structure of atom lesson2
•Download as DOCX, PDF•
1 like•191 views
structure of atom
Report
Share
Report
Share
Recommended
More Related Content
Similar to structure of atom lesson2
Similar to structure of atom lesson2 (20)
B.sc(microbiology and biotechnology and biochemistry) ii inorganic chemistry ...

B.sc(microbiology and biotechnology and biochemistry) ii inorganic chemistry ...
chapter_06pptfromBrownLeMayBurstendownloadedandmodified.ppt

chapter_06pptfromBrownLeMayBurstendownloadedandmodified.ppt
chapter_06pptfromBrownLeMayBurstendownloadedandmodified.ppt

chapter_06pptfromBrownLeMayBurstendownloadedandmodified.ppt
chapter_06pptfromBrownLeMayBurstendownloadedandmodified.ppt

chapter_06pptfromBrownLeMayBurstendownloadedandmodified.ppt
Study on the Energy Level Splitting of the Francium Atom Fr in an External Ma...

Study on the Energy Level Splitting of the Francium Atom Fr in an External Ma...
More from MISSRITIMABIOLOGYEXP
More from MISSRITIMABIOLOGYEXP (20)
PART 1 - CHAPTER 1 - CELL THE FUNDAMENTAL UNIT OF LIFE

PART 1 - CHAPTER 1 - CELL THE FUNDAMENTAL UNIT OF LIFE
CHAPTER 1 CROP PRODUCTION AND MANAGEMENT CLASS 8TH

CHAPTER 1 CROP PRODUCTION AND MANAGEMENT CLASS 8TH
HOW TO OBTAIN PURE COPPER SULPHATE FROM AN IMPURE SAMPLE

HOW TO OBTAIN PURE COPPER SULPHATE FROM AN IMPURE SAMPLE
HOW CAN WE OBTAIN COLOURED COMPONENT FROM BLUE OR BLACK INK?

HOW CAN WE OBTAIN COLOURED COMPONENT FROM BLUE OR BLACK INK?
Recently uploaded
Recently uploaded (20)
Call Girls in Mayapuri Delhi 💯Call Us 🔝9953322196🔝 💯Escort.

Call Girls in Mayapuri Delhi 💯Call Us 🔝9953322196🔝 💯Escort.
CALL ON ➥8923113531 🔝Call Girls Kesar Bagh Lucknow best Night Fun service 🪡

CALL ON ➥8923113531 🔝Call Girls Kesar Bagh Lucknow best Night Fun service 🪡
Artificial Intelligence In Microbiology by Dr. Prince C P

Artificial Intelligence In Microbiology by Dr. Prince C P
Scheme-of-Work-Science-Stage-4 cambridge science.docx

Scheme-of-Work-Science-Stage-4 cambridge science.docx
Neurodevelopmental disorders according to the dsm 5 tr

Neurodevelopmental disorders according to the dsm 5 tr
Engler and Prantl system of classification in plant taxonomy

Engler and Prantl system of classification in plant taxonomy
Recombinant DNA technology( Transgenic plant and animal)

Recombinant DNA technology( Transgenic plant and animal)
Recombination DNA Technology (Nucleic Acid Hybridization )

Recombination DNA Technology (Nucleic Acid Hybridization )
Grafana in space: Monitoring Japan's SLIM moon lander in real time

Grafana in space: Monitoring Japan's SLIM moon lander in real time
Analytical Profile of Coleus Forskohlii | Forskolin .pptx

Analytical Profile of Coleus Forskohlii | Forskolin .pptx
Traditional Agroforestry System in India- Shifting Cultivation, Taungya, Home...

Traditional Agroforestry System in India- Shifting Cultivation, Taungya, Home...
Discovery of an Accretion Streamer and a Slow Wide-angle Outflow around FUOri...

Discovery of an Accretion Streamer and a Slow Wide-angle Outflow around FUOri...
Analytical Profile of Coleus Forskohlii | Forskolin .pdf

Analytical Profile of Coleus Forskohlii | Forskolin .pdf
structure of atom lesson2
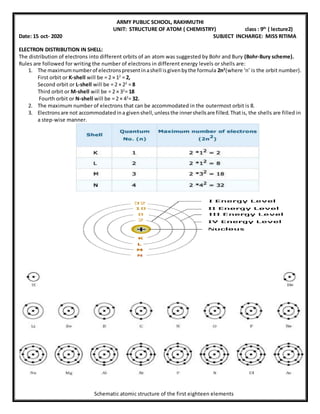
- 1. ARMY PUBLIC SCHOOL, RAKHMUTHI UNIT: STRUCTURE OF ATOM ( CHEMISTRY) class : 9th ( lecture2) Date: 15 oct- 2020 SUBJECT INCHARGE: MISS RITIMA ELECTRON DISTRIBUTION IN SHELL: The distribution of electrons into different orbits of an atom was suggested by Bohr and Bury (Bohr-Bury scheme). Rules are followed for writing the number of electrons in different energy levels or shells are: 1. The maximumnumberof electronspresentinashell isgivenbythe formula 2n2 (where ‘n’ is the orbit number). First orbit or K-shell will be = 2 × 12 = 2, Second orbit or L-shell will be = 2 × 22 = 8 Third orbit or M-shell will be = 2 × 32 = 18 Fourth orbit or N-shell will be = 2 × 42 = 32. 2. The maximum number of electrons that can be accommodated in the outermost orbit is 8. 3. Electronsare not accommodatedina givenshell,unlessthe innershellsare filled.Thatis, the shells are filled in a step-wise manner. Schematic atomic structure of the first eighteen elements
