IB Chemistry on Reactivity Series vs Electrochemical Series
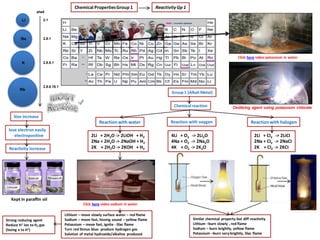
- 1. 2Li + CI2 -> 2LiCI 2Na + CI2 -> 2NaCI 2K + CI2 -> 2KCI Chemical PropertiesGroup 1 Size increase Reactionwith water Click here video potassium in water shell 2.1 2.8.1 2.8.8.1 2.8.8.18.1 Na Li K Rb lose electron easily electropositive Reactivity increase Group 1 (Alkali Metal) Chemicalreaction 2Li + 2H2O -> 2LiOH + H2 2Na + 2H2O -> 2NaOH + H2 2K + 2H2O -> 2KOH + H2 Reaction with oxygen Reactionwith halogen Lithium – move slowly surface water – red flame Sodium – move fast, hissing sound – yellow flame Potassium – move fast, ignite - lilac flame Turn red litmus blue- produce hydrogen gas Solution of metal hydroxide/alkaline produced Click here video sodium in water Similar chemical property but diff reactivity Lithium –burn slowly , red flame Sodium – burn brightly, yellow flame Potassium –burn very brightly, lilac flame Kept in paraffin oil Strong reducing agent Reduce H+ ion to H2 gas (losing e to H+) Oxidizing agent using potassium chlorate ReactivityGp 1 4Li + O2 -> 2Li2O 4Na + O2 -> 2Na2O 4K + O2 -> 2K2O
- 2. Reactivity Series Reactivityseries Metals with water, acids, oxygen Reactivity series Non metal, Hydrogen and Carbon Displacement rxn (H atom from H2O/HCI) Reactive metal displace H atom from water 2K + 2H2O → 2KOH + H2 Ca + 2H2O → Ca(OH)2 + H2 Less reactive metal displace H atom from acid Mg + 2HCI → MgCI2 + H2 Zn + H2SO4 → ZnSO4 + H2 Unreactive metal – No rxn with water /acid Au + HCI → Displacement rxn (REDOXreaction) Reactive metaldisplace less reactive metalfrom its solReactivity series Displacement rxn (O atom from less reactive) Reactive metal displace O from less reactive metal 2Al + Fe2O3 → Al2O3 + 2Fe Zn + PbO → ZnO + Pb Displacement rxn (O atom from less reactive) Reactive non metal displace O from less reactive metal C + 2Fe2O3→ 3CO2 + 4Fe H2 + CuO→ H2O + Cu Displacement rxn (less reactive ions) Reactive metal displace less reactive ions from its salt Zn + CuSO4 → ZnSO4 + Cu 2Al + 3CuCI2 → 2AlCI3 + 3Cu Reactive metal Click here AI/CuCI3 displacement Click here to view Flinn Scientific Click here Iron extraction (Thermite) • Metal arranged accordingto their ability to lose electron - form +ve ions • Measure tendency of metals in losing electrons (Undergooxidation) • Metals – lose electrons – form electropositiveions – Oxidation Process Click here microscale Fe reduction lithium How fast rxn happen? (Kinetics)
- 3. ElectrochemicalSeries STANDARD Reduction potential – H2 as std Oxidized sp ↔ Reduced sp Eθ/V Li+ + e- ↔ Li -3.04 K+ + e- ↔ K -2.93 Ca2+ + 2e- ↔ Ca -2.87 Na+ + e- ↔ Na -2.71 Mg 2+ + 2e- ↔ Mg -2.37 Al3+ + 3e- ↔ AI -1.66 Mn2+ + 2e- ↔ Mn -1.19 H2O + e- ↔ H2+OH- -0.83 Zn2+ + 2e- ↔ Zn -0.76 Fe2+ + 2e- ↔ Fe -0.45 Ni2+ + 2e- ↔ Ni -0.26 Sn2+ + 2e- ↔ Sn -0.14 Pb2+ + 2e- ↔ Pb -0.13 H+ + e- ↔ H2 0.00 Cu2+ + e- ↔ Cu+ +0.15 SO4 2- + 4H+ + 2e- ↔ H2SO3 + H2O +0.17 Cu2+ + 2e- ↔ Cu +0.34 1/2O2 + H2O +2e- ↔ 2OH- +0.40 Cu+ + e- ↔ Cu +0.52 1/2I2 + e- ↔ I- +0.54 Fe3+ + e- ↔ Fe2+ +0.77 Ag+ + e- ↔ Ag +0.80 1/2Br2 + e- ↔ Br- +1.07 1/2O2 + 2H+ +2e- ↔ H2O +1.23 Cr2O7 2-+14H+ +6e- ↔ 2Cr3+ +7H2O +1.33 1/2CI2 + e- ↔ CI- +1.36 MnO4 - + 8H+ + 5e- ↔ Mn2+ + 4H2O +1.51 1/2F2 + e- ↔ F +2.87 - ve reduction potential + ve reduction potential Compared to H2 as std Eθ cell/Cell Potential = EMF in volt EMF when half cell connect to SHE std condition Std potential written as std reduction potential TOP right • High ↑ tendency lose e • Li → Li + + e • Eθ Li = +3.04V • STRONG reducing Agent •Oxi favourable(Eθ = +ve) STRONG Reducing Agent WEAK Reducing Agent BOTTOM right • Low ↓ tendency lose e • F - → 1/2F2 + e • Eθ F2 = - 2.87V • WEAK reducing Agent •Oxi NOT favourable (Eθ =-ve) WEAK Oxidizing Agent Strong Oxidizing Agent TOP left • Low ↓ tendency gain e • Li+ + e → Li • Eθ Li= - 3.04V • WEAK oxidizingAgent • Red NOT favourable (Eθ = -ve) BOTTOM left • High ↑ tendency gain e • F2 + 2e → 2F- • Eθ F2= +2.87V • STRONG oxidizing Agent •Red favourable (Eθ = +ve) Thermodynamics measurement
