Oxonium and thionium ion as intermediate By Rezania.ppt
- 1. 1 Oxonium and Thionium ion as intermediate
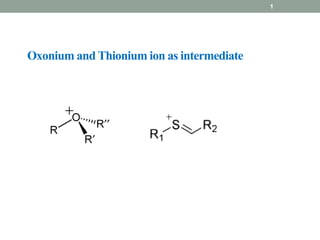
- 2. oxonium ion: In chemistry, the oxonium ion is any oxygen cation with three bonds. The simplest oxonium ion is the hydronium ion H3O+. Stable alkyloxonium salts exist; they are extensively used as alkylating agents. For example, triethyloxonium tetrafluoroborate (Et3O+)(BF4 −) is a white crystalline solid. 2Oxonium
- 3. In acidic media, the oxonium functional group produced by protonating an alcohol SN2: 3 The addition, follows Markovnikov’s rule Hydroxy-alkoxy-addition Oxonium +
- 4. Oxonium ions are excellent alkylating agents, and ethers can be conveniently prepared by treating them with alcohols or phenols Alkyl halides can be alkylated by ethers or ketones to give oxonium salts 4Oxonium
- 5. Oxonium in advance synthesis The term “carbenoid” refers to a metal-bound carbene, which results in a more stable species as compared to a “free” carbene. 5Oxonium
- 6. 6 Common Metal Carbenoid Reactions Insertion reactions Cyclopropanation Ylide generation Oxonium
- 7. 7 One of the earliest examples of oxonium ylide generation from catalytically generated metal carbenoids was provided by Nozaki et al. in 1966. Oxonium
- 8. 8 oxonium ylides reactions • [1,2]-shift reaction, • [2,3]-sigmatropic rearrangement, • insertion • β-hydride elimination. [1,2]-Shift Oxonium
- 9. 9 [1,2]-shift technique offers a new method for synthesising substituted carbocycles and cyclic ethers. Oxonium [1,2]-Shift
- 12. 12 Oxonium in the natural product (±)-vigulariol. Oxonium,[2,3]-Sigmatropic Rearrangement
- 16. 16 β-Hydride elimination Oxonium hydride elimination
- 17. 17 Thionium: Pummerer rearrangement: in fact Thionium is synonym for pummerer rearrangement
- 18. 18 Mechanism . Common activators besides acetic anhydride are trifluoroacetic anhydride and trifluoromethanesulfonic anhydride Common nucleophiles besides acetates are arenes, alkenes, amides, and phenols. Thionium:
- 19. 19 Even neutral nucleophiles can be used due to the highly electrophilic nature of the sulfonium. for example, the electron-rich aromatic ring of veratrole. Thionium:
- 20. 20Thionium:
- 21. 21 General Applications of the Pummerer Reaction 2.1 Pummerer Fragmentation Reactions Thionium:
- 22. 22 These results suggest that a pKR+ value greater than 14.5 is necessary for fragmentation to occur. only the Pummerer rearrangement A , B give fragmentation (4) 3C give a mixture Thionium:
- 23. 23 2.2. Vinylogous Pummerer Reactions Thionium:
- 24. 24 2.3. additive Pummerer Reactions Thionium:
- 25. 25 2.4. Interrupted Pummerer Reactions Successful reaction requires the absence of an amidic hydrogen; Thionium:
- 26. 26 Pummerer Reaction Using an Alternative Method of Thionium Ion Formation The Connective Pummerer Reaction Thionium:
- 28. 28 The Pummerer Reaction in Natural Product Synthesis Synthesis of Monomorine Thionium:
- 29. THANK YOY