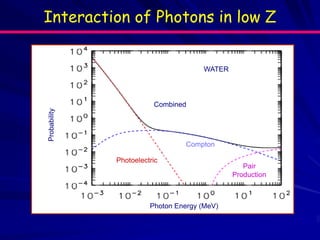
The document discusses various interactions of photons with matter. It describes three main interactions: the photoelectric effect, Compton scattering, and pair production. It provides details on the energy thresholds, probabilities, and products of these interactions. It also discusses attenuation of photons in materials and defines half value thickness and tenth value thickness. In addition, it summarizes interactions of charged particles and neutrons with matter, including ionization, bremsstrahlung, stopping power, linear energy transfer, and shielding considerations.