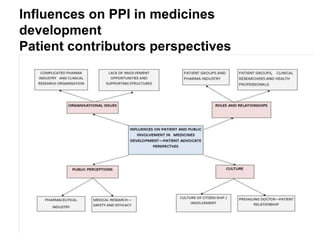
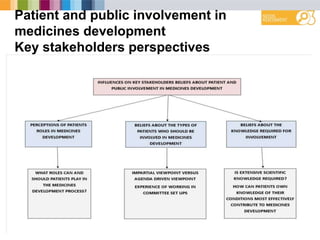
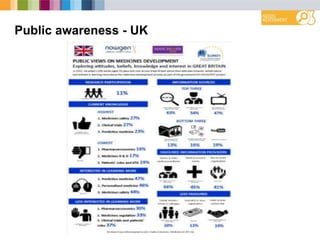
The document outlines a needs assessment conducted by Work Package 3 of EUPATI Nowgen at the University of Manchester, focusing on patient involvement in medicines development. It explores patient and stakeholder perspectives on knowledge, roles, and barriers to participation, while highlighting the complexities of relationships in the development process. The findings emphasize the importance of structured support for patient involvement and the value of shared experiences to enhance collaboration between patients and the pharmaceutical industry.