electronic properties of atoms 1.pptx
•Download as PPTX, PDF•
0 likes•77 views
The document summarizes the development of atomic theory from Dalton's billiard ball model to the modern quantum mechanical model. John Dalton proposed the first atomic theory in 1808, modeling atoms as solid spheres. J.J. Thomson's plum pudding model viewed atoms as positive spheres with electrons spread throughout. Ernest Rutherford's gold foil experiment in 1911 revealed the atom's small, dense nucleus. Niels Bohr incorporated orbits for electrons around the nucleus. The modern atomic model describes electrons as existing in probabilistic electron clouds rather than definite orbits.
Report
Share
Report
Share
Recommended
Class 9 structure of atom

This document discusses the structure of the atom. It describes the discovery of subatomic particles like electrons, protons, and neutrons through experiments. This led to the failure of Dalton's atomic theory which stated that atoms were indivisible. It discusses J.J. Thomson's "plum pudding" model and Rutherford's gold foil experiment, which resulted in Rutherford's nuclear model of the atom with electrons orbiting a small, dense nucleus. Later, Bohr improved on this model by proposing that electrons can only orbit in fixed, quantized energy levels. The document also discusses concepts like atomic number, mass number, isotopes, and isobars.
Structure Of The Atom.pdf notes important

The document provides information on the structure of atoms, including key experiments and models that helped reveal the internal structure of atoms. It discusses J.J. Thomson's cathode ray experiment that discovered electrons, Rutherford's alpha particle scattering experiment that showed atoms have a small, dense nucleus, and Bohr's model of electron orbits around the nucleus. It also covers topics like isotopes, mass number, atomic number, electron configuration, and valency.
History of the atom. Atomic theory. hIstory of the atom

The document summarizes the history and development of atomic theory from ancient Greece to modern times. It discusses early concepts from Democritus and the Bible, then outlines key discoveries and models proposed by scientists like Dalton, Thomson, Rutherford, and Bohr. Their models introduced ideas like atoms being indivisible particles, atoms containing electrons and a nucleus, electrons orbiting in shells, and electrons absorbing and emitting energy in fixed amounts. Each new model built upon previous work but also had limitations that were addressed by subsequent scientists.
Atomic structure

The document discusses the history of the development of atomic structure models from Thomson's plum pudding model to Rutherford's nuclear model. Key events include J.J. Thomson's discovery of the electron, Millikan's oil drop experiment determining the charge of an electron, discovery of the proton through canal ray experiments, Rutherford's alpha particle scattering experiment revealing the dense nucleus at the center of the atom, and Rutherford proposing the nuclear model of the atom. The nuclear model represented a major breakthrough but did not fully explain electron stability.
Structure of the Atom.pptx.pdf

The document discusses the structure of atoms. It describes how early scientists like Dalton, Thomson, Rutherford, and Bohr contributed to developing models of the atom through experiments. Thomson's "plum pudding" model depicted electrons distributed uniformly in a positively charged sphere, but it could not explain atomic stability. Rutherford's gold foil experiment showed that the positive charge and mass of atoms are concentrated in a tiny nucleus, leading to his nuclear model. Bohr's model incorporated discrete electron orbits to explain atomic stability. The document also discusses subatomic particles like protons, neutrons, isotopes, isobars, and how electrons are arranged in shells according to their quantum numbers.
Atomic structure presentation

Atoms are composed of electrons, protons, and neutrons. Electrons orbit around a small, dense nucleus composed of protons and neutrons. Over time, scientists have developed models of the atom based on new discoveries. Democritus first proposed atoms could not be further divided in 450 BC. In the 19th and 20th centuries, physicists discovered subatomic particles within atoms like electrons, proving atoms could still be divided. Now atoms are understood to have a nucleus surrounded by electron clouds.
General

This document provides an overview of general chemistry concepts related to atomic structure. It discusses several atomic models proposed over time, including Dalton's atomic theory, Thomson's "plum pudding" model, Rutherford's nuclear model, and Bohr's model. It also describes experiments like Thomson's cathode ray experiment and Rutherford's alpha scattering experiment that helped develop understanding of atomic structure. Key topics covered include the discovery of subatomic particles like protons, neutrons, and electrons, isotopes, ionization, and atomic spectra.
Atomic Nucleus...

