14.2 boltzmann factor
•Download as PPT, PDF•
0 likes•1,692 views
The document discusses the derivation of the Boltzmann factor e^(-ε/kT). It explains that processes occur when a particle's energy ε is a multiple of the average kinetic energy kT per particle, such as 20-30 times kT. While unlikely, particles can gain this extra energy through random collisions with other particles. The chances of gaining enough energy to overcome an energy barrier ε decreases exponentially with increasing ε/kT according to the Boltzmann factor. Therefore, the rate of a reaction is proportional to e^(-ε/kT).
Report
Share
Report
Share
Recommended
Fun with EMC2

1) A liter of water contains enough mass to produce 9 x 1016 Joules of energy if converted entirely, which could power a 1 Watt lightbulb for around 3 billion years.
2) The sun produces its energy through nuclear fusion, where hydrogen nuclei fuse into helium. A little bit of mass is lost in this process, appearing as energy according to E=mc2.
3) Scientists are trying to create controlled nuclear fusion on Earth for a sustainable energy source, but it requires maintaining temperatures over 15 million degrees, which is technically challenging.
Prasanth( e=mc2)

This document discusses Einstein's theory of mass-energy equivalence, E=mc2. It begins by explaining the formula and its implications, such as that mass and energy are interchangeable. It then provides examples of nuclear reactions like fusion and fission that release energy. Applications discussed include energy production in the sun via nuclear fusion and the development of atomic weapons. The document emphasizes how scientific achievements advance quality of life but also calls for responsible use of technology.
Nuclear Forces Pp

Nuclear forces keep nucleons together through powerful short-range attraction between protons and neutrons. The mass of a nucleus is less than the total mass of its constituent particles due to the nuclear mass defect. This mass defect is converted to nuclear binding energy, which is released when the nucleus forms. Nuclei with intermediate atomic weights have the greatest stability and binding energy per particle, while the lightest and heaviest nuclei are less stable.
Nuclear force

The document discusses the four fundamental forces: gravitational, electromagnetic, nuclear, and weak. It summarizes that the nuclear force was discovered after neutrons were discovered in 1932, and holds nucleons together in the nucleus. The nuclear force is charge independent, very strong but short range, repulsive at short distances, and acts through the exchange of pions between nucleons. The document provides details on the Yukawa potential and uncertainty principle as they relate to the nuclear force. It poses a multiple choice question about identifying an incorrect statement regarding the nuclear force.
Nuclear force 22.10.12

The nuclear force is responsible for binding protons and neutrons into atomic nuclei. It is powerfully attractive between nucleons at distances around 1 femtometer but rapidly decreases beyond 2.5 femtometers. At very short distances less than 0.7 femtometers, the nuclear force becomes repulsive. The nuclear force is mediated by the exchange of pi mesons between the pion clouds that surround the quark cores of protons and neutrons.
Martin Lindsey MSIMatter Presentation

This is the presentation I gave during a group interview for my position as a Fab Lab Facilitator at the Museum of Science and Industry in Chicago
Electric charges and fields 

COMPARING ELECTROSTATIC AND GRAVITATIONAL FORCES.
Electrostatic forces gravitational force. Electrostatic is the force of attraction or repulsion between two charges at rest while the gravitational force is the force of attraction between two bodies by virtue of their masses. masses.
https://bdslearningapp.blogspot.com/2020/09/electric-charges-and-fields_10.html
Magnetar

Magnetars are neutron stars with extremely powerful magnetic fields. This document calculates the maximum magnetic field at the surface of the magnetar SGR1806-20, which has a rotation period of 7.5 seconds and is slowing down rapidly. Assuming it is a standard neutron star, the calculation finds the surface magnetic field to be 1.7×1015 gauss, which is 38 times stronger than the quantum critical field above which electron-positron pair creation becomes highly probable. This explains how magnetars can emit energetic x-rays and gamma rays.
Recommended
Fun with EMC2

1) A liter of water contains enough mass to produce 9 x 1016 Joules of energy if converted entirely, which could power a 1 Watt lightbulb for around 3 billion years.
2) The sun produces its energy through nuclear fusion, where hydrogen nuclei fuse into helium. A little bit of mass is lost in this process, appearing as energy according to E=mc2.
3) Scientists are trying to create controlled nuclear fusion on Earth for a sustainable energy source, but it requires maintaining temperatures over 15 million degrees, which is technically challenging.
Prasanth( e=mc2)

This document discusses Einstein's theory of mass-energy equivalence, E=mc2. It begins by explaining the formula and its implications, such as that mass and energy are interchangeable. It then provides examples of nuclear reactions like fusion and fission that release energy. Applications discussed include energy production in the sun via nuclear fusion and the development of atomic weapons. The document emphasizes how scientific achievements advance quality of life but also calls for responsible use of technology.
Nuclear Forces Pp

Nuclear forces keep nucleons together through powerful short-range attraction between protons and neutrons. The mass of a nucleus is less than the total mass of its constituent particles due to the nuclear mass defect. This mass defect is converted to nuclear binding energy, which is released when the nucleus forms. Nuclei with intermediate atomic weights have the greatest stability and binding energy per particle, while the lightest and heaviest nuclei are less stable.
Nuclear force

The document discusses the four fundamental forces: gravitational, electromagnetic, nuclear, and weak. It summarizes that the nuclear force was discovered after neutrons were discovered in 1932, and holds nucleons together in the nucleus. The nuclear force is charge independent, very strong but short range, repulsive at short distances, and acts through the exchange of pions between nucleons. The document provides details on the Yukawa potential and uncertainty principle as they relate to the nuclear force. It poses a multiple choice question about identifying an incorrect statement regarding the nuclear force.
Nuclear force 22.10.12

