Case record... Acute disseminated encephalomyelitis
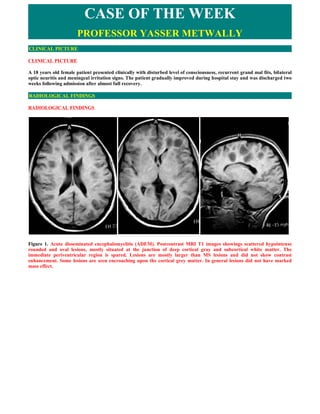
- 1. CASE OF THE WEEK PROFESSOR YASSER METWALLY CLINICAL PICTURE CLINICAL PICTURE A 18 years old female patient presented clinically with disturbed level of consciousness, recurrent grand mal fits, bilateral optic neuritis and meningeal irritation signs. The patient gradually improved during hospital stay and was discharged two weeks following admission after almost full recovery. RADIOLOGICAL FINDINGS RADIOLOGICAL FINDINGS Figure 1. Acute disseminated encephalomyelitis (ADEM). Postcontrast MRI T1 images showings scattered hypointense rounded and oval lesions, mostly situated at the junction of deep cortical gray and subcortical white matter. The immediate periventricular region is spared. Lesions are mostly larger than MS lesions and did not show contrast enhancement. Some lesions are seen encroaching upon the cortical grey matter. In general lesions did not have marked mass effect.
- 2. Figure 2. Acute disseminated encephalomyelitis (ADEM). Postcontrast MRI T1 images showings scattered hypointense rounded and oval lesions, mostly situated at the junction of deep cortical gray and subcortical white matter. The immediate periventricular region is spared. Lesions are mostly larger than MS lesions and did not show contrast enhancement. Some lesions are seen encroaching upon the cortical grey matter. In general lesions did not have marked mass effect. Figure 3. A case of acute disseminated encephalomyelitis. Notice that the multifocal cortical / subcortical hyperintense foci are sparing the periventricular region, and this is the classic pattern in ADEM. The foci, although of large size they have mild mass effect.
- 3. Figure 4. Acute disseminated encephalomyelitis (ADEM). Notice that the multifocal cortical / subcortical lesions are sparing the periventricular region. The ADEM lesions are hypointense on MRI T1 images and hyperintense on MRI T2 and FLAIR images. ADEM lesions, though large, exert mild mass effect. Figure 5. Acute disseminated encephalomyelitis (ADEM). Notice that the multifocal cortical / subcortical lesions are sparing the periventricular region. The ADEM lesions are hypointense on MRI T1 images and hyperintense on MRI T2 and FLAIR images. ADEM lesions, though large, exert mild mass effect. DIAGNOSIS: DIAGNOSIS: ACUTE DISSEMINATED ENCEPHALOMYELITIS (ADEM)
- 4. DISCUSSION DISCUSSION Acute disseminated encephalomyelitis (ADEM) presents as an acute-onset neurological dysfunction following a triggering event such as an infection or vaccination. Patients present with polysymptomatic neurological dysfunction, and imaging shows multifocal white matter lesions in the brain and spinal cord. Clinical evaluation, magnetic resonance imaging, and cerebrospinal fluid study are most useful in establishing the diagnosis and ruling out important differential diagnoses. Corticosteroids are the mainstay of treatment and the role of other modalities of treatment, such as plasma exchange and intravenous immunoglobulin, require further study. Prognosis is generally good. The recently proposed consensus definitions are likely to facilitate delineation of ADEM from other acquired demyelinating disorders. Acute disseminated encephalomyelitis (ADEM) has evolved considerably since the first case description of a patient with post-measles neurological dysfunction by Lucas in 1790. [1] Histopathologic and immunologic studies have provided considerable insight into the mechanism of the disease, and the advent of magnetic resonance imaging (MRI) has facilitated early diagnosis. With the recent introduction of consensus definitions, it is now established as a well-delineated entity. ADEM is a monophasic, inflammatory demyelinating disorder of the central nervous system (CNS). Clinical examination reveals polysymptomatic neurological involvement and imaging shows multifocal demyelinating lesions in the brain and spinal cord. It is an immune-mediated disorder that usually follows infections or vaccinations. [2,3] Though previously described as an illness with poor prognosis, recent studies have demonstrated improved outcomes following the introduction of effective immunomodulation. [4,5,6,7] This article will focus on the recent trends in the diagnosis and management of ADEM. Epidemiology The incidence of ADEM has been estimated to be 0.4 per 100,000 population per year [8] and it may represent 30% of all childhood encephalitic illnesses. [9] ADEM more commonly affects children and young adults, probably because of exposure to frequent vaccinations and exanthematous and upper respiratory tract infections in this age-group. [6,7,10,11,12,66] The mean age of onset in children ranges from 5 to 8 years. [6,12,13] ADEM generally does not show a predilection for any one sex, though two recent studies have shown a male predominance among the victims. [13,14] ADEM occurs throughout the year, but there is a significant increase in incidence during winter and spring. [6,7] Triggering Events ADEM typically follows an antigenic challenge, such as infection or vaccination, which activates the immune system. Exanthematous fevers such as measles, mumps, and rubella are the usual triggering infections. Recently, there has been a distinct change in the