The atomic nucleus is at the center of atoms and is composed of protons and neutrons. It was discovered in 1911 by Ernest Rutherford based on experiments showing that atoms have small, dense, positively charged nuclei. The nucleus contains nearly all of an atom's mass. Protons and neutrons are bound together in the nucleus by the strong nuclear force. Nuclear chemistry deals with the composition, properties, and reactions of atomic nuclei. Key discoveries included the neutron by Chadwick in 1932 and the development of nuclear models showing electrons orbiting the positively charged nucleus.
Recommended
Class 9 structure of atom

This document discusses the structure of the atom. It describes the discovery of subatomic particles like electrons, protons, and neutrons through experiments. This led to the failure of Dalton's atomic theory which stated that atoms were indivisible. It discusses J.J. Thomson's "plum pudding" model and Rutherford's gold foil experiment, which resulted in Rutherford's nuclear model of the atom with electrons orbiting a small, dense nucleus. Later, Bohr improved on this model by proposing that electrons can only orbit in fixed, quantized energy levels. The document also discusses concepts like atomic number, mass number, isotopes, and isobars.
Structure Of The Atom.pdf notes important

The document provides information on the structure of atoms, including key experiments and models that helped reveal the internal structure of atoms. It discusses J.J. Thomson's cathode ray experiment that discovered electrons, Rutherford's alpha particle scattering experiment that showed atoms have a small, dense nucleus, and Bohr's model of electron orbits around the nucleus. It also covers topics like isotopes, mass number, atomic number, electron configuration, and valency.
History of the atom. Atomic theory. hIstory of the atom

The document summarizes the history and development of atomic theory from ancient Greece to modern times. It discusses early concepts from Democritus and the Bible, then outlines key discoveries and models proposed by scientists like Dalton, Thomson, Rutherford, and Bohr. Their models introduced ideas like atoms being indivisible particles, atoms containing electrons and a nucleus, electrons orbiting in shells, and electrons absorbing and emitting energy in fixed amounts. Each new model built upon previous work but also had limitations that were addressed by subsequent scientists.
Atomic structure

The document discusses the history of the development of atomic structure models from Thomson's plum pudding model to Rutherford's nuclear model. Key events include J.J. Thomson's discovery of the electron, Millikan's oil drop experiment determining the charge of an electron, discovery of the proton through canal ray experiments, Rutherford's alpha particle scattering experiment revealing the dense nucleus at the center of the atom, and Rutherford proposing the nuclear model of the atom. The nuclear model represented a major breakthrough but did not fully explain electron stability.
Structure of the Atom.pptx.pdf

The document discusses the structure of atoms. It describes how early scientists like Dalton, Thomson, Rutherford, and Bohr contributed to developing models of the atom through experiments. Thomson's "plum pudding" model depicted electrons distributed uniformly in a positively charged sphere, but it could not explain atomic stability. Rutherford's gold foil experiment showed that the positive charge and mass of atoms are concentrated in a tiny nucleus, leading to his nuclear model. Bohr's model incorporated discrete electron orbits to explain atomic stability. The document also discusses subatomic particles like protons, neutrons, isotopes, isobars, and how electrons are arranged in shells according to their quantum numbers.
Atomic structure presentation

Atoms are composed of electrons, protons, and neutrons. Electrons orbit around a small, dense nucleus composed of protons and neutrons. Over time, scientists have developed models of the atom based on new discoveries. Democritus first proposed atoms could not be further divided in 450 BC. In the 19th and 20th centuries, physicists discovered subatomic particles within atoms like electrons, proving atoms could still be divided. Now atoms are understood to have a nucleus surrounded by electron clouds.
General

This document provides an overview of general chemistry concepts related to atomic structure. It discusses several atomic models proposed over time, including Dalton's atomic theory, Thomson's "plum pudding" model, Rutherford's nuclear model, and Bohr's model. It also describes experiments like Thomson's cathode ray experiment and Rutherford's alpha scattering experiment that helped develop understanding of atomic structure. Key topics covered include the discovery of subatomic particles like protons, neutrons, and electrons, isotopes, ionization, and atomic spectra.
Atomic Nucleus...

The atomic nucleus is at the center of atoms and is composed of protons and neutrons. It was discovered in 1911 by Ernest Rutherford based on experiments showing that atoms have small, dense, positively charged nuclei. The nucleus contains nearly all of an atom's mass. Protons and neutrons are bound together in the nucleus by the strong nuclear force. Nuclear chemistry deals with the composition, properties, and reactions of atomic nuclei. Key discoveries included the neutron by Chadwick in 1932 and the development of nuclear models showing electrons orbiting the positively charged nucleus.
Atomic Model Timeline

1. The document outlines the development of atomic models from Dalton's atomic theory in 1803 to the quantum model in 1924. It describes key contributors including Dalton, Thomson, Millikan, Rutherford, Bohr, de Broglie, Schrodinger, and Born and their major discoveries.
2. Major milestones included Dalton establishing atoms as the basic unit of matter, Thomson discovering the electron, Rutherford discovering the nucleus, Bohr explaining electron energy levels, and the development of the quantum model explaining wave-particle duality of electrons.
3. Each new model built upon the previous work and corrected limitations, moving science closer to a full understanding of atomic structure.
Atoms Physics class 12th Nonmed

