Report
Share
Download to read offline
Recommended
More Related Content
Similar to MINDMAP PHENOL.pdf
Similar to MINDMAP PHENOL.pdf (20)
Heterocyclic compounds unit III as per PCI syllabus

Heterocyclic compounds unit III as per PCI syllabus
B. Sc. Part - I (Sem-II) Unit-IV (A) Phenols by Dr Pramod R Padole

B. Sc. Part - I (Sem-II) Unit-IV (A) Phenols by Dr Pramod R Padole
Recently uploaded
Antibiotics are medicines that fight infections caused by bacteria in humans and animals by either killing the bacteria or making it difficult for the bacteria to grow and multiply. Bacteria are germsABHISHEK ANTIBIOTICS PPT MICROBIOLOGY // USES OF ANTIOBIOTICS TYPES OF ANTIB...

ABHISHEK ANTIBIOTICS PPT MICROBIOLOGY // USES OF ANTIOBIOTICS TYPES OF ANTIB...ABHISHEK SONI NIMT INSTITUTE OF MEDICAL AND PARAMEDCIAL SCIENCES , GOVT PG COLLEGE NOIDA
Recently uploaded (20)
Vital Signs of Animals Presentation By Aftab Ahmed Rahimoon

Vital Signs of Animals Presentation By Aftab Ahmed Rahimoon
Fun for mover student's book- English book for teaching.pdf

Fun for mover student's book- English book for teaching.pdf
SaffronCrocusGenomicsThessalonikiOnlineMay2024TalkOnline.pptx

SaffronCrocusGenomicsThessalonikiOnlineMay2024TalkOnline.pptx
Costs to heap leach gold ore tailings in Karamoja region of Uganda

Costs to heap leach gold ore tailings in Karamoja region of Uganda
ABHISHEK ANTIBIOTICS PPT MICROBIOLOGY // USES OF ANTIOBIOTICS TYPES OF ANTIB...

ABHISHEK ANTIBIOTICS PPT MICROBIOLOGY // USES OF ANTIOBIOTICS TYPES OF ANTIB...
MSC IV_Forensic medicine - Mechanical injuries.pdf

MSC IV_Forensic medicine - Mechanical injuries.pdf
Harry Coumnas Thinks That Human Teleportation is Possible in Quantum Mechanic...

Harry Coumnas Thinks That Human Teleportation is Possible in Quantum Mechanic...
Classification of Kerogen, Perspective on palynofacies in depositional envi...

Classification of Kerogen, Perspective on palynofacies in depositional envi...
Heat Units in plant physiology and the importance of Growing Degree days

Heat Units in plant physiology and the importance of Growing Degree days
dkNET Webinar: The 4DN Data Portal - Data, Resources and Tools to Help Elucid...

dkNET Webinar: The 4DN Data Portal - Data, Resources and Tools to Help Elucid...
Information science research with large language models: between science and ...

Information science research with large language models: between science and ...
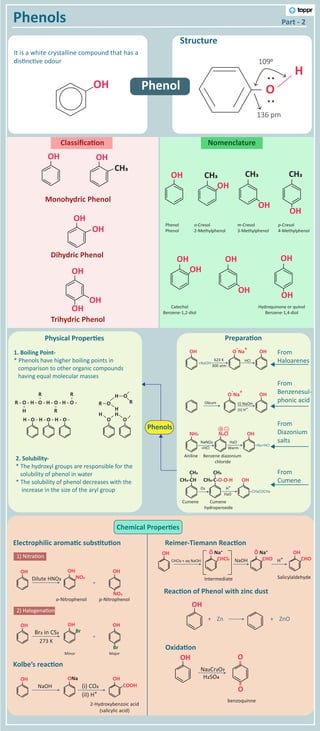
MINDMAP PHENOL.pdf
- 1. Chemical Proper�es It is a white crystalline compound that has a dis�nc�ve odour Phenols Structure Monohydric Phenol Part - 2 Nomenclature Classifica�on Dihydric Phenol Trihydric Phenol Electrophilic aroma�c subs�tu�on Reimer-Tiemann Reac�on Oxida�on Reac�on of Phenol with zinc dust Kolbe’s reac�on Phenols 1) Nitra�on 2) Halogena�on OH O H 136 pm 1090 : : OH OH CH3 OH OH OH OH OH OH OH CH3 OH OH OH CH3 OH OH OH CH3 OH OH Phenol Phenol Catechol Benzene-1,2-diol Hydroquinone or quinol Benzene-1,4-diol o-Cresol 2-Methylphenol m-Cresol 3-Methylphenol p-Cresol 4-Methylphenol Prepara�on From Haloarenes From Benzenesul- phonic acid From Diazonium salts From Cumene OH O - Na + OH +NaOH +N2+HCl 623 K 300 atm HCl NH2 - + N2Cl OH NaNO2 +HCl Warm H2O O - Na + OH Oleum (i) NaOH (ii) H⁺ +CH3COCH3 CH3-CH CH3 CH3-C-O-O-H CH3 OH O2 H2O H⁺ 1. Boiling Point- * Phenols have higher boiling points in comparison to other organic compounds having equal molecular masses 2. Solubility- * The hydroxyl groups are responsible for the solubility of phenol in water * The solubility of phenol decreases with the increase in the size of the aryl group Physical Proper�es H - O - H - O - H - O - R - O - H - O - H - O - H - O - H H R R R O O H H O R H O R OH NO2 OH NO2 OH Dilute HNO3 o-Nitrophenol + p-Nitrophenol OH Br OH Br OH Br2 in CS2 Minor + Major 273 K OH ONa OH COOH NaOH 2-Hydroxybenzoic acid (salicylic acid) (i) CO2 (iI) H⁺ OH CHCl2 O Na⁺ CHCl3 + aq NaOH Intermediate benzoquinne Salicylaldehyde NaOH CHO O Na⁺ H⁺ CHO OH OH OH + Zn H2SO4 Na2Cr2O7 + ZnO O O Phenol Benzene diazonium chloride Aniline Cumene hydroperoxide Cumene