This document discusses potential environmental issues from dyeing processes. It notes that most emissions are to water, as substances in dye baths have low vapour pressure. Exceptions include thermosol dyeing and pigment dyeing where pollutants can be released to air. Emissions may include unfixed dye, auxiliaries, heavy metals, salts, and reducing/oxidizing agents. The document focuses on issues related to specific substances like reactive dyes with low fixation rates, dyes containing halogens contributing to AOX, and metals as dye impurities or structural elements. It also discusses toxicity concerns from things like sulphides, oxidizing agents, and the potential for some azo dyes to cleave into





![destructive techniques (e.g coagulation, adsorption).
However, it should be noted that AOX from dyes do not have
the same effect as the AOX derived from chlorine reactions
(haloform reaction, in particular) arising from textile
processes such as bleaching, wool shrink-resist treatments,
etc.
Dyestliffs are not biodegradable compounds and the
halogens in their molecule should not give rise the haloform
reaction (main cause of hazardous AOX). In this respect it is
interesting to consider that PARCOM 97/1 does not set a
general discharge limit value For AOX. but rather allows
discrimination between hazardous and non-hazardous AOX
[50,OSPAR, 1997].
Heavy metal emission Metals can be present in dyes for two
reasons. First, metals are used as catalysts during the
manufacture of some dyes and can be present as impurities.
Second, in some dyes the metal is chelated with the dye
molecule, forming an integral structural element.
Dye manufactutrers are now putting more effort into
reducing the amount of metals present as impurities. This
can be done by selection of starting products, removal of
heavy metal and substitution of the solvent where the
reaction takes place.
ETAD has established limits in the content of heavy metal in
dyestuffs. The values have been set to ensure that emission
levels from a 2 % dyeing and a total dilution of the dye of
1:2500, will meet the known waste water requirements [64,
BASF, 1994].
Examples of dyes containing bound metals are copper and
nickel in phthalocyanine groups, copper in blue copper-azo-
complex reactive dyes and chromium in metal-complex dyes
used for wool silk and polyamide. The total amount of
metallised dye used is decreasing, but there remain domains
(certain shades such as greens, certain levels of fastness to
light) where phthalocyanine dyes,, for example, cannot be
easily substituted.](https://image.slidesharecdn.com/2environmentalissuesindyeing-110711044039-phpapp01/85/2-environmental-issues-in-dyeing-6-320.jpg)
![The presence of the metal in these metallised dyes can be
regarded as a less relevant problem compared to the
presence of free metal impurities. Provided that high
exhaustion and Fixation levels are achieved and that
measures are taken to minimise losses from handling,
weighing, drum cleaning, etc., only a little unconsumed dye
should end up in the waste water. Moreover, since the metal
is an integral part of the dye molecule, which is itself non-
biodegradable, there is very little potential for it to become
bio-available.
It is also important to take into account that treatment
methods such as Filtration and adsorption on activated
sludge, which remove the dye from the waste water, also
reduce nearly proportionally the amount of bound metal in
the final effluent. Conversely, other methods such as
advance oxidation may free the metal.
Toxicitv Dyestuffs
showing aquatic toxicity and/ or allergenic effects are
highlighted in Section 9. Here it is also important to mention
that about 60 % to 70 % of the dyes used nowadays are azo
dyes [77, ELJRATEX. 2000]. Under reductive conditions,
these dyes may produce amines and some of them are
carcinogenic. A list of carcinogenic amines that can be
formed by cleavage of certain azo dves is shown in the Table
2.13. SO
81](https://image.slidesharecdn.com/2environmentalissuesindyeing-110711044039-phpapp01/85/2-environmental-issues-in-dyeing-7-320.jpg)
![Table 2.13: List of carcinogenic amines
The use of azo-dyes that may cleave to one of the 22
potentially carcinogenic aromatic amines listed above is
banned according to the 19th amendment of Directive
76/769/EWG on dangerous However, more than 100 dyes
with the potential to form carcinogenic amines are still
available on the market [77, EURATEX, 2000].
Auxiliaries contained in dye formulations
Depending on the dye class and the application method
employed (e.g. batch or continuous dyeing, printing)
different additives are present in the dye formulations. Since
these substances are not absorbed/ fixed by the fibres, they
are completely discharged in the waste water. Typical
additives are listed in the table below.
82](https://image.slidesharecdn.com/2environmentalissuesindyeing-110711044039-phpapp01/85/2-environmental-issues-in-dyeing-8-320.jpg)
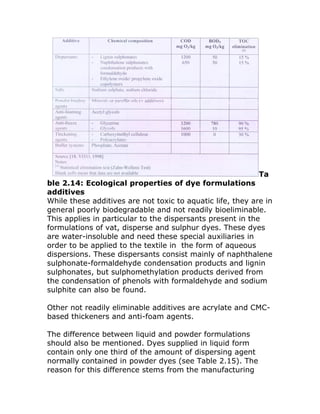