1) The document discusses Thomson's plum pudding model of the atom and its limitations. It then describes Rutherford's alpha scattering experiment, the conclusions he drew, and his nuclear model of the atom.
2) Bohr's model improved upon Rutherford's by proposing quantized electron orbits and energy levels based on Planck's quantum theory. This allowed Bohr to explain the hydrogen spectral series.
3) The document provides details of Thomson, Rutherford, and Bohr's atomic models, including their postulates and limitations. It discusses key experiments and conclusions that improved understanding of atomic structure over time.
Strcture of atom

The document discusses the development of atomic theory and models of atomic structure based on experiments. Key points include:
1. Early experiments with cathode ray tubes led Thomson to discover the electron and determine its small mass and negative charge.
2. Rutherford's gold foil experiment showed that the mass and positive charge of atoms are concentrated in a very small, dense nucleus.
3. Later experiments discovered the proton in the nucleus and the neutron, establishing the main subatomic particles that make up all elements.
4. Models evolved from Thomson's "plum pudding" model to Rutherford's nuclear model to better explain experimental results and the stability of atoms.
Basic Electronics 

Atoms are composed of protons, neutrons, and electrons. Protons and neutrons are located in the nucleus, while electrons orbit around the nucleus in different shells. An atom's atomic number refers to the number of protons, while its atomic mass includes the number of protons and neutrons. Ions are formed when atoms gain or lose electrons. Electronics involves the flow of electrons and concepts like voltage, current, resistance, and power. Circuits allow electricity to flow through a closed conductive loop. Materials are classified as conductors, insulators, or semiconductors based on how easily electrons can flow through them.
structure of an atom by romit jain

The document summarizes the development of atomic models over time. Early models included Thomson's "plum pudding" model which viewed the atom as electrons embedded in a uniform positive charge. Rutherford's gold foil experiment showed atoms have a small, dense nucleus and led to the nuclear model. Bohr incorporated quantized electron orbits to explain the stability of atoms. Later, Chadwick discovered the neutron, completing the standard model of the atom consisting of electrons orbiting a nucleus of protons and neutrons.
Electrons in atoms

The document discusses the historical development of atomic models from Dalton to Bohr and beyond. It introduces John Dalton's early model of atoms as indivisible particles with no internal structure in 1863. Later models incorporated the discoveries of the electron by J.J. Thomson in 1897 and the nuclear structure of atoms by Ernest Rutherford in 1911. Niels Bohr's 1913 model proposed that electrons orbit the nucleus in fixed, quantized energy levels. This laid the foundations for understanding atomic emission spectra and the quantum mechanical model that later replaced Bohr's model.
CBSE Class 9 Science Chapter 4- structure of atom

This document discusses the structure of atoms and the development of atomic models over time. It covers John Dalton's early atomic theory that atoms are indivisible and different for each element. J.J. Thomson's plum pudding model described atoms as electrically neutral spheres. Rutherford determined atoms have a small, dense nucleus through alpha particle scattering experiments. Niels Bohr incorporated quantum theory, proposing electrons orbit in discrete energy levels. The document also discusses atomic number, mass number, isotopes, electron configuration, and valency.
Radiography introduction

Radiography uses x-rays to generate images of the internal structures of objects. X-rays are generated using an x-ray tube, which accelerates electrons toward a metal target. When the electrons collide with the target, x-rays are produced. These x-rays are used to expose radiographic film, creating a latent image. The film is then developed using chemical processes similar to photographic film development, making the latent image visible. The visible image reveals the internal structures and densities of the object in a manner similar to shadows.
Atomic structure Grade 11 Chemistry

This presentation is specially made for the students of grades 11 and 12 of High School. This is the presentation of chapter Atomic Structure with proper diagrams, figures, facts, mnemonics, and some repeated past questions. Here you will get a chance to know about Atomic theory, Daltons Law, particles and so on.
Structure of atom

This document discusses the historical development of atomic models from Thomson's plum pudding model to Bohr's model of electron shells. It describes key experiments and findings, including:
1) Rutherford's gold foil experiment which showed that the positive charge of atoms is concentrated in a small, dense nucleus.
2) Bohr's model which explained atomic stability by proposing discrete electron orbits where electrons do not radiate energy.
3) The discovery of the neutron by Chadwick in 1932, completing understanding of atomic structure with protons and neutrons in the nucleus and electrons in shells surrounding it.
Nikhil duggal

The document discusses the atomic structure and models of the atom. It begins with an acknowledgement and table of contents. It then covers Dalton's atomic theory, subatomic particles like electrons and protons, cathode rays and the discovery of electrons. It discusses the charge to mass ratio of electrons, the discovery of protons and neutrons, and models of the atom including Thomson's model and Rutherford's nuclear model. It also addresses isotopes, limitations of models, wave nature of radiation, and the electromagnetic spectrum.
Stricture of Atoms(Class 11)(Chapter 2) 

Structure of Atoms some basic concepts of atomic structure its history of modelling and also the present and accepted model including the quantum model of atomic structure.
Atomic Theory Reg