The nuclear force is responsible for binding protons and neutrons into atomic nuclei. It is powerfully attractive between nucleons at distances around 1 femtometer but rapidly decreases beyond 2.5 femtometers. At very short distances less than 0.7 femtometers, the nuclear force becomes repulsive. The nuclear force is mediated by the exchange of pi mesons between the pion clouds that surround the quark cores of protons and neutrons.
Martin Lindsey MSIMatter Presentation

This is the presentation I gave during a group interview for my position as a Fab Lab Facilitator at the Museum of Science and Industry in Chicago
Electric charges and fields 

COMPARING ELECTROSTATIC AND GRAVITATIONAL FORCES.
Electrostatic forces gravitational force. Electrostatic is the force of attraction or repulsion between two charges at rest while the gravitational force is the force of attraction between two bodies by virtue of their masses. masses.
https://bdslearningapp.blogspot.com/2020/09/electric-charges-and-fields_10.html
Magnetar

Magnetars are neutron stars with extremely powerful magnetic fields. This document calculates the maximum magnetic field at the surface of the magnetar SGR1806-20, which has a rotation period of 7.5 seconds and is slowing down rapidly. Assuming it is a standard neutron star, the calculation finds the surface magnetic field to be 1.7×1015 gauss, which is 38 times stronger than the quantum critical field above which electron-positron pair creation becomes highly probable. This explains how magnetars can emit energetic x-rays and gamma rays.
6563.nuclear models

1. Nuclear models like the liquid drop model and shell model describe aspects of nuclear structure and behavior. The liquid drop model treats the nucleus like a liquid drop while the shell model treats nucleons as moving independently in nuclear orbits.
2. The shell model explains nuclear magic numbers and properties like spin and parity. Magic numbers correspond to nuclear stability when the number of protons or neutrons equals 2, 8, 20, 28, 50, 82, etc. The shell model accounts for magic numbers in terms of closed nuclear shells.
3. While insightful, nuclear models have limitations and do not fully describe all nuclear phenomena. The liquid drop model cannot explain magic numbers while the shell model fails to explain the stability of certain
Notes lab 04b the atom structure

Elements are made of atoms, which are the basic building blocks of matter. Atoms consist of protons, neutrons, and electrons. Protons and neutrons are located in the nucleus, while electrons surround the nucleus in electron clouds. Protons are positively charged, electrons are negatively charged, and neutrons have no charge. The number of protons determines the element and its atomic number. The total number of protons and neutrons determines the atom's mass number. Elements are organized on the periodic table based on their atomic structure.
Plasma

This document discusses plasma oscillations in a highly ionized gas. It describes how an initial uniform distribution of electrons and ions can become perturbed and accumulate, changing their densities. This accumulation can cause the electrons and ions to oscillate around their equilibrium positions. The document derives an equation showing the evolution of this density perturbation with time, which takes the form of a harmonic oscillator with the plasma frequency wp. Plasma oscillations at frequencies below wp will be reflected, while above wp waves can propagate freely through the plasma.
Effcharge

The document discusses the structure of an atom's nucleus which contains protons and neutrons. Electrons orbit the nucleus in shells labeled with numbers and letters, with lower shells feeling a stronger pull from the nucleus due to less shielding from other electrons. The effective nuclear charge decreases for electrons in higher, farther shells that experience more shielding from inner electrons.
Mass and energy

In fission, the mass of a U-235 nucleus is less after splitting than before, with the missing mass converted to energy. In pair production, a high-energy photon disappears and an electron-positron pair is created, with energy converted to mass. Both involve mass-energy interconversion - fission converts mass to energy while pair production converts energy to mass. Every particle has a rest mass when stationary, and moving particles effectively have more mass due to their kinetic energy. Rest energy is the energy equivalent of a particle's rest mass.
Unit 4 - Foundations of Astronomy

The document outlines key concepts in astronomy and physics, including the scientific method, fundamental forces, elementary particles, and atomic structure. It discusses the four fundamental forces (gravitational, electromagnetic, strong, and weak), elementary particles like protons, neutrons, electrons, and quarks, and provides classical and modern views of atomic structure. Key terms are defined related to these topics.
Big bang diagram travis tricker

This document provides a timeline of major events in the early universe from the Planck epoch shortly after the Big Bang to the present day. It describes the unification of fundamental forces, phase transitions as the universe expanded and cooled, nucleosynthesis forming new atomic nuclei, recombination of hydrogen and helium atoms, formation of the first galaxies, and notes that today life exists on Earth within the solar system alongside distant quasars.
History of the universe

The document describes the evolution of the universe from the Big Bang to the present day in three main epochs: the photon epoch, where photons took over the mass of the universe around 380,000 years after the Big Bang; the lepton epoch, where leptons took over the mass around 1 second after the Big Bang; and the quark epoch, where gravitation and electromagnetism began to take form around 10-10 seconds after the Big Bang. It outlines how the early universe transitioned from a high pressure, high density singularity at the moment of the Big Bang to forming the first light elements like hydrogen and helium and eventually galaxies and stars over billions of years as it expanded and cooled.
chemistry of radiation nuclear activatin

The document discusses various types of ionizing radiation, their properties and interactions with matter. It describes the dual wave-particle nature of radiation and defines key terms like half-value layer, linear attenuation coefficient, and interaction mechanisms including the photoelectric effect, Compton scattering, pair production, and bremsstrahlung. It also covers particulate radiations like electrons and neutrons, and their penetration and energy deposition in tissues.
Interaction of ionizing