trend and, in regions with extensive immunization coverage, nonspecific upper respiratory tract infections are the most common triggering events. [11] However, ADEM following vaccine-preventable diseases is still prevalent in Egypt. [15] The association with infection may be difficult to establish and only 70% of ADEM patients report a triggering event. Though extensive microbiological investigations have led to the inclusion of a wide spectrum of infections in the list of those alleged to cause ADEM [Table - 1], they may fail to identify a cause in the majority of cases. [6,7,10] Table 1. Infections alleged to cause ADEM Viral Measles, Mumps, Varicella, Rubella, Influenza, A,B Hepatitis, A,B Coxsackie, Epstein-Barr, Dengue [16] and HIV [17] Bacterial Mycoplasma pneumoniae, Borrelia burgdorferi, Mycobacterium tuberculosis, Brucella, Chlamydia, Legionella, Salmonella typhi, and Leptospira, Campylobacter, Streptococcus pyogenes Vaccination Rabies, Measles, Rubella, Smallpox, Diphtheria, Mumps, Tetanus, antitoxin, Pertussis, Japanese encephalitis, Polio, Hepatitis B, Influenza, and Meningococcal A, and C Drugs Gold, Arsenical compounds, Sulfonamides, streptomycin/PAS Miscellaneous Allogenic bone marrow transplantation [19], Heart-lung transplantation [19] Herbal extracts [20], Ventriculo-atrial shunts, [21] Stings, Leprosy type I reaction Post-immunization ADEM occurs most frequently following measles, rubella, or mumps vaccination. Many vaccines have been implicated in the causation of ADEM [Table - 1]. In countries where neural tissue-based vaccines are still used, antirabies immunization with either BPL (betapropionolactone inactivated) or Semple (phenol inactivated) vaccines are important causes for ADEM. [4] Clinical Features The spectrum of neurological symptoms and signs in ADEM is broad, reflecting widespread central and peripheral nervous system (PNS) involvement. There may be a prodrome consisting of fever, headache, vomiting, and malaise.
- 5. Neurological dysfunction generally occurs within 3-6 weeks of the triggering event and may appear abruptly or may progress over several days. [6,7] Generally, ADEM is a monophasic illness and its severity may range from mild disease, with headache, subtle drowsiness, and irritability, to fulminant disease characterized by coma, decerebration, and respiratory failure. The distribution of the lesions in the nervous system determines the clinical presentation, and the commonest features are altered sensorium, pyramidal dysfunction, cerebellar ataxia, optic neuritis, and/or myelitis. Retention of urine, urinary frequency, urgency, or incontinence may occur during the acute stage and lower urinary tract dysfunction may persist even after disappearance of other neurological deficits. [23] Seizures are not uncommon and may be of focal or generalized type . [24] There may be additional features such as fever, headache, and meningismus, with the disease mimicking meningitis. Children under 3 years generally present only with encephalopathy, probably because they have immature myelin. [2,7] ADEM may have various atypical presentations. Behavioral disturbances may occasionally be the sole symptom. Presence of flaccidity and areflexia in an otherwise typical case of ADEM betrays additional PNS involvement, which is most commonly at the level of the spinal roots; [25] this picture is frequently seen in antirabies vaccination-related ADEM. [4] Combined CNS and PNS demyelination may suggest the possibility of shared pathological epitopes. [26] There is evidence to suggest that central (ADEM) and peripheral (acute and chronic inflammatory demyelinating polyradiculoneuropathy) demyelinating disorders represent two ends of a spectrum and overlap of clinical features may occur. [27] Extrapyramidal manifestations such as chorea and dystonia are rare but may be prominent in ADEM following group A streptococcal infection. [28] Occasional patients may present with focal deficits, clinical features of raised intracranial pressure, and imaging results suggesting a mass lesion. Such a presentation of ADEM, tumefactive demyelination , may be mistaken for neoplasm till histopathology establishes the diagnosis. The pattern of neurological dysfunction may be influenced by the type of triggering event. For example, post-mumps demyelination commonly presents as myelitis, post-varicella demyelination has cerebellar ataxia as its hallmark, and rubella-associated ADEM often has an explosive onset of symptoms, seizures and mild pyramidal dysfunction. [29] Acute hemorrhagic leukoencephalitis (AHLE) and acute necrotizing hemorrhagic leukoencephalitis (ANHLE) of Weston Hurst represent the hyperacute, fulminant form of postinfectious demyelination [2] [Table - 2]. Table 2. ADEM versus Acute hemorrhagic leukoencephalitis (AHLE) Feature ADEM AHLE Age group Children Young adult Triggering events Viral exanthema, respiratory tract infection, respiratory tract infection vaccination Onset Acute Hyperacute, fulminant Blood picture Normal Leukocytosis ESR/acute phase Normal Elevated reactant Urine Normal Proteinuria CSF cellularity Mononuclear Neutrophils, red blood cells Neuroimaging Multifocal white matter lesions Large lesions, mass effect, hemorrhage, necrosis Pathology Periventricular demyelination, inflammation