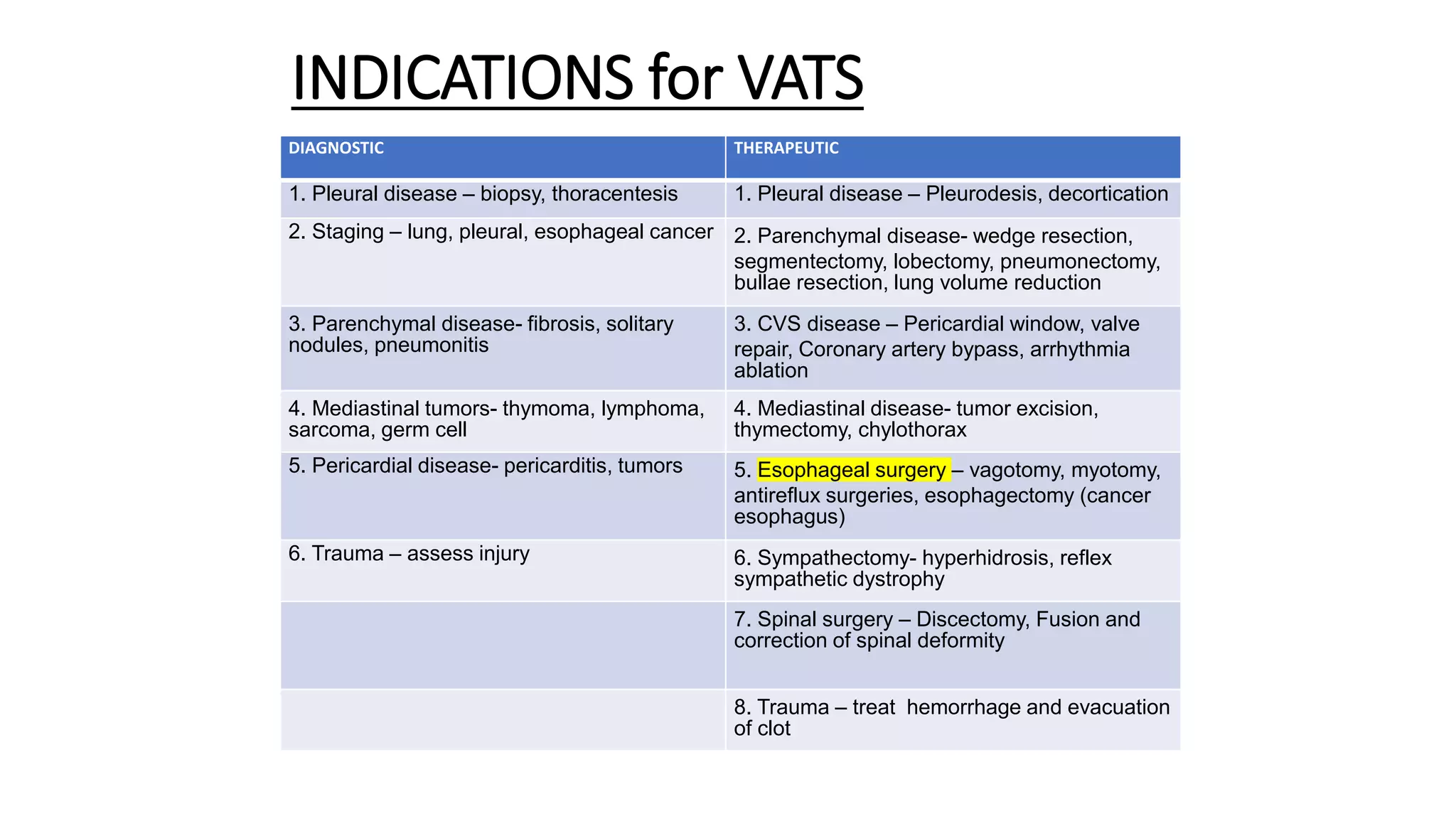
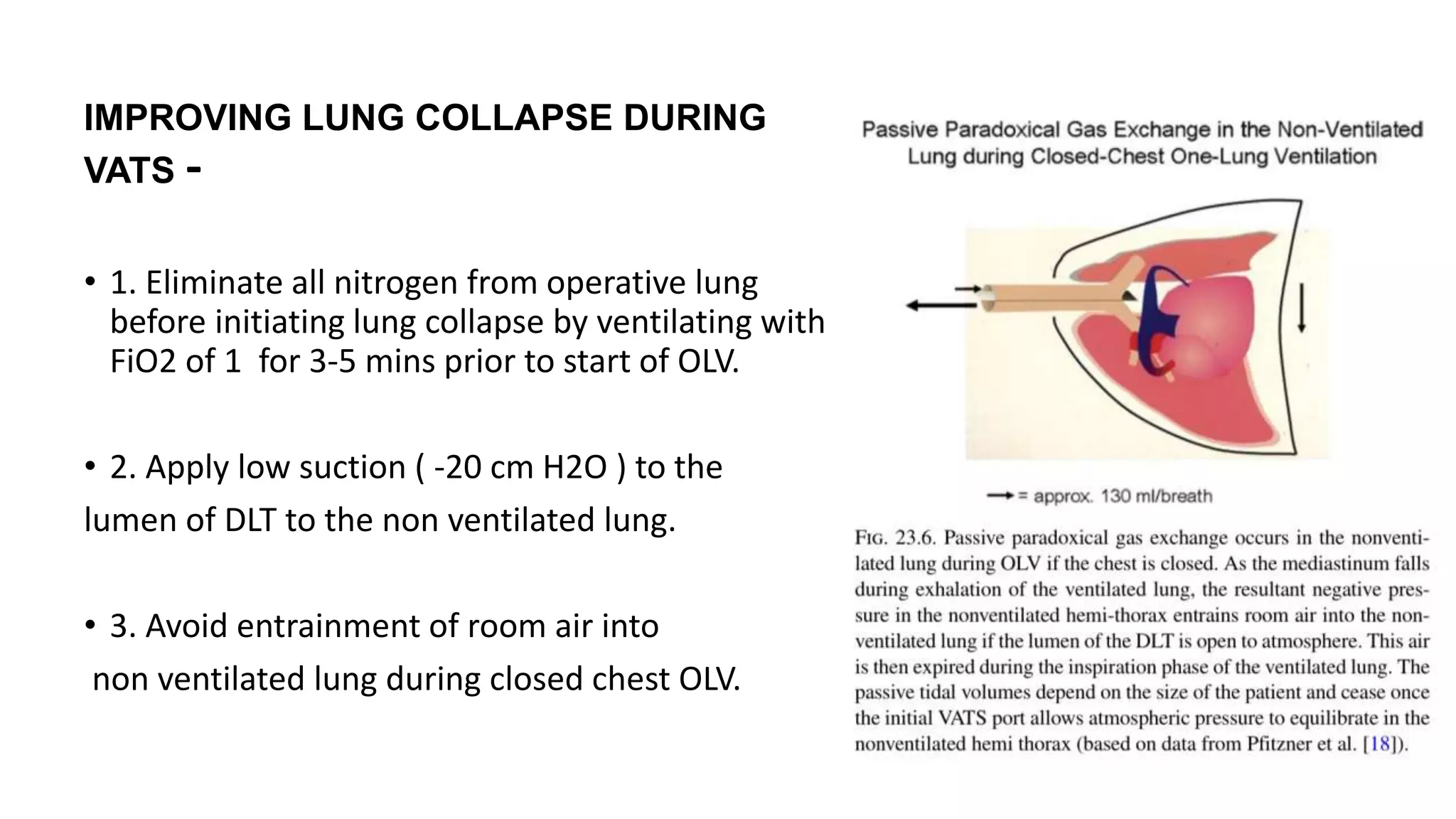
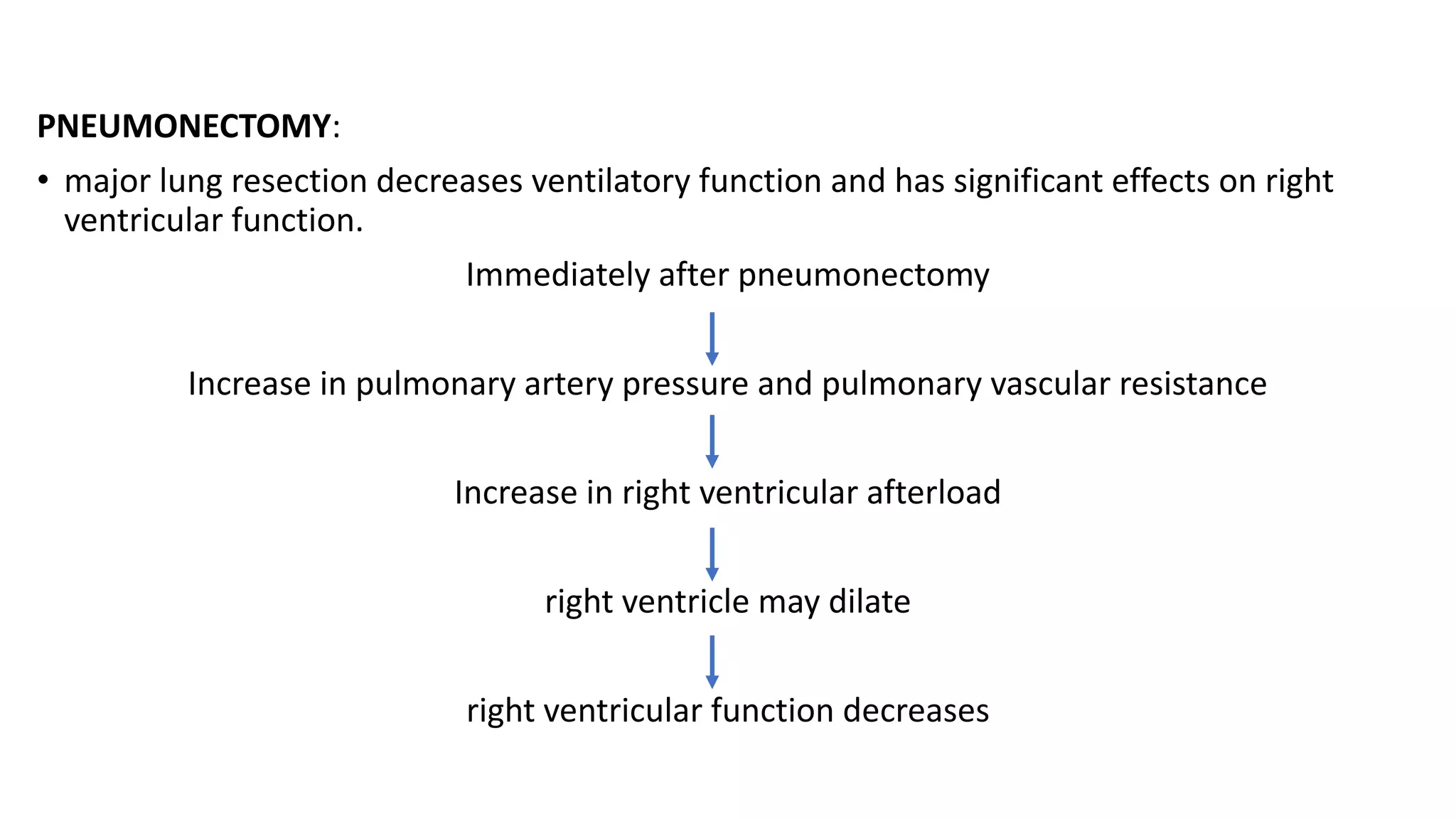
The document discusses anesthesia considerations for thoracoscopy and VATS procedures. It covers preoperative assessment and optimization, intraoperative anesthetic management including lung isolation techniques, ventilation strategies, positioning, and management of issues like hypoxemia. Protective lung ventilation principles with low tidal volumes, PEEP, and recruitment maneuvers are emphasized for lung protection during one-lung ventilation.