Ionizing radiation interacts with matter by ejecting electrons through processes like ionization and excitation. The three main interaction processes between photons and atoms are the photoelectric effect, Compton effect, and pair production. The probability of each interaction depends on the photon energy and atomic number of the absorbing material. Charged particles like electrons and protons mainly interact through ionization and excitation via radiative collisions.
rad-onc-matney-interactions.pdf

The document discusses various interactions between photons and matter, including ionizing and non-ionizing interactions. It describes in detail photon interactions such as the photoelectric effect, Compton scattering, and pair production. It explains how these interactions work and how their probability depends on factors like the photon energy and atomic number (Z) of the absorbing material. Characteristics of the interactions like half value layer and tenth value layer used in shielding calculations are also covered.
Interaction of radiation with Matter - Dr. Vandana

The document discusses the interaction of radiation with matter. It describes the various types of interactions including photoelectric effect, Compton scattering, pair production and their dependence on photon energy. It also discusses the linear attenuation coefficient, half value layer, mass attenuation coefficient and energy absorption coefficient. The different effects of ionizing and non-ionizing radiation are summarized along with the radiobiological implications of radiation interactions.
Interaction_photon_electron_with_matter.ppt

The document discusses various interactions of photons with matter. It describes three main interactions: the photoelectric effect, Compton scattering, and pair production. It provides details on the energy thresholds, probabilities, and products of these interactions. It also discusses attenuation of photons in materials and defines half value thickness and tenth value thickness. In addition, it summarizes interactions of charged particles and neutrons with matter, including ionization, bremsstrahlung, stopping power, linear energy transfer, and shielding considerations.
Size effects

The document discusses how the surface-to-volume ratio affects the properties of nanomaterials. It explains that nanomaterials have an extremely high surface area to volume ratio compared to larger materials, meaning the surface plays a larger role in determining properties. Different shapes like spheres, cylinders, and cubes are examined, showing how their surface-to-volume ratios change with size. The large increase in surface area for a given volume is demonstrated by reducing a 10 micrometer particle to billions of 10 nanometer particles, increasing the surface area by a factor of 1000. The high surface-to-volume ratio is a key factor making nanomaterial properties dependent on surface effects.
Thoughts of thermoelectric material

Thermoelectric materials can convert waste heat into electricity, helping address issues of declining fossil fuels and global warming. Researchers aim to improve the figure of merit (ZT) of these materials, which indicates efficiency - the best currently have ZT between 2-3 but 4-10 could reach 15-20% efficiency. The key is increasing the power factor through properties like the Seebeck coefficient while decreasing the thermal conductivity, such as through nanostructuring, alloying, or composites to scatter phonons transporting heat. Continued research optimizes these various material parameters to further enhance thermoelectric performance.
Elementsofstatisticalmechanics

1) Statistical mechanics deals with relating the macroscopic behavior of a system to the microscopic properties of its particles. A system's macrostate is defined by the distribution of particles among compartments, while each distinct microscopic arrangement is a microstate.
2) Phase space combines position and momentum space, specifying the complete state of a system. For classical particles, the Maxwell-Boltzmann distribution describes average particle numbers. Quantum statistics include Bose-Einstein and Fermi-Dirac distributions.
3) A photon gas in an enclosure reaches thermal equilibrium where the Bose-Einstein distribution applies. The number of photon energy states is calculated from phase space considerations.
Compton effect and pair production

The document discusses various types of ionizing radiation and their interactions with matter. It describes electromagnetic radiation as composed of photons that can interact via photoelectric effect, Compton scattering, pair production, and other processes. Compton scattering results in energy transfer between photons and recoil electrons. The probability of interaction depends on photon energy and material properties like atomic number. Higher energy photons have a greater chance of depositing energy through secondary electrons.
Charged particle interaction with matter

This document discusses charged particle interactions with matter. It begins by outlining the topics to be covered, including interactions of heavy charged particles like protons, electrons, and light ions. It then explains that charged particle interactions are mediated by Coulomb forces and may involve ionization or excitation of orbital electrons or interactions with atomic nuclei. Different types of interactions like elastic and inelastic collisions are described. Equations for energy loss by heavy charged particles during collisions are shown. The interactions of protons, electrons, neutrons, and light and heavy ions are then discussed in more detail.
3 thermodynamics fall Energy 101 fall 2015

1. The document discusses the laws of thermodynamics, including that energy cannot be created or destroyed according to the first law, but tends to disperse and lose usefulness according to the second law of increasing entropy. The third law states that at absolute zero, entropy approaches zero and all activity ceases.
2. Solar energy provides a high quality energy source to offset the increasing entropy of systems on Earth and allows for regeneration through photosynthesis. Meditation also offsets entropy effects in the mind and body.
3. A deep understanding of thermodynamics illustrates why perpetual motion is impossible and why energy must be continually replenished to maintain order in systems.
Interactions of radiation_with_matter

This document discusses the interactions of various types of radiation with matter. It describes five main interactions that x-rays and gamma rays can undergo: photoelectric effect, Compton scattering, coherent scattering, pair production, and photodisintegration. It explains that the photoelectric effect and Compton scattering are most important for diagnostic and therapeutic radiology respectively. It also discusses the interactions of particulate radiation such as electrons, protons, neutrons, alpha particles, and heavy ions with matter and how they deposit energy.
Interaction of radiation with matter.pptx

1. When photons interact with matter, they can transfer energy to electrons through ionization or excitation, ejecting electrons from atoms.
2. The major interactions of photons with matter are photoelectric effect, Compton scattering, pair production, and photodisintegration. These depend on photon energy and atomic number (Z) of the absorbing medium.
3. Charged particles like electrons and protons primarily cause ionization through collisions with atoms. Neutrons can cause recoiling or nuclear disintegrations upon interaction.
CHAPTER 10 Molecules and Solids