Fibrinoid necrosis, hemorrhage, necrosis, demyelination Prognosis Good Bad Consensus Definition for ADEM Clinical research in ADEM has always been hampered by the lack of a uniform case definition, resulting in heterogeneous, incomparable study populations. The recent introduction of consensus definitions for children by the International Pediatric MS Study Group is likely to facilitate the differentiation of monophasic ADEM from other acquired demyelinating conditions [Table - 3]. [30] According to the consensus definition patients should have an acute or subacute onset of the first clinical event of multifocal CNS affection, characterized by polysymptomatic neurological dysfunction. Encephalopathy is an essential clinical feature; it is defined as either behavioral changes, alteration in consciousness, or both. MRI brain must show focal or multifocal lesions greater than 1-2 cm in size, which must be predominantly in the white matter, though it may also be present in the grey matter such as the basal ganglia or thalamus. In addition, the spinal cord may show confluent lesions. The clinical event must have a presumed inflammatory or demyelinating basis, without other etiologies. It should be followed by improvement, either clinically or on MRI, though there may be residual deficits. New or fluctuating symptoms and signs or MRI findings within three months of the initial event are considered part of the initial acute event. Table 3. Monophasic ADEM and other acquired demyelinating conditions Monophasic multifocal disease
- 6. Acute disseminated encephalomyelitis Acute hemorrhagic leukoencephalitis Clinically isolated syndromes Transverse myelitis Optic neuritis Cerebellitis Brain stem dysfunction Multiphasic and recurrent acute disseminated encephalomyelitis Is ADEM always a monophasic disease? Though ADEM is classically described as a monophasic illness, a biphasic temporal profile has been documented in 0-20% of patients. [6,7,13,14] Such patients do not proceed to develop multiple sclerosis (MS). [7] It is now possible to define patients of ADEM who develop relapses, i.e., multiphasic or recurrent ADEM. [30] A new clinical event should have occurred that meets the case definition of ADEM. The new event should occur three or more months after the initial episode. The event should occur when the patient is not on steroids and at least one month after completion of therapy. In multiphasic ADEM , clinical examination and MRI should demonstrate involvement of new areas of the CNS during the relapse. In recurrent ADEM , clinical findings and MRI should suggest involvement of the same areas that had been involved in the initial ADEM episode. Investigations Cerebrospinal fluid Cerebrospinal fluid (CSF) should be analyzed to exclude the differential diagnoses of ADEM, especially the CNS infections. CSF is abnormal in nearly three-fourth of ADEM patients and is characterized by pleocytosis, elevated proteins, and normal sugar values. [6,7] Cellular response is usually lymphocytic and counts are moderately elevated. Intrathecal oligoclonal immunoglobulin synthesis is rare and usually disappears after clinical recovery. [7,13] Electrophysiology Generalized slowing is the most common finding in electroencephalography and is nonspecific. Patients with spinal cord, brain stem, or optic nerve involvement may have abnormal somatosensory, brain stem auditory, or visual evoked potentials, respectively. [2] Imaging Computed tomography (CT) may demonstrate hypodense lesions in the white matter and basal ganglia. However, initial CT imaging may be normal in 40% of cases, as lesions may appear only 5-14 days after the onset of clinical signs. [2,31,66] MRI forms the cornerstone for diagnosis of ADEM and is more sensitive than CT in detecting lesions. Abnormalities are best appreciated in T2-weighted and fluid-attenuated inversion recovery (FLAIR) sequences as patchy, multifocal, poorly marginated lesions of increased signal intensity. White matter lesions are usually asymmetric and most frequently situated subcortically, in the cerebellum, brain stem, and spinal cord. [32,33] Thalamic and basal ganglia lesions are seen in nearly one-third of cases and may be symmetric. Corpus callosum may be affected when involvement is extensive. [3] The spinal cord becomes swollen and may show increased signal intensities, most commonly in the thoracic region. Enhancement after contrast administration is variable and occurs in acute lesions due to disruption of the blood-brain barrier. [32,33] Lesions may show complete ring, incomplete ring ('open-ring sign'), nodular, gyral, or spotty patterns of enhancement. [33,34] Tumefactive demyelination appears as a large white matter lesion with mass effect. Though clinically a monophasic illness, new lesions may appear serially over several weeks and hence, may appear to be of varying ages on MRI. [33] Occasionally, initial MR images may be normal and lesions may appear in images repeated later in the course of the disease or even during the stage of clinical improvement. [35] Though lesions generally resolve with treatment, hyperintensities may persist in MRI long after clinical recovery and is due to astrocytic hyperplasia, gliosis, or cystic changes. [36,37] Resolution of MRI abnormalities within six months of the demyelinating episode favors the diagnosis of ADEM. [38]