1. Early atomic models proposed by philosophers like Democritus and Dalton proposed that atoms were the fundamental indivisible units of matter.
2. Rutherford's gold foil experiment in the early 1900s showed that the atom has a small, dense nucleus at its center containing positive charge.
3. Later models like Bohr's incorporated the idea that electrons orbit the nucleus in fixed energy levels, accounting for the emission and absorption of photons.
A T O M I C T H E O R Y Reg

1. Early atomic models proposed by philosophers like Democritus and Dalton proposed that atoms were the fundamental building blocks of matter.
2. Rutherford's gold foil experiment in the early 1900s showed that the atom has a small, dense nucleus at its center containing most of its mass.
3. Later discoveries found that the nucleus contains protons and neutrons, while electrons orbit the nucleus, leading to the modern understanding of atomic structure.
ELECTRON-theory ppt industrials arts part2

Electron theory aims to explain the structure and properties of matter through its electronic structure. It states that all matter is comprised of molecules made up of atoms, which contain protons, neutrons, and electrons. Electrons play a key role in electricity as their movement constitutes electric current. Within atoms, electrons exist in specific energy levels or shells around the nucleus, and their behavior is described by quantum mechanics.
Dt

Rutherford conducted a gold foil experiment where he observed that some alpha particles passed through the gold foil, some were slightly deflected, and some rebounded backwards. From this, he inferred that atoms have a small, dense, positively charged nucleus surrounded by electrons. This Rutherford model of the atom likened the atom to the solar system, with electrons orbiting the nucleus similarly to planets orbiting the sun. Later, Bohr proposed modifications where electrons can only orbit in certain distances corresponding to specific energy levels, explaining the stability of atoms.
Dt

Rutherford conducted a gold foil experiment where he bombarded a thin gold foil with alpha particles. He discovered that some alpha particles bounced back, indicating there was a small, dense nucleus at the center of the atom. This led Rutherford to propose an atomic model where a small, positively charged nucleus is surrounded by electrons. Later, Bohr added to this model by suggesting electrons can only orbit the nucleus in certain fixed energy levels. The Rutherford-Bohr model helped explain the stability of atoms and how electrons can move between energy levels by absorbing or emitting photons.
Dt

Rutherford conducted a gold foil experiment where he bombarded a thin gold foil with alpha particles. He discovered that some alpha particles bounced back, indicating there was a small, dense nucleus at the center of the atom. This led Rutherford to propose an atomic model where a small, positively charged nucleus is surrounded by electrons. Later, Bohr added to this model by suggesting electrons can only orbit the nucleus in certain fixed energy levels. The Rutherford-Bohr model helped explain the stability of atoms and how electrons can move between energy levels by absorbing or emitting photons.
Dt

Rutherford conducted a gold foil experiment where he observed that some alpha particles passed through the gold foil, some were slightly deflected, and some rebounded backwards. From this, he inferred that atoms have a small, dense, positively charged nucleus surrounded by electrons. This Rutherford model of the atom likened the atom to the solar system, with electrons orbiting the nucleus similarly to planets orbiting the sun. Later, Bohr proposed modifications where electrons can only orbit in certain distances corresponding to specific energy levels, explaining the stability of atoms.
Community pharmacy- Social and preventive pharmacy UNIT 5

Covered community pharmacy topic of the subject Social and preventive pharmacy for Diploma and Bachelor of pharmacy
Digital Artifact 1 - 10VCD Environments Unit

Digital Artifact 1 - 10VCD Environments Unit - NGV Pavilion Concept Design
More Related Content
Similar to electronic properties of atoms 1.pptx
Atomic Model Timeline

1. The document outlines the development of atomic models from Dalton's atomic theory in 1803 to the quantum model in 1924. It describes key contributors including Dalton, Thomson, Millikan, Rutherford, Bohr, de Broglie, Schrodinger, and Born and their major discoveries.
2. Major milestones included Dalton establishing atoms as the basic unit of matter, Thomson discovering the electron, Rutherford discovering the nucleus, Bohr explaining electron energy levels, and the development of the quantum model explaining wave-particle duality of electrons.
3. Each new model built upon the previous work and corrected limitations, moving science closer to a full understanding of atomic structure.
Atoms Physics class 12th Nonmed

1) The document discusses Thomson's plum pudding model of the atom and its limitations. It then describes Rutherford's alpha scattering experiment, the conclusions he drew, and his nuclear model of the atom.
2) Bohr's model improved upon Rutherford's by proposing quantized electron orbits and energy levels based on Planck's quantum theory. This allowed Bohr to explain the hydrogen spectral series.
3) The document provides details of Thomson, Rutherford, and Bohr's atomic models, including their postulates and limitations. It discusses key experiments and conclusions that improved understanding of atomic structure over time.
Strcture of atom