CHAPTER 10 Molecules and Solids
10.1 Molecular Bonding and Spectra
10.2 Stimulated Emission and Lasers
10.3 Structural Properties of Solids
10.4 Thermal and Magnetic Properties of Solids
10.5 Superconductivity
10.6 Applications of Superconductivity
More Related Content
What's hot
6563.nuclear models

1. Nuclear models like the liquid drop model and shell model describe aspects of nuclear structure and behavior. The liquid drop model treats the nucleus like a liquid drop while the shell model treats nucleons as moving independently in nuclear orbits.
2. The shell model explains nuclear magic numbers and properties like spin and parity. Magic numbers correspond to nuclear stability when the number of protons or neutrons equals 2, 8, 20, 28, 50, 82, etc. The shell model accounts for magic numbers in terms of closed nuclear shells.
3. While insightful, nuclear models have limitations and do not fully describe all nuclear phenomena. The liquid drop model cannot explain magic numbers while the shell model fails to explain the stability of certain
Notes lab 04b the atom structure

Elements are made of atoms, which are the basic building blocks of matter. Atoms consist of protons, neutrons, and electrons. Protons and neutrons are located in the nucleus, while electrons surround the nucleus in electron clouds. Protons are positively charged, electrons are negatively charged, and neutrons have no charge. The number of protons determines the element and its atomic number. The total number of protons and neutrons determines the atom's mass number. Elements are organized on the periodic table based on their atomic structure.
Plasma

This document discusses plasma oscillations in a highly ionized gas. It describes how an initial uniform distribution of electrons and ions can become perturbed and accumulate, changing their densities. This accumulation can cause the electrons and ions to oscillate around their equilibrium positions. The document derives an equation showing the evolution of this density perturbation with time, which takes the form of a harmonic oscillator with the plasma frequency wp. Plasma oscillations at frequencies below wp will be reflected, while above wp waves can propagate freely through the plasma.
Effcharge

The document discusses the structure of an atom's nucleus which contains protons and neutrons. Electrons orbit the nucleus in shells labeled with numbers and letters, with lower shells feeling a stronger pull from the nucleus due to less shielding from other electrons. The effective nuclear charge decreases for electrons in higher, farther shells that experience more shielding from inner electrons.
Mass and energy

In fission, the mass of a U-235 nucleus is less after splitting than before, with the missing mass converted to energy. In pair production, a high-energy photon disappears and an electron-positron pair is created, with energy converted to mass. Both involve mass-energy interconversion - fission converts mass to energy while pair production converts energy to mass. Every particle has a rest mass when stationary, and moving particles effectively have more mass due to their kinetic energy. Rest energy is the energy equivalent of a particle's rest mass.
Unit 4 - Foundations of Astronomy

The document outlines key concepts in astronomy and physics, including the scientific method, fundamental forces, elementary particles, and atomic structure. It discusses the four fundamental forces (gravitational, electromagnetic, strong, and weak), elementary particles like protons, neutrons, electrons, and quarks, and provides classical and modern views of atomic structure. Key terms are defined related to these topics.
Big bang diagram travis tricker

This document provides a timeline of major events in the early universe from the Planck epoch shortly after the Big Bang to the present day. It describes the unification of fundamental forces, phase transitions as the universe expanded and cooled, nucleosynthesis forming new atomic nuclei, recombination of hydrogen and helium atoms, formation of the first galaxies, and notes that today life exists on Earth within the solar system alongside distant quasars.
History of the universe

The document describes the evolution of the universe from the Big Bang to the present day in three main epochs: the photon epoch, where photons took over the mass of the universe around 380,000 years after the Big Bang; the lepton epoch, where leptons took over the mass around 1 second after the Big Bang; and the quark epoch, where gravitation and electromagnetism began to take form around 10-10 seconds after the Big Bang. It outlines how the early universe transitioned from a high pressure, high density singularity at the moment of the Big Bang to forming the first light elements like hydrogen and helium and eventually galaxies and stars over billions of years as it expanded and cooled.
What's hot (8)
Similar to 14.2 boltzmann factor
chemistry of radiation nuclear activatin

The document discusses various types of ionizing radiation, their properties and interactions with matter. It describes the dual wave-particle nature of radiation and defines key terms like half-value layer, linear attenuation coefficient, and interaction mechanisms including the photoelectric effect, Compton scattering, pair production, and bremsstrahlung. It also covers particulate radiations like electrons and neutrons, and their penetration and energy deposition in tissues.
Interaction of ionizing

Ionizing radiation interacts with matter by ejecting electrons through processes like ionization and excitation. The three main interaction processes between photons and atoms are the photoelectric effect, Compton effect, and pair production. The probability of each interaction depends on the photon energy and atomic number of the absorbing material. Charged particles like electrons and protons mainly interact through ionization and excitation via radiative collisions.
rad-onc-matney-interactions.pdf

The document discusses various interactions between photons and matter, including ionizing and non-ionizing interactions. It describes in detail photon interactions such as the photoelectric effect, Compton scattering, and pair production. It explains how these interactions work and how their probability depends on factors like the photon energy and atomic number (Z) of the absorbing material. Characteristics of the interactions like half value layer and tenth value layer used in shielding calculations are also covered.
Interaction of radiation with Matter - Dr. Vandana

The document discusses the interaction of radiation with matter. It describes the various types of interactions including photoelectric effect, Compton scattering, pair production and their dependence on photon energy. It also discusses the linear attenuation coefficient, half value layer, mass attenuation coefficient and energy absorption coefficient. The different effects of ionizing and non-ionizing radiation are summarized along with the radiobiological implications of radiation interactions.
Interaction_photon_electron_with_matter.ppt