- 7. Figure 6. Acute disseminated encephalomyelitis. Notice the contrast enhancement which is characteristic of acute lesions. Also notice that many lesions are situated at the junction of deep cortical gray and subcortical white matter which is characteristic of ADEM Figure 7. In acute disseminated encephalomyelitis tumefactive demyelination appears as a large white matter lesion with mass effect. Newer modalities Quantitative proton MR spectroscopic imaging has shown low N-acetylaspartate (NAA) and high lactate levels in acute lesions, which normalize after recovery. [39] Further studies are required to assess the role of MR imaging techniques such as magnetization transfer and diffusion tensor imaging in detecting early or small lesions. [40] Abnormalities in 99m Tc- HMPAO SPECT appear as areas of hypoperfusion, which are more extensive than the abnormalities seen in MRI and may parallel the time course of the disease more accurately. [41] Notably, in patients with residual cognitive deficits, SPECT with acetazolamide is able to detect persistent abnormalities from areas where lesions had apparently resolved on MRI. [42,43] Pathogenesis Immunology The lesions of ADEM are due to autoimmune-mediated inflammation of the CNS, and the absence of viral or bacterial antigens in the CNS is nearly universal. [5,7] T-cells have been shown to play an important role, possibly through molecular mimicry or by nonspecific activation of autoreactive T-cell clones. [3] Interleukin-6 may be associated with proliferation of B-lymphocytes and immunoglobulin G synthesis. [44] Anti-basal ganglia antibodies have been demonstrated in children with classical features of ADEM following streptococcal infection. [28] A complex interplay between cytokines and adhesion molecules is responsible for the cellular events of inflammatory encephalomyelitis and oligodendrocyte death. An association has been established between ADEM and certain class II HLA alleles, indicating
- 8. that genetic factors may play a role in immunoregulation and progression from infection/vaccination to ADEM. [29,66] Pathology Histopathology studies have demonstrated perivenous cuffing with inflammatory cells, especially lymphocytes and macrophages, and loss of myelin. Most of the changes are seen in white matter. However, grey matter also shows changes, including chromatolysis and neuronal loss, especially in the thalami, deep cortical lamina, and hypothalamus. The meninges may also show inflammatory reaction. [2,45] Figure 8. Histopathology studies In ADEM have demonstrated perivenous cuffing with inflammatory cells, especially lymphocytes and macrophages, and loss of myelin Differential Diagnosis In the absence of specific biological markers, the diagnosis of ADEM is based upon clinical and imaging features. Clinical evaluation, neuroimaging, and blood and CSF analysis can help to distinguish ADEM from other conditions. [46] Herpes simplex encephalitis commonly presents with abnormal behavior and focal/secondary generalized seizures, which are more frequent and difficult to treat than in ADEM. MRI, EEG, and CSF polymerase chain reaction for Herpes simplex virus help in confirmation of the diagnosis. Japanese encephalitis presents with acute encephalopathy. MRI may show bilateral thalamic lesions, akin to ADEM. History, EEG findings, and CSF evidence of antibodies to the virus are useful for differentiation. Other infections such as bacterial meningitis and brain abscess can be ruled out by relevant investigations such as imaging and lumbar puncture. Complicated tuberculous meningitis may sometimes mimic ADEM and can be excluded if CSF sugar is normal and cultures are sterile. Neuromyelitis optica is characterized by optic neuritis and myelitis, with spinal lesions extending over three or more segments, features that may also be seen in ADEM. It may be distinguished from ADEM by the relative paucity of white matter lesions in MRI of the brain and presence of antibodies to aquaporin 4 (NMO-IgG). [47] Patients with Behcet's disease may present with multifocal neurological signs due to brain and spinal cord involvement. Clinical and imaging features may resemble ADEM, and the history of recurrent mucocutaneous ulcers of the mouth and genitalia will be essential to establish the diagnosis. Antiphospholipid antibody syndrome may also mimic the clinical and MRI features of ADEM and should be ruled out by measuring the specific antibodies. Immune-mediated disorders such as systemic lupus erythematosus, Sjögren syndrome, and sarcoidosis may present with neurological dysfunction and multifocal white matter changes and can be diagnosed by history and relevant blood tests. Susac's syndrome may present with subacute encephalopathy, with MRI of the brain showing multiple white matter lesions; however, it can be differentiated from ADEM based on additional features such as the presence of headache, visual impairment due to retinal artery branch occlusion, sensorineural hearing loss, and specific involvement of central corpus callosum in MRI. [48,66] The diagnosis of recurrent ADEM should be made only after excluding other lesions such as arteriovenous malformations, neoplasms, and vasculitis.
