electronic properties of atoms 1.pptx
- 1. DEVELOPMENT OF THE ATOMIC THEORY WSU CHY15M0 - electronic properties of an atom
- 2. PURPOSE OF ATOMIC MODELS The models of the atom do not provide a complete understanding of the atom. The models help us to make useful predictions about how matter will behave. WSU CHY15M0 - electronic properties of an atom
- 3. Dalton Thomson Rutherford Bohr Schrodinger WSU CHY15M0 - electronic properties of an atom
- 4. JOHN DALTON (1766-1844), ❑Proposed the first atomic theory in 1808. WSU CHY15M0 - electronic properties of an atom
- 5. JOHN DALTON (1766-1844), ❑The atom is a solid sphere – billiard ball model. ⮚All matter consists of tiny particles called atoms, which cannot be broken up. ⮚All the atoms of a particular element are the same. Atoms of different elements are different. WSU CHY15M0 - electronic properties of an atom
- 6. JOHN DALTON (1766-1844), ❑The atom is a solid sphere – billiard ball model. ⮚When atoms combine, they form compounds. ⮚Atoms cannot be created or destroyed in a chemical reaction WSU CHY15M0 - electronic properties of an atom
- 7. Thomson WSU CHY15M0 - electronic properties of an atom
- 8. Thomson WSU CHY15M0 - electronic properties of an atom
- 9. JOSEPH J THOMSON ❑JJ Thomson discovered electrons in 1897. ❑The atom is a positive sphere where the electrons were randomly spread out inside the atom. ❑This model was known as the “plum- pudding” model. WSU CHY15M0 - electronic properties of an atom
- 10. Rutherford WSU CHY15M0 - electronic properties of an atom
- 11. Ruther- ford WSU CHY15M0 - electronic properties of an atom
- 12. ERNEST RUTHERFORD ❖In 1911 Ernest Rutherford used the discovery of an alpha particle to do further experiments on the atom. ❖The alpha(α) particle has the same mass as a helium atom but as a positive charge. ❖Rutherford directed a beam of alpha particles at a thin gold foil so that he could explore the inside of the atom. ❖The alpha particle scattering experiment. WSU CHY15M0 - electronic properties of an atom
- 13. ERNEST RUTHERFORD p38 ❖Almost all the mass of the atom is found in a very small positively charged nucleus in the centre of the atom. He later found the positive charge comes from protons. ❖The space around the nucleus is empty for the negatively charged electrons to be found there. ❖Rutherford’s nuclear atomic model. ❖One drawback: Opposite charges must attract each other and electrons should lose energy and spiral inward. WSU CHY15M0 - electronic properties of an atom
- 14. Bohr WSU CHY15M0 - electronic properties of an atom
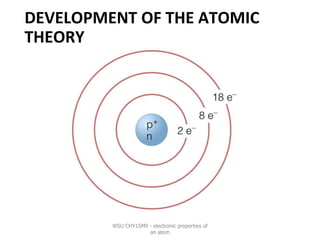
- 15. NIELS BOHR ❖Electrons move in orbits(or energy levels) around the nucleus. ❖Electrons with the same energy move around in the same orbit. ❖Electrons in orbits that are further away from the nucleus have a higher energy than the orbits closer to the nucleus. WSU CHY15M0 - electronic properties of an atom
- 16. MODERN ATOMIC MODEL ❖The quantum mechanical model or electron cloud model ❖An atom consists of a nucleus and electrons spinning around the nucleus. ❖The electrons move in a cloud around the nucleus. ❖We cannot tell exactly where the electrons are at any given time. ❖We do know that electrons exist more frequently in certain regions around the nucleus than others. WSU CHY15M0 - electronic properties of an atom
- 17. MODERN ATOMIC MODEL p39 WSU CHY15M0 - electronic properties of an atom
- 18. WSU CHY15M0 - electronic properties of an atom