The document discusses various interactions of photons with matter. It describes three main interactions: the photoelectric effect, Compton scattering, and pair production. It provides details on the energy thresholds, probabilities, and products of these interactions. It also discusses attenuation of photons in materials and defines half value thickness and tenth value thickness. In addition, it summarizes interactions of charged particles and neutrons with matter, including ionization, bremsstrahlung, stopping power, linear energy transfer, and shielding considerations.
Size effects

The document discusses how the surface-to-volume ratio affects the properties of nanomaterials. It explains that nanomaterials have an extremely high surface area to volume ratio compared to larger materials, meaning the surface plays a larger role in determining properties. Different shapes like spheres, cylinders, and cubes are examined, showing how their surface-to-volume ratios change with size. The large increase in surface area for a given volume is demonstrated by reducing a 10 micrometer particle to billions of 10 nanometer particles, increasing the surface area by a factor of 1000. The high surface-to-volume ratio is a key factor making nanomaterial properties dependent on surface effects.
Thoughts of thermoelectric material

Thermoelectric materials can convert waste heat into electricity, helping address issues of declining fossil fuels and global warming. Researchers aim to improve the figure of merit (ZT) of these materials, which indicates efficiency - the best currently have ZT between 2-3 but 4-10 could reach 15-20% efficiency. The key is increasing the power factor through properties like the Seebeck coefficient while decreasing the thermal conductivity, such as through nanostructuring, alloying, or composites to scatter phonons transporting heat. Continued research optimizes these various material parameters to further enhance thermoelectric performance.
Elementsofstatisticalmechanics

1) Statistical mechanics deals with relating the macroscopic behavior of a system to the microscopic properties of its particles. A system's macrostate is defined by the distribution of particles among compartments, while each distinct microscopic arrangement is a microstate.
2) Phase space combines position and momentum space, specifying the complete state of a system. For classical particles, the Maxwell-Boltzmann distribution describes average particle numbers. Quantum statistics include Bose-Einstein and Fermi-Dirac distributions.
3) A photon gas in an enclosure reaches thermal equilibrium where the Bose-Einstein distribution applies. The number of photon energy states is calculated from phase space considerations.
Compton effect and pair production

The document discusses various types of ionizing radiation and their interactions with matter. It describes electromagnetic radiation as composed of photons that can interact via photoelectric effect, Compton scattering, pair production, and other processes. Compton scattering results in energy transfer between photons and recoil electrons. The probability of interaction depends on photon energy and material properties like atomic number. Higher energy photons have a greater chance of depositing energy through secondary electrons.
Charged particle interaction with matter

This document discusses charged particle interactions with matter. It begins by outlining the topics to be covered, including interactions of heavy charged particles like protons, electrons, and light ions. It then explains that charged particle interactions are mediated by Coulomb forces and may involve ionization or excitation of orbital electrons or interactions with atomic nuclei. Different types of interactions like elastic and inelastic collisions are described. Equations for energy loss by heavy charged particles during collisions are shown. The interactions of protons, electrons, neutrons, and light and heavy ions are then discussed in more detail.
3 thermodynamics fall Energy 101 fall 2015

1. The document discusses the laws of thermodynamics, including that energy cannot be created or destroyed according to the first law, but tends to disperse and lose usefulness according to the second law of increasing entropy. The third law states that at absolute zero, entropy approaches zero and all activity ceases.
2. Solar energy provides a high quality energy source to offset the increasing entropy of systems on Earth and allows for regeneration through photosynthesis. Meditation also offsets entropy effects in the mind and body.
3. A deep understanding of thermodynamics illustrates why perpetual motion is impossible and why energy must be continually replenished to maintain order in systems.
Interactions of radiation_with_matter

This document discusses the interactions of various types of radiation with matter. It describes five main interactions that x-rays and gamma rays can undergo: photoelectric effect, Compton scattering, coherent scattering, pair production, and photodisintegration. It explains that the photoelectric effect and Compton scattering are most important for diagnostic and therapeutic radiology respectively. It also discusses the interactions of particulate radiation such as electrons, protons, neutrons, alpha particles, and heavy ions with matter and how they deposit energy.
Interaction of radiation with matter.pptx

1. When photons interact with matter, they can transfer energy to electrons through ionization or excitation, ejecting electrons from atoms.
2. The major interactions of photons with matter are photoelectric effect, Compton scattering, pair production, and photodisintegration. These depend on photon energy and atomic number (Z) of the absorbing medium.
3. Charged particles like electrons and protons primarily cause ionization through collisions with atoms. Neutrons can cause recoiling or nuclear disintegrations upon interaction.
CHAPTER 10 Molecules and Solids

CHAPTER 10 Molecules and Solids
10.1 Molecular Bonding and Spectra
10.2 Stimulated Emission and Lasers
10.3 Structural Properties of Solids
10.4 Thermal and Magnetic Properties of Solids
10.5 Superconductivity
10.6 Applications of Superconductivity
Chapter 6 Thermally Activated Process and Diffusion in Solids.