The document discusses the development of atomic theory and models of atomic structure based on experiments. Key points include:
1. Early experiments with cathode ray tubes led Thomson to discover the electron and determine its small mass and negative charge.
2. Rutherford's gold foil experiment showed that the mass and positive charge of atoms are concentrated in a very small, dense nucleus.
3. Later experiments discovered the proton in the nucleus and the neutron, establishing the main subatomic particles that make up all elements.
4. Models evolved from Thomson's "plum pudding" model to Rutherford's nuclear model to better explain experimental results and the stability of atoms.
Basic Electronics 

Atoms are composed of protons, neutrons, and electrons. Protons and neutrons are located in the nucleus, while electrons orbit around the nucleus in different shells. An atom's atomic number refers to the number of protons, while its atomic mass includes the number of protons and neutrons. Ions are formed when atoms gain or lose electrons. Electronics involves the flow of electrons and concepts like voltage, current, resistance, and power. Circuits allow electricity to flow through a closed conductive loop. Materials are classified as conductors, insulators, or semiconductors based on how easily electrons can flow through them.
structure of an atom by romit jain

The document summarizes the development of atomic models over time. Early models included Thomson's "plum pudding" model which viewed the atom as electrons embedded in a uniform positive charge. Rutherford's gold foil experiment showed atoms have a small, dense nucleus and led to the nuclear model. Bohr incorporated quantized electron orbits to explain the stability of atoms. Later, Chadwick discovered the neutron, completing the standard model of the atom consisting of electrons orbiting a nucleus of protons and neutrons.
Electrons in atoms

The document discusses the historical development of atomic models from Dalton to Bohr and beyond. It introduces John Dalton's early model of atoms as indivisible particles with no internal structure in 1863. Later models incorporated the discoveries of the electron by J.J. Thomson in 1897 and the nuclear structure of atoms by Ernest Rutherford in 1911. Niels Bohr's 1913 model proposed that electrons orbit the nucleus in fixed, quantized energy levels. This laid the foundations for understanding atomic emission spectra and the quantum mechanical model that later replaced Bohr's model.
CBSE Class 9 Science Chapter 4- structure of atom

This document discusses the structure of atoms and the development of atomic models over time. It covers John Dalton's early atomic theory that atoms are indivisible and different for each element. J.J. Thomson's plum pudding model described atoms as electrically neutral spheres. Rutherford determined atoms have a small, dense nucleus through alpha particle scattering experiments. Niels Bohr incorporated quantum theory, proposing electrons orbit in discrete energy levels. The document also discusses atomic number, mass number, isotopes, electron configuration, and valency.
Radiography introduction

Radiography uses x-rays to generate images of the internal structures of objects. X-rays are generated using an x-ray tube, which accelerates electrons toward a metal target. When the electrons collide with the target, x-rays are produced. These x-rays are used to expose radiographic film, creating a latent image. The film is then developed using chemical processes similar to photographic film development, making the latent image visible. The visible image reveals the internal structures and densities of the object in a manner similar to shadows.
Atomic structure Grade 11 Chemistry

This presentation is specially made for the students of grades 11 and 12 of High School. This is the presentation of chapter Atomic Structure with proper diagrams, figures, facts, mnemonics, and some repeated past questions. Here you will get a chance to know about Atomic theory, Daltons Law, particles and so on.
Structure of atom

This document discusses the historical development of atomic models from Thomson's plum pudding model to Bohr's model of electron shells. It describes key experiments and findings, including:
1) Rutherford's gold foil experiment which showed that the positive charge of atoms is concentrated in a small, dense nucleus.
2) Bohr's model which explained atomic stability by proposing discrete electron orbits where electrons do not radiate energy.
3) The discovery of the neutron by Chadwick in 1932, completing understanding of atomic structure with protons and neutrons in the nucleus and electrons in shells surrounding it.
Nikhil duggal

The document discusses the atomic structure and models of the atom. It begins with an acknowledgement and table of contents. It then covers Dalton's atomic theory, subatomic particles like electrons and protons, cathode rays and the discovery of electrons. It discusses the charge to mass ratio of electrons, the discovery of protons and neutrons, and models of the atom including Thomson's model and Rutherford's nuclear model. It also addresses isotopes, limitations of models, wave nature of radiation, and the electromagnetic spectrum.
Stricture of Atoms(Class 11)(Chapter 2) 

Structure of Atoms some basic concepts of atomic structure its history of modelling and also the present and accepted model including the quantum model of atomic structure.
Atomic Theory Reg

1. Early atomic models proposed by philosophers like Democritus and Dalton proposed that atoms were the fundamental indivisible units of matter.
2. Rutherford's gold foil experiment in the early 1900s showed that the atom has a small, dense nucleus at its center containing positive charge.
3. Later models like Bohr's incorporated the idea that electrons orbit the nucleus in fixed energy levels, accounting for the emission and absorption of photons.
A T O M I C T H E O R Y Reg

