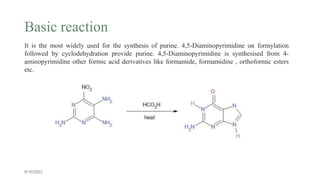
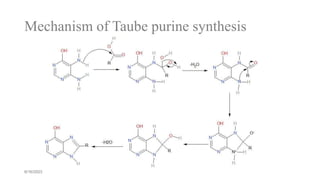
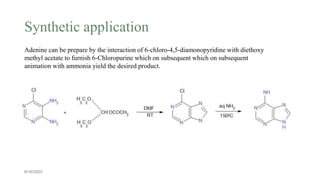
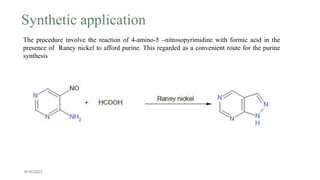
This document summarizes the Traube purine synthesis reaction. The Traube synthesis allows for the introduction of a carbon fragment to bridge the nitrogen atoms of the amino group at positions C-5 and C-6 of a pyrimidine ring. It involves the formylation of 4,5-diaminopyrimidine followed by cyclodehydration to produce a purine. 4,5-Diaminopyrimidine is synthesized from 4-aminopyrimidine using formic acid or its derivatives. The mechanism involves the initial formation of an iminium ion intermediate followed by ring closure. Adenine can be synthesized using this reaction by reacting 6-chloro-4,5-di