This document discusses thermally activated processes and diffusion in solids. It covers topics like rate processes, the probability of atoms acquiring activation energy, vacancy and interstitial diffusion mechanisms, and industrial applications of diffusion like surface hardening and integrated circuits. The key points are:
- Reactions in solids require atoms to gain enough energy to overcome activation energy barriers. Higher temperatures provide more energy.
- Diffusion occurs through vacancy or interstitial mechanisms as atoms move into vacant spaces in the crystal lattice. It is described by equations like Fick's laws.
- Industrial uses of diffusion include carburizing steel surfaces to introduce carbon and dope silicon wafers with impurities to create integrated circuits. Diffusion
Radiation Interactions.ppt

This document discusses radiation interactions with matter. It describes how different types of ionizing radiation interact with and deposit energy in matter. Photons can interact via photoelectric effect, Compton scattering, and pair production. Charged particles like electrons cause ionization and excitation as they pass through matter. Neutrons interact with nuclei via collisions or nuclear disintegration. The effectiveness of different types of interactions depends on the energy of the radiation and the atomic properties of the absorbing material. Depth dose profiles are compared for different particle beams like photons, electrons, protons, and neutrons.
Atomic and nuclear physics

The document discusses several topics in atomic and nuclear physics including:
1) The photoelectric effect describes how light shining on a metal surface can eject electrons. Compton scattering demonstrates that X-rays lose energy when scattered by electrons, showing the particle nature of light.
2) X-rays are produced when high-energy electrons collide with atoms, either ejecting inner electrons or through braking radiation when deflected by the nucleus.
3) Light exhibits both wave and particle properties in experiments like the photoelectric effect, Compton scattering, and the Young's interference experiment, known as wave-particle duality.
4) Electrons falling between energy levels emit photons with energy equal to the level difference
Collision theory and Boltzmann

Higher temperatures increase the rate of reaction by providing particles with more kinetic energy, leading to more frequent and more energetic collisions that are more likely to exceed the activation energy needed for the reaction to occur. Specifically, for every 10 degree C rise in temperature, the rate of reaction doubles and the time taken halves, as increased particle energy from heat causes more collisions per unit time and a greater chance of particles reaching the activation energy required to react upon colliding.
lecture_6_20.ppt

This document discusses the interactions of ionizing radiation with matter. It begins by introducing the objectives of understanding how radiation interacts with matter and the effects on physical, chemical, and biological levels. It then covers the three main interactions of photons (photoelectric effect, Compton scattering, pair production) and how they lead to attenuation. Key points include how probability depends on photon energy and atomic number, and the byproducts of each interaction. It also discusses the interactions of particulate radiation like electrons, protons, neutrons, and how they ionize matter through excitation, ionization, and bremsstrahlung. Factors like linear energy transfer and specific ionization are addressed. The document is intended to provide background knowledge on radiation detection and
Laser lecture01

This document provides an introduction to lasers and their applications. It begins with recommended textbooks on the subject, then provides a chart showing the laser spectrum and examples of different laser types and their wavelengths. The remainder of the document discusses the basic components and functioning of lasers, including the gain medium that provides stimulated emission, the pump source to create population inversion, and the optical cavity formed by mirrors. It also provides brief histories of the development of masers and the first ruby laser.
Similar to 14.2 boltzmann factor (20)
Interaction of radiation with Matter - Dr. Vandana

Interaction of radiation with Matter - Dr. Vandana
Chapter 6 Thermally Activated Process and Diffusion in Solids.

Chapter 6 Thermally Activated Process and Diffusion in Solids.
More from mrrayner
7La/1

The document summarizes key facts about the Earth:
1) The planet we live on is called Earth. It gets its heat and light from the Sun. The Moon is Earth's natural satellite and orbits Earth once every 28 days.
2) A diagram labels that one side of Earth experiences nighttime while the other experiences daytime due to its rotation on its axis.
3) Additional facts provided are that Earth's year has 365 days, a leap year occurs every 4 years, Earth orbits the Sun in 365.25 days, and Earth completes one spin every 24 hours.
7 l1 the earth in space

A day is the time it takes for the Earth to spin on its axis. A month is the time it takes for the Moon to orbit the Earth. A year is the time it takes for the Earth to orbit once around the Sun. The document contains information about the definitions of a day, month, and year according to the movement and positions of the Earth, Moon, and Sun in space. It also lists learning objectives about describing and explaining the motions that cause days, months, years, and seasons.
7 l target sheet

This document outlines learning objectives related to understanding the motion of objects in space, including: how the Earth revolves around the sun and rotates on its axis, causing day/night and seasons; how the tilt of the Earth's axis causes seasons; why we see phases of the Moon and solar/lunar eclipses occur; how to distinguish between stars and planets based on their motion; and how our understanding of the universe has developed over time through scientific observation.
Making a loudspeaker

Sounds are produced by vibrations that travel through a medium such as air. The document discusses how to construct and test a loudspeaker. It instructs the reader to plug in the speaker, turn on the voltage, switch it on at the mains, and listen as they move their ear closer to identify how the sound changes and what causes this. The learning objectives are to describe how sounds are produced, construct a loudspeaker, and explain how a loudspeaker works.
Sound homework

Pupils conducted a sound experiment in a playground to investigate how walls cast sound shadows. They took sound level measurements at various points around the playground using a sound meter after making a standard sound. The measurements showed lower sound levels in positions located behind walls and structures relative to the sound source, demonstrating the shadowing effect of walls on sound propagation.
May the forces be with you

This document discusses forces, mass, and acceleration. It provides examples of calculating acceleration using the equation F=ma. It includes sample problems such as determining the force needed to accelerate a space shuttle or the acceleration of a cyclist pedaling with a given force. Practice problems are provided for students to calculate acceleration, force, or mass given two of the three variables.
Forces making a force meter

A force meter measures force in Newtons. It works by using a rubber band of known length that stretches when a force is applied. The amount the rubber band stretches corresponds to the amount of force applied, allowing the force to be measured in Newtons on a scale.
6.3 food glorious food

Food contains chemical energy that originally comes from the Sun. The amount of energy a person needs each day can vary depending on factors like their body size, age, gender, activity levels, and environmental conditions like temperature. While two people may consume the same amount of energy, one may still gain weight due to differences in their metabolism or physical activity levels.
Assignment 6.1