- 9. Metabolic leukoencephalopathies such as metachromatic leukodystrophy (MLD), adrenoleukodystrophy, and MELAS (mitochondrial myopathy, encephalopathy, lactic acidosis, and stroke-like episodes) may mimic childhood ADEM. Leukodystrophies may have a stepladder clinical course, with the exacerbations associated with febrile illnesses. History may reveal prior neurological dysfunction. MRI may show symmetric, confluent white matter lesions, hallmarks of inherited rather than acquired leukoencephalopathies. Specific investigations such as serum aryl sulfatase for MLD and very long-chain fatty acids (VLCFA) levels for adrenoleukodystrophy will confirm the diagnosis. The clinical features of mitochondrial cytopathies such as MELAS may be similar to that of ADEM and lesions seen on MRI may be transient. Serum and CSF lactate levels may be elevated during the acute episode and MR spectroscopic imaging may show elevated lactate in the affected tissues. ADEM or multiple sclerosis? In a patient presenting with neurological dysfunction and MRI showing multiple white matter lesions, the most important differential diagnosis is MS. Distinguishing between ADEM and MS is a diagnostic challenge and has important therapeutic and prognostic implications. There are several clinical, imaging, and laboratory parameters that may be useful to distinguish between the two [Table - 4]. CSF electrophoresis has shown a significant reduction in the beta-1 globulin fraction in patients with MS as compared to those with ADEM and this may be a potential CSF marker. [49] Features that strongly favor ADEM include a history of preceding infection, polysymptomatic neurological dysfunction, encephalopathy, grey matter involvement on MRI, and absence of oligoclonal bands in CSF. [6,7] Often, distinction between these two conditions cannot be made with certainty and follow-up with serial MRI may be necessary to establish the diagnosis. [3,36,66] Table 4. Differences between ADEM and multiple sclerosis Feature ADEM MS Onset Abrupt Subacute Triggering events Preceding infection or vaccination in 70 Uncommon % of cases Age group More common in children More common in young adult Temporal profile Monophasic, rarely relapsing Relapsing Clinical features Altered sensorium More common Rare Seizures More common Rare Neurological deficit Multifocal Usually single deficit Optic neuritis Bilateral Unilateral Myelitis Complete, transverse myelitis Partial myelopathy Lower motor neuron signs More common Rare Headache More common Rare Meningismus More common Rare Neuroimaging findings Distribution of the lesions Bilateral extensive lesions Scattered asymmetric lesions White matter lesions Confluent, ill-defined periventricular and Well-defined, with periventricular subcortical white matter lesions preponderance Corpus callosum lesions Rare Common Grey matter involvement Thalamic and basal ganglionic lesions are Uncommon common Edema and mass effect May be present Uncommon Follow-up MRI No new lesions New lesions with dissemination in time and place Cerebrospinal fluid Cell count Mild to moderate pleocytosis Normal to mild pleocytosis Protein Increased Normal Oligoclonal bands Uncommon, transient Common and persistent Mortality 10-25 Uncommon Treatment
- 10. Public health initiatives are important for minimizing exposure to the triggering events of ADEM. These include implementing effective vaccination programs and avoiding vaccines containing neural tissues or enveloped viruses. [2] Early initiation of antibiotics for infections may not prevent the immune trigger that culminates in ADEM. [50] Treatment of ADEM involves immunomodulation that aims to counter autoimmune-mediated inflammation. Though spontaneous resolution has been described, recovery is usually incomplete and hence patients presenting in the acute stage with significant, progressive neurological deficits should receive treatment. [51] In view of the rarity of ADEM, treatment regimes for children and adults are based on anecdotal reports and lack validation by randomized controlled trials. High- dose corticosteroids have become the first line of management and are associated with rapid recovery when given early in the disease. [6,7,13,14,52] Earlier studies used ACTH, but nowadays either intravenous methylprednisolone (10-30 mg/kg/day, up to maximum of 1 gm/day) or dexamethasone (1 mg/kg/day) for 3 to 5 days are being used. [6,7,13,14] Full recovery was reported in 50-80% of patients. Methylprednisolone treated patients have significantly better outcome with respect to disability compared to those treated with dexamethasone. [13] In addition to immunosuppression, steroids also have anti-inflammatory and antiedema actions, and the immediate improvement following administration of steroids is likely to be due to reduction in cerebral edema. [53] Oral corticosteroids are continued and gradually tapered over six weeks to minimize the risk of recurrence. [7] The role of corticosteroids in patients presenting late in the course of the disease is questionable. [54] Vaccinations should be avoided during the first six months after recovery. [7] Plasma exchange is another therapeutic modality, and its role in the management of demyelinating disorders that are nonresponsive to corticosteroids has been established in one randomized control trial. [55] In most studies, 1-1.4 plasma volumes were exchanged by continuous flow centrifugation and 70% of volume was replaced by 5% serum albumin, fresh- frozen plasma, or hydroxyethyl starch. Patients received 2-20 exchanges, depending on the severity of illness and clinical response. It is likely to be effective in patients of ADEM not responding to corticosteroids, especially when given early in the course of the disease. [56,57] Intravenous immunoglobulin is yet another option and the results so far are impressive, especially in corticosteroid nonresponders. In most studies, 0.4 gm/kg/day was given over 5 days and improvement was noted within 2 to 3 days. [58,59,60] However, in most reported cases, patients had received prior corticosteroids and the delayed effects of these drugs could not be ruled out as the reason for the improvement. At present, the evidence is insufficient to design a treatment algorithm for ADEM. However, based on the available reports, plasma exchange and intravenous immunoglobulin are likely to be second-line therapies when corticosteroids fail. The relative efficacies of the two have not been evaluated. The choice of second-line therapy should be individualized, depending upon the type of CNS involvement, complications, and comorbidities. For example, autonomic dysfunction and hypotension would preclude the use of plasma exchange. Anecdotal reports suggest that intravenous immunoglobulin may be more effective in patients with who also have PNS involvement [25] and plasma exchange in patients with tumefactive demyelination. [61] Combination therapy with methylprednisolone and immunoglobulin has been reported to be successful in atypical cases of ADEM. [62] Cyclophosphamide [52] and hypothermia [63] have been successfully tried in fulminant ADEM. Hemicraniectomy has been reported to be lifesaving in patients with massive life-threatening cerebral edema refractory to conventional medical management. [64,65] Outcome ADEM is usually a monophasic illness lasting 2-4 weeks. [2] However recovery may continue for weeks or months. While earlier studies reported mortality of 20% and significant morbidity amongst survivors, recent reports suggest excellent outcomes, especially in children. [7,10,13,14] Visual impairment, weakness, lower urinary tract dysfunction, cognitive impairment, and behavioral changes may persist at follow-up. [7,13,23] Children thought to have made a full recovery may show subtle neurocognitive and behavioral changes on formal assessment, though these may be less severe than the changes seen in children with MS. [43] Patients having progressive neurological dysfunction , altered sensorium, and seizures have poor long-term outcomes. [24] SUMMARY SUMMARY ADEM presents as an acute onset neurological dysfunction following infection or vaccination. Clinical evaluation, MRI, and CSF study are most useful to establish the diagnosis and rule out important differential diagnoses. The recently proposed consensus definitions are likely to facilitate delineation of ADEM from other acquired demyelinating disorders. Corticosteroids are the most important treatment modality, while the role of other agents requires further study. Prognosis is generally good.