1. Early atomic models proposed by philosophers like Democritus and Dalton proposed that atoms were the fundamental building blocks of matter.
2. Rutherford's gold foil experiment in the early 1900s showed that the atom has a small, dense nucleus at its center containing most of its mass.
3. Later discoveries found that the nucleus contains protons and neutrons, while electrons orbit the nucleus, leading to the modern understanding of atomic structure.
ELECTRON-theory ppt industrials arts part2

Electron theory aims to explain the structure and properties of matter through its electronic structure. It states that all matter is comprised of molecules made up of atoms, which contain protons, neutrons, and electrons. Electrons play a key role in electricity as their movement constitutes electric current. Within atoms, electrons exist in specific energy levels or shells around the nucleus, and their behavior is described by quantum mechanics.
Dt

Rutherford conducted a gold foil experiment where he observed that some alpha particles passed through the gold foil, some were slightly deflected, and some rebounded backwards. From this, he inferred that atoms have a small, dense, positively charged nucleus surrounded by electrons. This Rutherford model of the atom likened the atom to the solar system, with electrons orbiting the nucleus similarly to planets orbiting the sun. Later, Bohr proposed modifications where electrons can only orbit in certain distances corresponding to specific energy levels, explaining the stability of atoms.
Dt

Rutherford conducted a gold foil experiment where he bombarded a thin gold foil with alpha particles. He discovered that some alpha particles bounced back, indicating there was a small, dense nucleus at the center of the atom. This led Rutherford to propose an atomic model where a small, positively charged nucleus is surrounded by electrons. Later, Bohr added to this model by suggesting electrons can only orbit the nucleus in certain fixed energy levels. The Rutherford-Bohr model helped explain the stability of atoms and how electrons can move between energy levels by absorbing or emitting photons.
Dt

Rutherford conducted a gold foil experiment where he bombarded a thin gold foil with alpha particles. He discovered that some alpha particles bounced back, indicating there was a small, dense nucleus at the center of the atom. This led Rutherford to propose an atomic model where a small, positively charged nucleus is surrounded by electrons. Later, Bohr added to this model by suggesting electrons can only orbit the nucleus in certain fixed energy levels. The Rutherford-Bohr model helped explain the stability of atoms and how electrons can move between energy levels by absorbing or emitting photons.
Dt

Rutherford conducted a gold foil experiment where he observed that some alpha particles passed through the gold foil, some were slightly deflected, and some rebounded backwards. From this, he inferred that atoms have a small, dense, positively charged nucleus surrounded by electrons. This Rutherford model of the atom likened the atom to the solar system, with electrons orbiting the nucleus similarly to planets orbiting the sun. Later, Bohr proposed modifications where electrons can only orbit in certain distances corresponding to specific energy levels, explaining the stability of atoms.
Similar to electronic properties of atoms 1.pptx (20)
Recently uploaded
Community pharmacy- Social and preventive pharmacy UNIT 5

Covered community pharmacy topic of the subject Social and preventive pharmacy for Diploma and Bachelor of pharmacy
Digital Artifact 1 - 10VCD Environments Unit

Digital Artifact 1 - 10VCD Environments Unit - NGV Pavilion Concept Design
RPMS TEMPLATE FOR SCHOOL YEAR 2023-2024 FOR TEACHER 1 TO TEACHER 3

RPMS Template 2023-2024 by: Irene S. Rueco
How to Fix the Import Error in the Odoo 17

An import error occurs when a program fails to import a module or library, disrupting its execution. In languages like Python, this issue arises when the specified module cannot be found or accessed, hindering the program's functionality. Resolving import errors is crucial for maintaining smooth software operation and uninterrupted development processes.
The Diamonds of 2023-2024 in the IGRA collection

A review of the growth of the Israel Genealogy Research Association Database Collection for the last 12 months. Our collection is now passed the 3 million mark and still growing. See which archives have contributed the most. See the different types of records we have, and which years have had records added. You can also see what we have for the future.
How to Make a Field Mandatory in Odoo 17

In Odoo, making a field required can be done through both Python code and XML views. When you set the required attribute to True in Python code, it makes the field required across all views where it's used. Conversely, when you set the required attribute in XML views, it makes the field required only in the context of that particular view.
The History of Stoke Newington Street Names

Presented at the Stoke Newington Literary Festival on 9th June 2024
www.StokeNewingtonHistory.com
Advanced Java[Extra Concepts, Not Difficult].docx![Advanced Java[Extra Concepts, Not Difficult].docx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Advanced Java[Extra Concepts, Not Difficult].docx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
This is part 2 of my Java Learning Journey. This contains Hashing, ArrayList, LinkedList, Date and Time Classes, Calendar Class and more.
Main Java[All of the Base Concepts}.docx

This is part 1 of my Java Learning Journey. This Contains Custom methods, classes, constructors, packages, multithreading , try- catch block, finally block and more.
LAND USE LAND COVER AND NDVI OF MIRZAPUR DISTRICT, UP

