate which is facilitated by