- 11. Addendum A new version of this PDF file (with a new case) is uploaded in my web site every week (every Saturday and remains available till Friday.) To download the current version follow the link quot;http://pdf.yassermetwally.com/case.pdfquot;. You can also download the current version from my web site at quot;http://yassermetwally.comquot;. To download the software version of the publication (crow.exe) follow the link: http://neurology.yassermetwally.com/crow.zip The case is also presented as a short case in PDF format, to download the short case follow the link: http://pdf.yassermetwally.com/short.pdf At the end of each year, all the publications are compiled on a single CD-ROM, please contact the author to know more details. Screen resolution is better set at 1024*768 pixel screen area for optimum display REFERENCES References 1. Lucas J. An account of uncommon symptoms succeeding the measles: With additional remarks on the infection of measles and smallpox. London Med J 1790;11:325-31. 2. Coyle PK. Post infectious encephalomyelitis. In : Kennedy PGE, Davis LE, editors. Infectious Diseases of the Nervous System. Butterworth-Heinemann: Oxford; 2000. p. 83-108. 3. Garg RK. Acute disseminated encephalomyelitis. Postgrad Med J 2003;79:11-7. 4. Swamy HS, Shankar SK, Chandra PS, Aroor SR, Krishna AS, Perumal VG. Neurological complications due to beta- propiolactone (BPL)-inactivated antirabies vaccination. Clinical, electrophysiological and therapeutic aspects. J Neurol Sci 1984;63:111-28. 5. Johnson RT, Griffin DE, Hirsch RL, Wolinsky JS, Roedenbeck S, de Soriano IL, et al . Measles encephalomyelitis- clinical and immunologic studies. N Engl J Med 1984;310:137-41. 6. Hynson JL, Kornberg AJ, Coleman LT, Shield L, Harvey AS, Kean MJ. Clinical and neuroradiologic features of acute disseminated encephalomyelitis in children. Neurology 2001;56:1308-12. 7. Dale RC, de Sousa C, Chong WK, Cox TC, Harding B, Neville BG. Acute disseminated encephalomyelitis, multiphasic disseminated encephalomyelitis and multiple sclerosis in children. Brain 2000;123:2407-22. 8. Leake JA, Albani S, Kao AS, Senac MO, Billman GF, Nespeca MP, et al . Acute disseminated encephalomyelitis in childhood: Epidemiologic, clinical and laboratory features. Pediatr Infect Dis J 2004;23:756-64. 9. Kennard C, Swash M. Acute viral encephalitis: Its diagnosis and outcome. Brain 1981;104:129-48. 10. Murthy SN, Faden HS, Cohen ME, Bakshi R. Acute disseminated encephalomyelitis in children. Pediatrics 2002;110:e1-7. 11. Menge T, Hemmer B, Nessler S, Wiendl H, Neuhaus O, Hartung HP, et al . Acute disseminated encephalomyelitis: An update. Arch Neurol 2005;62:1673-80. 12. Anlar B, Basaran C, Kose G, Guven A, Haspolat S, Yakut A, et al . Acute disseminated Encephalomyelitis in Children: Outcome and Prognosis. Neuropediatrics 2003;34:194-9. 13. Tenembaum S, Chamoles N, Fejerman N. Acute disseminated encephalomyelitis: A long-term follow-up study of 84 pediatric patients. Neurology 2002;59:1224-31. 14. Singhi PD, Ray M, Singhi S, Khandelwal NK. Acute disseminated encephalomyelitis in North Indian children: Clinical profile and follow-up. J Child Neurol 2006;21:851-7. 15. Murthy JM, Yangala R, Meena AK, Jaganmohan Reddy J. Acute disseminated encephalomyelitis: clinical and MRI Study from South India. J Neurol Sci 1999;165:133-8. 16. Yamamoto Y, Takasaki T, Yamada K, Kimura M, Washizaki K, Yoshikawa K, et al . Acute disseminated encephalomyelitis following dengue fever. J Infect Chemother 2002;8:175-7.