This document contains instructions and questions for Assignment 6.1. Students are instructed to show all working and include relevant units. The questions involve calculating quantities using given values and units, including multiplying and dividing measurements in m, m3, ml, and kg/m3. Conversions between standard and scientific notation are also required.
Assignment 6.2

This document contains a possible table of results that shows temperature readings in degrees Celsius taken at 11:00, 12:00, 13:00 and 14:00 hours each day from Monday to Friday. The temperatures generally increase throughout the day, with the highest readings occurring between 13:00 and 14:00 hours each afternoon.
6.4 bending light

The document discusses the refraction of light and how it causes optical illusions. It explains that when light travels from one material to another of different density, it changes direction. Specifically, light bends toward the normal when moving to a denser material, and away from the normal when moving to a less dense material. The document provides instructions for an experiment to observe and measure the refraction of light through a glass block at different angles of incidence.
6.4 bending light

Light changes direction when moving between different materials due to refraction. An experiment is described where a glass block is used to refract light rays entering at various angles, and the angles are measured and graphed. The graph shows the relationship between the incident and refracted angles, with the refracted angle increasing as the incident angle increases. This property of refraction is important for applications like lenses and understanding optical illusions.
Assignment 6.1 answers

This document contains 6 math questions requiring calculations with units. Question 1 involves multiplying two lengths. Question 2 gives a length. Question 3 involves (a) multiplying three lengths and (b) multiplying the results of part a. Question 4 involves (a) converting ml to m3 and (b) writing the answer in scientific notation. Question 5 involves (a) calculating length and volume and (b) calculating density. Question 6 involves calculating volume and density. Full working and units are required for all answers.
6.4 bending light

1. Light changes direction when moving between different materials due to refraction. Scientists studying refracting telescopes need accurate information on how light refracts when moving between air and glass.
2. An experiment is described where light passes through a semicircular glass block and the angle of refraction is measured for different angles of incidence.
3. The results are plotted on a graph showing the relationship between incident and refracted angles, helping to understand how optics can correct vision problems.
5.8 current and pd in a circuit

The document discusses a circuit with two components in series, a resistor and a buzzer, that have different resistances. It asks what the potential difference (p.d.) is across each component and what this reveals about their relative resistances. It then provides learning objectives and example calculations for current, potential difference, and resistance in series circuits.
Speed and velocity

The document discusses key concepts related to speed, velocity, distance, and time. It provides definitions of speed, distance, displacement, velocity, and average and instantaneous speed. Examples are given to illustrate the difference between distance and displacement. Graphs showing variations in distance and velocity over time are presented, and the relationships between distance, time, speed, velocity, and their equations are summarized in a table.
Document in windows internet explorer

The document describes an experiment to determine the laws of reflection. [1] Students are instructed to use a mirror, light source, and protractor to measure the angle of incidence and reflection of light rays. [2] They will shine light at a mirror from various angles and measure the corresponding reflected angles to see if the angle of reflection equals the angle of incidence. [3] By plotting their results on a graph, students can evaluate whether the evidence supports the statement that the angle of reflection equals the angle of incidence.
6.3 giant mirror

A village in Italy installed a large mirror on a mountain opposite their village to direct sunlight into the village. The mirror helped brighten the village by reflecting sunlight into the dark areas between the mountains. Villagers could now grow crops that previously did not receive enough sunlight. The mirror demonstrated how reflected light can help illuminate dark spaces.
6.2 energy everywhere

This document discusses different types of energy, including potential energy which is stored or hidden energy that has the ability to do work, and kinetic energy which is energy of motion. It lists learning objectives about forms of energy and energy transfers. The rest of the document appears to contain questions at different levels about energy, asking about heat energy, potential vs kinetic energy, energy transfers, examples of potential energy storage, and drawing energy transfer diagrams.
Mark scheme

This document is a coursework assessment form for a GCE Advanced Subsidiary physics course. It provides criteria for evaluating student coursework in 5 areas: [1] the quality and independence of the student's research briefing, [2] the use and understanding of physics demonstrated, and [3] the selection, summarizing, explanation, and [4] understanding/critical thinking shown in the student's work. Scores from 1-5 are given for each criterion, with 5 being the highest score. The form also includes the student's name and ID number for record keeping purposes.
More from mrrayner (20)
Recently uploaded
CACJapan - GROUP Presentation 1- Wk 4.pdf

Macroeconomics- Movie Location
This will be used as part of your Personal Professional Portfolio once graded.
Objective:
Prepare a presentation or a paper using research, basic comparative analysis, data organization and application of economic information. You will make an informed assessment of an economic climate outside of the United States to accomplish an entertainment industry objective.
Executive Directors Chat Leveraging AI for Diversity, Equity, and Inclusion

Let’s explore the intersection of technology and equity in the final session of our DEI series. Discover how AI tools, like ChatGPT, can be used to support and enhance your nonprofit's DEI initiatives. Participants will gain insights into practical AI applications and get tips for leveraging technology to advance their DEI goals.
PCOS corelations and management through Ayurveda.