This Dissertation explores the particular circumstances of Mirzapur, a region located in the
core of India. Mirzapur, with its varied terrains and abundant biodiversity, offers an optimal
environment for investigating the changes in vegetation cover dynamics. Our study utilizes
advanced technologies such as GIS (Geographic Information Systems) and Remote sensing to
analyze the transformations that have taken place over the course of a decade.
The complex relationship between human activities and the environment has been the focus
of extensive research and worry. As the global community grapples with swift urbanization,
population expansion, and economic progress, the effects on natural ecosystems are becoming
more evident. A crucial element of this impact is the alteration of vegetation cover, which plays a
significant role in maintaining the ecological equilibrium of our planet.Land serves as the foundation for all human activities and provides the necessary materials for
these activities. As the most crucial natural resource, its utilization by humans results in different
'Land uses,' which are determined by both human activities and the physical characteristics of the
land.
The utilization of land is impacted by human needs and environmental factors. In countries
like India, rapid population growth and the emphasis on extensive resource exploitation can lead
to significant land degradation, adversely affecting the region's land cover.
Therefore, human intervention has significantly influenced land use patterns over many
centuries, evolving its structure over time and space. In the present era, these changes have
accelerated due to factors such as agriculture and urbanization. Information regarding land use and
cover is essential for various planning and management tasks related to the Earth's surface,
providing crucial environmental data for scientific, resource management, policy purposes, and
diverse human activities.
Accurate understanding of land use and cover is imperative for the development planning
of any area. Consequently, a wide range of professionals, including earth system scientists, land
and water managers, and urban planners, are interested in obtaining data on land use and cover
changes, conversion trends, and other related patterns. The spatial dimensions of land use and
cover support policymakers and scientists in making well-informed decisions, as alterations in
these patterns indicate shifts in economic and social conditions. Monitoring such changes with the
help of Advanced technologies like Remote Sensing and Geographic Information Systems is
crucial for coordinated efforts across different administrative levels. Advanced technologies like
Remote Sensing and Geographic Information Systems
9
Changes in vegetation cover refer to variations in the distribution, composition, and overall
structure of plant communities across different temporal and spatial scales. These changes can
occur natural.
Pollock and Snow "DEIA in the Scholarly Landscape, Session One: Setting Expec...

Pollock and Snow "DEIA in the Scholarly Landscape, Session One: Setting Expec...National Information Standards Organization (NISO)
This presentation was provided by Steph Pollock of The American Psychological Association’s Journals Program, and Damita Snow, of The American Society of Civil Engineers (ASCE), for the initial session of NISO's 2024 Training Series "DEIA in the Scholarly Landscape." Session One: 'Setting Expectations: a DEIA Primer,' was held June 6, 2024.Pengantar Penggunaan Flutter - Dart programming language1.pptx

Pengantar Penggunaan Flutter - Dart programming language1.pptx
ANATOMY AND BIOMECHANICS OF HIP JOINT.pdf

it describes the bony anatomy including the femoral head , acetabulum, labrum . also discusses the capsule , ligaments . muscle that act on the hip joint and the range of motion are outlined. factors affecting hip joint stability and weight transmission through the joint are summarized.
How to Manage Your Lost Opportunities in Odoo 17 CRM

Odoo 17 CRM allows us to track why we lose sales opportunities with "Lost Reasons." This helps analyze our sales process and identify areas for improvement. Here's how to configure lost reasons in Odoo 17 CRM
Walmart Business+ and Spark Good for Nonprofits.pdf

"Learn about all the ways Walmart supports nonprofit organizations.
You will hear from Liz Willett, the Head of Nonprofits, and hear about what Walmart is doing to help nonprofits, including Walmart Business and Spark Good. Walmart Business+ is a new offer for nonprofits that offers discounts and also streamlines nonprofits order and expense tracking, saving time and money.
The webinar may also give some examples on how nonprofits can best leverage Walmart Business+.
The event will cover the following::
Walmart Business + (https://business.walmart.com/plus) is a new shopping experience for nonprofits, schools, and local business customers that connects an exclusive online shopping experience to stores. Benefits include free delivery and shipping, a 'Spend Analytics” feature, special discounts, deals and tax-exempt shopping.
Special TechSoup offer for a free 180 days membership, and up to $150 in discounts on eligible orders.
Spark Good (walmart.com/sparkgood) is a charitable platform that enables nonprofits to receive donations directly from customers and associates.
Answers about how you can do more with Walmart!"
Recently uploaded (20)
Community pharmacy- Social and preventive pharmacy UNIT 5

Community pharmacy- Social and preventive pharmacy UNIT 5
RPMS TEMPLATE FOR SCHOOL YEAR 2023-2024 FOR TEACHER 1 TO TEACHER 3

RPMS TEMPLATE FOR SCHOOL YEAR 2023-2024 FOR TEACHER 1 TO TEACHER 3
Liberal Approach to the Study of Indian Politics.pdf

Liberal Approach to the Study of Indian Politics.pdf
Pride Month Slides 2024 David Douglas School District

Pride Month Slides 2024 David Douglas School District
LAND USE LAND COVER AND NDVI OF MIRZAPUR DISTRICT, UP

LAND USE LAND COVER AND NDVI OF MIRZAPUR DISTRICT, UP
Pollock and Snow "DEIA in the Scholarly Landscape, Session One: Setting Expec...