- 12. 17. Narciso P, Galgani S, Del Grosso B, De Marco M, De Santis A, Balestra P, et al . Acute disseminated encephalomyelitis as manifestation of primary HIV infection. Neurology 2001;57:1493-6. 18. Tomonari A, Tojo A, Adachi D, Iseki T, Ooi J, Shirafuji N, et al . Acute disseminated encephalomyelitis (ADEM) after allogeneic bone marrow transplantation for acute myeloid leukemia. Ann Hematol 2003;82:37-40. 19. Horowitz MB, Comey C, Hirsch W, Marion D, Griffith B, Martinez J. Acute disseminated encephalomyelitis (ADEM) or ADEM-like inflammatory changes in a heart-lung transplant recipient: A case report. Neuroradiology 1995;37:434-7. 20. Schwarz S, Knauth M, Schwad S, Walter-Sack I, Bonmann E, Storch-Hagenlocher B. Acute disseminated encephalomyelitis after parenteral therapy with herbal extracts: A report of two cases. J Neurol Neurosurg Psychiatry 2000;69:516-8. 21. Boz C, Velioglu S, Ozmenoglu M. Acute disseminated encephalomyelitis after bee sting. Neurol Sci 2003;23:313-5. 22. Jacob J, Alexander M, Aaron S, Pulimood S, Walter N, Gnanamuthu C. Acute disseminated encephalomyelitis in Hansen's disease. Ann Indian Acad Neurol 2006;9:166-8. 23. Sakakibara R, Hattori T, Yasuda K, Yamanishi T. Micturitional disturbance in acute disseminated encephalomyelitis (ADEM). J Auton Nerv Syst 1996;60:200-5. 24. Panicker JN. Acute Disseminated Encephalomyelitis: Clinical Profile and Predictors of Outcome (dissertation). Bangalore: National Institute of Mental Health and Neurosciences, NIMHANS (Deemed University); 2004. 25. Marchioni E, Ravaglia S, Piccolo G, Furione M, Zardini E, Franciotta D, et al . Postinfectious inflammatory disorders: Subgroups based on prospective follow-up. Neurology 2005;65:1057-65. 26. Kinoshita A, Hayashi M, Miyamoto K, Oda M, Tanabe H. Inflammatory demyelinating polyradiculitis in a patient with acute disseminated encephalomyelitis (ADEM). J Neurol Neurosurg Psychiatry 1996;60:87-90. 27. Krivickas LS, Hochberg FH, Freeman S. Chronic inflammatory demyelinating polyradiculoneuropathy with tumefactive central demyelination. Muscle Nerve 2006;33:283-8. 28. Dale RC, Church AJ, Cardoso F, Goddard E, Cox TC, Chong WK, et al . Post streptococcal acute disseminated encephalomyelitis with basal ganglia involvement and auto-reactive antibasal ganglia antibodies. Ann Neurol 2001;50:588- 95. 29. Idrissova ZR, Boldyreva MN, Dekonenko EP, Malishev NA, Leontyeva IY, Martinenko IN, et al . Acute disseminated encephalomyelitis in children: Clinical features and HLA-DR linkage. Eur J Neurol 2003;10:537-46. 30. Krupp LB, Banwell B, Tenembaum S; International Pediatric MS Study Group. Consensus definitions proposed for pediatric multiple sclerosis and related disorders. Neurology 2007;68:S7-12. 31. Lukes SA, Norman D. Computed tomography in acute disseminated encephalomyelitis. Ann Neurol 1983;13:567-72. 32. Mader I, Stock KW, Ettlin T, Probst A. Acute disseminated encephalomyelitis: MR and CT features. AJNR Am J Neuroradiol 1996;17:104-9. 33. Caldemeyer KS, Smith RR, Harris TM, Edwards MK. MRI in acute disseminated encephalomyelitis. Neuroradiology 1994;36:216-20. 34. van der Meyden CH, de Villiers JF, Middlecote BD, Terblanche J. Gadolinium ring enhancement and mass effect in acute disseminated encephalomyelitis. Neuroradiology 1994;36:221-3. 35. Honkaniemi J, Dastidar P, Kdhdrd V, Haapasalo H. Delayed MR imaging changes in acute disseminated encephalomyelitis. AJNR Am J Neuroradiol 2001;22:1117-24. 36. Kesselring J, Miller DH, Robb SA, Kendall BE, Moseley IF, Kingsley D, et al . Acute disseminated encephalomyelitis. MRI findings and the distinction from multiple sclerosis. Brain 1990;113:291-302. 37. Go T, Imai T. A residual cystic lesion in acute disseminated encephalomyelitis. Neuroradiology 2000;42:682-4. 38. Richer LR, Sinclair DB, Bhargava R. Neuroimaging features of acute disseminated encephalomyelitis in childhood. Pediatr Neurol 2005;32:30-6. 39. Mader I, Wolff M, Nagele T, Niemann G, Grodd W, Kuker W. MRI and proton MR spectroscopy in acute disseminated encephalomyelitis. Childs Nerv Syst 2005;21:566-72.