This presentation includes basic of PCOS their pathology and treatment and also Ayurveda correlation of PCOS and Ayurvedic line of treatment mentioned in classics.
Azure Interview Questions and Answers PDF By ScholarHat

Azure Interview Questions and Answers PDF By ScholarHat
A Strategic Approach: GenAI in Education

Artificial Intelligence (AI) technologies such as Generative AI, Image Generators and Large Language Models have had a dramatic impact on teaching, learning and assessment over the past 18 months. The most immediate threat AI posed was to Academic Integrity with Higher Education Institutes (HEIs) focusing their efforts on combating the use of GenAI in assessment. Guidelines were developed for staff and students, policies put in place too. Innovative educators have forged paths in the use of Generative AI for teaching, learning and assessments leading to pockets of transformation springing up across HEIs, often with little or no top-down guidance, support or direction.
This Gasta posits a strategic approach to integrating AI into HEIs to prepare staff, students and the curriculum for an evolving world and workplace. We will highlight the advantages of working with these technologies beyond the realm of teaching, learning and assessment by considering prompt engineering skills, industry impact, curriculum changes, and the need for staff upskilling. In contrast, not engaging strategically with Generative AI poses risks, including falling behind peers, missed opportunities and failing to ensure our graduates remain employable. The rapid evolution of AI technologies necessitates a proactive and strategic approach if we are to remain relevant.
Main Java[All of the Base Concepts}.docx

This is part 1 of my Java Learning Journey. This Contains Custom methods, classes, constructors, packages, multithreading , try- catch block, finally block and more.
A Survey of Techniques for Maximizing LLM Performance.pptx

A Survey of Techniques for Maximizing LLM Performance
How to Fix the Import Error in the Odoo 17

An import error occurs when a program fails to import a module or library, disrupting its execution. In languages like Python, this issue arises when the specified module cannot be found or accessed, hindering the program's functionality. Resolving import errors is crucial for maintaining smooth software operation and uninterrupted development processes.
DRUGS AND ITS classification slide share

Any substance (other than food) that is used to prevent, diagnose, treat, or relieve symptoms of a
disease or abnormal condition
Natural birth techniques - Mrs.Akanksha Trivedi Rama University

Natural birth techniques - Mrs.Akanksha Trivedi Rama UniversityAkanksha trivedi rama nursing college kanpur.
Natural birth techniques are various type such as/ water birth , alexender method, hypnosis, bradley method, lamaze method etcHow to Manage Your Lost Opportunities in Odoo 17 CRM

Odoo 17 CRM allows us to track why we lose sales opportunities with "Lost Reasons." This helps analyze our sales process and identify areas for improvement. Here's how to configure lost reasons in Odoo 17 CRM
বাংলাদেশ অর্থনৈতিক সমীক্ষা (Economic Review) ২০২৪ UJS App.pdf

বাংলাদেশের অর্থনৈতিক সমীক্ষা ২০২৪ [Bangladesh Economic Review 2024 Bangla.pdf] কম্পিউটার , ট্যাব ও স্মার্ট ফোন ভার্সন সহ সম্পূর্ণ বাংলা ই-বুক বা pdf বই " সুচিপত্র ...বুকমার্ক মেনু 🔖 ও হাইপার লিংক মেনু 📝👆 যুক্ত ..
আমাদের সবার জন্য খুব খুব গুরুত্বপূর্ণ একটি বই ..বিসিএস, ব্যাংক, ইউনিভার্সিটি ভর্তি ও যে কোন প্রতিযোগিতা মূলক পরীক্ষার জন্য এর খুব ইম্পরট্যান্ট একটি বিষয় ...তাছাড়া বাংলাদেশের সাম্প্রতিক যে কোন ডাটা বা তথ্য এই বইতে পাবেন ...
তাই একজন নাগরিক হিসাবে এই তথ্য গুলো আপনার জানা প্রয়োজন ...।
বিসিএস ও ব্যাংক এর লিখিত পরীক্ষা ...+এছাড়া মাধ্যমিক ও উচ্চমাধ্যমিকের স্টুডেন্টদের জন্য অনেক কাজে আসবে ...
South African Journal of Science: Writing with integrity workshop (2024)

South African Journal of Science: Writing with integrity workshop (2024)Academy of Science of South Africa
A workshop hosted by the South African Journal of Science aimed at postgraduate students and early career researchers with little or no experience in writing and publishing journal articles.Recently uploaded (20)
Executive Directors Chat Leveraging AI for Diversity, Equity, and Inclusion

Executive Directors Chat Leveraging AI for Diversity, Equity, and Inclusion
Azure Interview Questions and Answers PDF By ScholarHat

Azure Interview Questions and Answers PDF By ScholarHat
A Survey of Techniques for Maximizing LLM Performance.pptx

A Survey of Techniques for Maximizing LLM Performance.pptx
Film vocab for eal 3 students: Australia the movie

Film vocab for eal 3 students: Australia the movie
Natural birth techniques - Mrs.Akanksha Trivedi Rama University

Natural birth techniques - Mrs.Akanksha Trivedi Rama University
How to Manage Your Lost Opportunities in Odoo 17 CRM

How to Manage Your Lost Opportunities in Odoo 17 CRM
বাংলাদেশ অর্থনৈতিক সমীক্ষা (Economic Review) ২০২৪ UJS App.pdf

বাংলাদেশ অর্থনৈতিক সমীক্ষা (Economic Review) ২০২৪ UJS App.pdf
South African Journal of Science: Writing with integrity workshop (2024)

South African Journal of Science: Writing with integrity workshop (2024)
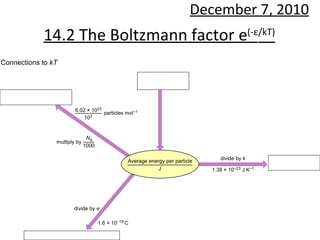
14.2 boltzmann factor
- 1. 14.2 The Boltzmann factor e (- ε /kT) December 7, 2010
- 4. Think about playing pool…
- 11. Flying high Dense Less dense Gravity Diffusion
- 13. fraction of molecules mgh The idea is generally applicable
- 18. rate of reaction is proportional to exp(-e/kT)
- 19. Soft matter
- 20. Soap, cell membranes and wristwatches
- 21. The molecules don’t care
- 22. Quick check questions (page 132)
- 23. Questions 1-7 (pg 134)
- 24. Test