![In addition to the use of salt as raw material, neutralization
of commonly used acids and alkalis produces salts as a by-
products. Salts are not removed in conventional waste water
treatment systems and they are therefore ultimately
discharged in the receiving water.
Although the mammalian and aquatic toxicity of the
commonly employed salts are very low, in arid or semi-arid
regions their large-scale use can produce concentrations
above the toxic limit and increase the salinity of the
groundwater. Countries have set emission limits at 2000
ppm or below. River quality standards must also be taken
into account.
Carriers The use of these auxiliaries, which were widely
employed in the past, has now been reduced due to
ecological and health problems. They are still an issue in
dyeing of polyester in blend with wool.
Carriers may already be added to the dyes by
manufacturers. In this case textile finishers will have little
knowledge of the loads discharged ([4, Tebodin, 1991] and
[61, L. Bettens, 1999]).
Carriers (sec Section 8.6.7) include a wide group of organic
compounds, many of them steam volatile, poorlv
biodegradable and toxic to humans and aquatic life.
However, as the active substances usually have high affinity
for the fibre (hydrophobic types), 75 - 90 % are absorbed by
the textile and only the emulsifiers and the hydrophilic-type
carriers such as phenols and benzoate derivatives are found
in the waste water.
The carriers that remain on the fibre after dyeing and
washing, are partially volatilised during drying and fixing
operations and can give rise to air emissions. Traces can still
he found on the finished product, thus representing a
potential problem for the consumer. Alternative options are
described in Sections 4.6.1 and 4.6.2.
Other auxiliaries of environment interest](https://image.slidesharecdn.com/2environmentalissuesindyeing-110711044039-phpapp01/85/2-environmental-issues-in-dyeing-13-320.jpg)


![In conclusion, the use of low liquor ratio machinery, or
selection of the most adequate machine for the size of the
lot to be processed, is fundamental to the resultant
environmental performance of the process. Having said that.
high energy and water consumption in batch dyeing is not
only the result of high liquor ratios.
Another factor to take into consideration is the
discontinuous nature of the batch dyeing operating mode,
especially with regard to operations such as cooling,
heating, washing and rinsing.
Furthermore, shade matching can be responsible for higher
water and energy consumption, especially when dyeing is
carried out without the benefit of laboratory instruments. In
a manual regime the bulk of the dyestuff is normally applied
in the first phase to obtain a shade which is close to that
required in the final product. This is followed by a number of
matching operations, during which small quantities of dye
are applied to achieve the final shade. Shades which are
difficult to match may require repeated shade additions with
cooling and reheating between each addition [32, ENco,
2001].
Increased energy and water consumption may also be
caused by inappropriate handling techniques and/or poorly
performing process control systems. For example, in some
cases displacement spillage may occur during immersion of
the fibre in the machine, while the potential for overfilling
and spillage exists where the machines are only equipped
with manual control valves, which fail to control the liquor
level and temperature correctly (see also Section 4.1.4).
Continuous and semi-continuous dyeing processes
consume less water, but this also means a higher dyestuff
concentration in the dye liquor. In discontinuous dyeing the
dye concentration varies from O.I to I g/l, while in
continuous processes this value is in the range of 10 to 100
g/l. The residual padding liquor in the troughs, pumps and
pipes must be discarded when a new colour is started. The
discharge of this concentrated effluent can result in a higher](https://image.slidesharecdn.com/2environmentalissuesindyeing-110711044039-phpapp01/85/2-environmental-issues-in-dyeing-16-320.jpg)


![Pollutants that are likely to be encountered in waste water
are listed in the table below.
98
Ta
ble 2.17: Pollutants that are more likely to be encountered
in waste water from printing processes
Volatile organic compounds from drying and fixing
Drying and fixing are another important emission source in
printing processes. The following pollutants may be
encountered in the exhaust air [179, DBA, 2001]:
• aliphatic hydrocarbons (C10-C20) from binders
• Monomers such as acrylates, vinyl acetates, styrene,
acrylonitrile, methylol acrylamide, butadiene,
• • methanol from fixing agents](https://image.slidesharecdn.com/2environmentalissuesindyeing-110711044039-phpapp01/85/2-environmental-issues-in-dyeing-19-320.jpg)







![formaldehyde). Emissions, for example, of formaldehyde up
to 300 g/h (2 - 60 mg/m3) have been observed in some
cases, which were attributable to inefficient combustion of
the gas in the stenter frame [179, UBA, 2001].
It is therefore obvious - when speaking about air emissions -
that the environmental benefit obtained with the use of
formaldehyde-free finishing recipes is totally lost if the
burners in the stenter frames are poorly adjusted and
produce high formaldehyde emissions. The active
substances in the most common finishing agents and the
possible associated air emissions are discussed in Section
8.8. Moreover a more comprehensive list of pollutants that
can be found in the exhaust air from heat treatments in
general, is reported in Section 12.
Environmental issues associated with discontinuous
processes The application of functional finishes in long
liquor by means of batch processes is used mainly in varn
finishing and in the wool carpet yam industry in particular.
Since the functional finishes are generally applied either in
the dye baths or in the rinsing baths after dyeing,, this
operation does not entail additional water consumption with
respect to dyeing.
For the resulting water emissions, as with batch dyeing, the
efficiency of the transfer of the active substance from the
liquor to the fibre is the key factor which influences the
emission loads. The efficiency depends on the liquor ratio
znd on many parameters such as pH, temperature and the
type of emulsion (micro- or macro-emulsion).
Maximising the efficiency is particularly important when
biocides are applied in mothproofing finishing. As
mothproofing agents are not water-soluble they are applied
from emulsions. The degree of emulsification and the pH are
critical in the application of mothproofing agents (i.e. the
efficiency of the process is higher when the active substance](https://image.slidesharecdn.com/2environmentalissuesindyeing-110711044039-phpapp01/85/2-environmental-issues-in-dyeing-27-320.jpg)