Pollock and Snow "DEIA in the Scholarly Landscape, Session One: Setting Expec...
Pengantar Penggunaan Flutter - Dart programming language1.pptx

Pengantar Penggunaan Flutter - Dart programming language1.pptx
How to Manage Your Lost Opportunities in Odoo 17 CRM

How to Manage Your Lost Opportunities in Odoo 17 CRM
Walmart Business+ and Spark Good for Nonprofits.pdf

Walmart Business+ and Spark Good for Nonprofits.pdf
electronic properties of atoms 1.pptx
- 1. DEVELOPMENT OF THE ATOMIC THEORY WSU CHY15M0 - electronic properties of an atom
- 2. PURPOSE OF ATOMIC MODELS The models of the atom do not provide a complete understanding of the atom. The models help us to make useful predictions about how matter will behave. WSU CHY15M0 - electronic properties of an atom
- 3. Dalton Thomson Rutherford Bohr Schrodinger WSU CHY15M0 - electronic properties of an atom
- 4. JOHN DALTON (1766-1844), ❑Proposed the first atomic theory in 1808. WSU CHY15M0 - electronic properties of an atom
- 5. JOHN DALTON (1766-1844), ❑The atom is a solid sphere – billiard ball model. ⮚All matter consists of tiny particles called atoms, which cannot be broken up. ⮚All the atoms of a particular element are the same. Atoms of different elements are different. WSU CHY15M0 - electronic properties of an atom
- 6. JOHN DALTON (1766-1844), ❑The atom is a solid sphere – billiard ball model. ⮚When atoms combine, they form compounds. ⮚Atoms cannot be created or destroyed in a chemical reaction WSU CHY15M0 - electronic properties of an atom
- 7. Thomson WSU CHY15M0 - electronic properties of an atom
- 8. Thomson WSU CHY15M0 - electronic properties of an atom
- 9. JOSEPH J THOMSON ❑JJ Thomson discovered electrons in 1897. ❑The atom is a positive sphere where the electrons were randomly spread out inside the atom. ❑This model was known as the “plum- pudding” model. WSU CHY15M0 - electronic properties of an atom
- 10. Rutherford WSU CHY15M0 - electronic properties of an atom
- 11. Ruther- ford WSU CHY15M0 - electronic properties of an atom
- 12. ERNEST RUTHERFORD ❖In 1911 Ernest Rutherford used the discovery of an alpha particle to do further experiments on the atom. ❖The alpha(α) particle has the same mass as a helium atom but as a positive charge. ❖Rutherford directed a beam of alpha particles at a thin gold foil so that he could explore the inside of the atom. ❖The alpha particle scattering experiment. WSU CHY15M0 - electronic properties of an atom
- 13. ERNEST RUTHERFORD p38 ❖Almost all the mass of the atom is found in a very small positively charged nucleus in the centre of the atom. He later found the positive charge comes from protons. ❖The space around the nucleus is empty for the negatively charged electrons to be found there. ❖Rutherford’s nuclear atomic model. ❖One drawback: Opposite charges must attract each other and electrons should lose energy and spiral inward. WSU CHY15M0 - electronic properties of an atom
- 14. Bohr WSU CHY15M0 - electronic properties of an atom
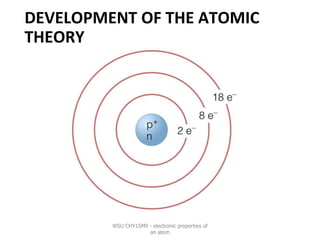
- 15. NIELS BOHR ❖Electrons move in orbits(or energy levels) around the nucleus. ❖Electrons with the same energy move around in the same orbit. ❖Electrons in orbits that are further away from the nucleus have a higher energy than the orbits closer to the nucleus. WSU CHY15M0 - electronic properties of an atom
- 16. MODERN ATOMIC MODEL ❖The quantum mechanical model or electron cloud model ❖An atom consists of a nucleus and electrons spinning around the nucleus. ❖The electrons move in a cloud around the nucleus. ❖We cannot tell exactly where the electrons are at any given time. ❖We do know that electrons exist more frequently in certain regions around the nucleus than others. WSU CHY15M0 - electronic properties of an atom
- 17. MODERN ATOMIC MODEL p39 WSU CHY15M0 - electronic properties of an atom
- 18. WSU CHY15M0 - electronic properties of an atom