- 13. 40. Inglese M, Salvi F, Iannucci G, Mancardi GL, Mascalchi M, Filippi M. Magnetization transfer and Diffusion tensor MR imaging of acute disseminated encephalomyelitis. AJNR Am J Neuroradiol 2002;23:267-72. 41. Itti E, Huff K, Cornford ME, Itti L, Poruri K, Mishkin FS. Postinfectious encephalitis. A co registered SPECT and magnetic resonance imaging study. Clin Nucl Med 2002;27:129-30. 42. Okamoto M, Ashida KI, Imaizumi M. Hypoperfusion following encephalitis: SPECT with acetazolamide. Eur J Neurol 2001;8:471-4. 43. Hahn CD, Miles BS, MacGregor DL, Blaser SI, Banwell BL, Hetherington CR. Neurocognitive outcome after acute disseminated encephalomyelitis. Pediatr Neurol 2003;29:117-23. 44. Dale RC, Morovat A. Interleukin-6 and oligoclonal IgG synthesis in children with acute disseminated encephalomyelitis. Neuropediatrics 2003;34:141-5. 45. Kumar A, Swamy HS, Santhosh V, Taly AB, Arunodaya GR, Shankar SK. Pathology of allergic encephalomyelitis following Semple type antirabies vaccine from India. Neurol Infect Epidemiol 1997;2:239-48. 46. Hahn JS, Pohl D, Rensel M, Rao S; International Pediatric MS Study Group. Differential diagnosis and evaluation in pediatric multiple sclerosis. Neurology 2007;68:S13-22. 47. Wingerchuk DM, Lennon VA, Pittock SJ, Lucchinetti CF, Weinshenker BG. Revised diagnostic criteria for neuromyelitis optica. Neurology 2006;66:1485-9. 48. Susac JO. Susac syndrome: The triad of microangiopathy of the brain and retina with hearing loss in women. Neurology 1994;44:591-3. 49. Chopra B, Abraham R, Abraham A. CSF beta-1 Globulin- a potential marker in differentiating multiple sclerosis and acute disseminated encephalomyelitis: A preliminary study. Neurol India 2002;50:41-4. 50. Pellegrini M, O'Brien TJ, Hoy J, Sedal L. Mycoplasma pneumoniae infection associated with an acute brainstem syndrome. Acta Neurol Scand 1996;93:203-6. 51. Rust RS, Dodson W, Prensky A, Chun R, DeVivo D, Dodge P, et al . Classification and outcome of acute disseminated encephalomyelitis. Ann Neurol 1997;42:491. 52. Schwarz S, Mohr A, Knauth M, Wildemann B, Storch-Hagenlocher B. Acute disseminated encephalomyelitis: A follow- up study of 40 adult patients. Neurology 2001;56:1313-8. 53. Barkhof F, Hommes OR, Scheltens P, Valk J. Quantitative MRI changes in gadolinium-DTPA enhancement after high dose intravenous methyl prednisolone in multiple sclerosis. Neurology 1991;41:1219-22. 54. Straub J, Chofflon M, Delavelle J. Early high-dose intravenous methylprednisolone in acute disseminated encephalomyelitis: A successful recovery. Neurology 1997;49:1145-7. 55. Weinshenker BG, O'Brien PC, Petterson TM, Noseworthy JH, Lucchinetti CF, Dodick DW, et al . A randomized trial of plasma exchange in acute central nervous system inflammatory demyelinating disease. Ann Neurol 1999;46:878-86. 56. Lin CH, Jeng JS, Yip PK. Plasmapheresis in acute disseminated encephalomyelitis. J Clin Apheresis 2004;19:154-9. 57. Keegan M, Pineda AA, McClelland RL, Darby CH, Rodriguez M, Weinshenker BG. Plasma exchange for severe attacks of CNS demyelination: Predictors of response. Neurology 2002;58:143-6. 58. Pradhan S, Gupta RP, Shashank S, Pandey N. Intravenous immunoglobulin therapy in acute disseminated encephalomyelitis. J Neurol Sci 1999;165:56-61. 59. Sahlas DJ, Miller SP, Guerin M, Veilleux M, Francis G. Treatment of acute disseminated encephalomyelitis with intravenous immunoglobulin. Neurology 2000;54:1370-2. 60. Kleiman M, Brunquell P. Acute disseminated encephalomyelitis: Response to intravenous immunoglobulin? J Child Neurol 1995;10:481-3. 61. Mao-Draayer Y, Braff S, Pendlebury W, Panitch H. Treatment of steroid-unresponsive tumefactive demyelinating disease with plasma exchange. Neurology 2002;59:1074-7. 62. Straussberg R, Schonfeld T, Weitz R, Karmazyn B, Harel L. Improvement of atypical acute disseminated encephalomyelitis with steroids and intravenous immunoglobulins. Pediatr Neurol 2001;24:139-43.
- 14. 63. Takata T, Hirakawa M, Sakurai M, Kanazawa I. Fulminant form of acute disseminated encephalomyelitis: Successful treatment with hypothermia. J Neurol Sci 1999;165:94-7. 64. von Stuckrad-Barre S, Klippel E, Foerch C, Lang JM, du Mesnil de Rochemont R, Sitzer M. Hemicraniectomy as a successful treatment of mass effect in acute disseminated encephalomyelitis. Neurology 2003;61:420-1. 65. Refai D, Lee MC, Goldenberg FD, Frank JI. Decompressive hemicraniectomy for acute disseminated encephalomyelitis: Case report. Neurosurgery 2005;56:E872. 66. Metwally, MYM: Textbook of neurimaging, A CD-ROM publication, (Metwally, MYM editor) WEB-CD agency for electronic publishing, version 9.1a January 2008
