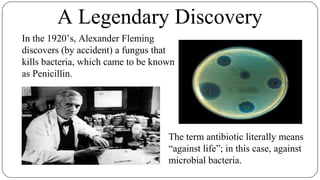
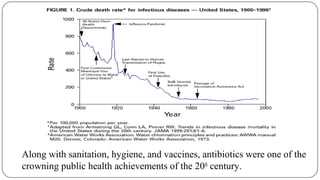
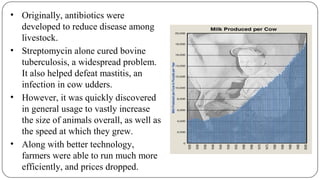
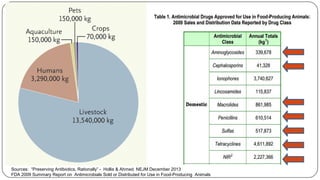
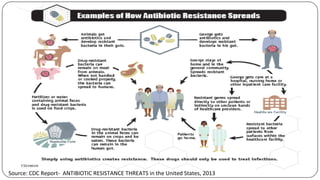
The document discusses the historical and current use of antibiotics in livestock, highlighting their initial use for disease control and subsequent overuse, leading to antibiotic resistance in both animals and humans. It outlines economic impacts, healthcare costs due to antibiotic-resistant infections, and the need for policy regulations to curb unnecessary antibiotic use in agriculture. The document emphasizes the importance of continued research and responsible practices to preserve antibiotic effectiveness for both human and animal health.