- 4. ReactivitySeries lithium Li Potassium > Sodium > Lithium ElectrochemicalSeries Reactivityvs ElectrochemicalSeries Oxidized sp ↔ Reduced sp Eθ/V Li+ + e- ↔ Li -3.04 K+ + e- ↔ K -2.93 Ca2+ + 2e- ↔ Ca -2.87 Na+ + e- ↔ Na -2.71 Mg 2+ + 2e- ↔ Mg -2.37 Al3+ + 3e- ↔ AI -1.66 Zn2+ + 2e- ↔ Zn -0.76 Fe2+ + 2e- ↔ Fe -0.45 Ni2+ + 2e- ↔ Ni -0.26 Pb2+ + 2e- ↔ Pb -0.13 Cu2+ + 2e- ↔ Cu +0.34 Ag+ + e- ↔ Ag +0.80 Lithium > Potassium > Sodium Electrochemical Series - Thermodynamics measurement ↓ Eθ value give – energetics feasibility of rxn- not rate/kinetics ↓ Rxn may be feasible, but to slow to happen/no observable sign – Ea too high ↓ Measurement of voltage/potential using Std H2 Electrode Reactivity – Kinetics ↓ How fast/metal with water and acid ↓ Due to low Ea – easier to react ↓ Potassium + water = faster/reactive Lithium + water = slower/less reactive Strong Correlation but may not be the same ↓ Li to Li+ ion more thermodynamically favourable than K to K+ ion ↓ K more reactive than Li in water/acid – due to kinetics factor
- 5. ElectrochemicalSeries - Thermodynamics measurement M(s) → M+ (g) + e ∆Ha/kJ mol-1 ∆Hhyd/kJ mol-1 Li +161 +519 -499 Na +108 +494 -390 K +90 +418 -305 3 Steps rxn: M (s) → M (g) ∆H = enthalpy of atomization M (g) → M+ (g) ∆H = enthalpy of ionization M+ (g) → M+ (aq) ∆H = enthalpy of hydration ElectrochemicalSeries STD Oxidation potential Reduced sp ↔ Oxidized sp Eθ/V Li ↔ Li+ + e +3.04 K ↔ K+ + e +2.93 Na ↔ Na+ + e +2.71 Li(s) Li → Li+ (g) ∆Ha = +161 ∆HI = +519 ∆Hhyd = - 499 Li+ (g) → Li+ (aq) Li(s) → Li + (aq) ∆H = +181 Li(s) → Li (g) ∆Ha = +90 K (s) K (s) → K (g) ∆HI = +418 ∆Hhyd = - 305 K+ (g) → K+ (aq) K(s) → K + (aq) ∆H = +203 Na (s) ∆Ha = +108 Na(s) → Na(g) ∆HI = +494 K → K+ (g) Na → Na+ (g) ∆Hhyd = - 390 Na+ (g) → Na+ (aq) Na(s) → Na+ (aq) ∆H = +212 Lithium – least ∆H change - Most energetically favourable -∆H = spontaneous/favourable -∆H = spontaneous/favourable ↓ Li → Li+ + e +Eθ Potassium – High ∆H change - Less energetically favourable -∆H = spontaneous/favourable -∆H = spontaneous/favourable ↓ K → K+ + e +Eθ Sodium – Highest∆H change - Least energeticallyfavourable +∆H = NON spontaneous/favourable +∆H = NON spontaneous/favourable ↓ Na → Na+ + e +Eθ Li Na K Lithium – Size smaller ↓ Easily hydrated → - ∆H favourable ↓ IE High – strong NC due to small size Potassium– Size bigger ↓ Diff hydrated → +∆H non favourable ↓ IE Low – weak NC due to large size
- 6. ElectrochemicalSeries STD Oxidation potential Reduced sp ↔ Oxidized sp Eθ/V Li ↔ Li+ + e +3.04 K ↔ K+ + e +2.93 Na ↔ Na+ + e +2.71 Li(s) Li → Li+ (g) ∆Ha = +161 ∆HI = +519 ∆Hhyd = - 499 Li+ (g) → Li+ (aq) Li(s) → Li + (aq) ∆H = +181 Li(s) → Li (g) ∆Ha = +90 K (s) K (s) → K (g) ∆HI = +418 ∆Hhyd = - 305 K+ (g) → K+ (aq) K(s) → K + (aq) ∆H = +203 Na (s) ∆Ha = +108 Na(s) → Na(g) ∆HI = +494 K → K+ (g) Na → Na+ (g) ∆Hhyd = - 390 Na+ (g) → Na+ (aq) Na(s) → Na+ (aq) ∆H = +212 Lithium – least ∆H change - Most energetically favourable -∆H = spontaneous/favourable -∆H = spontaneous/favourable ↓ Li → Li+ + e +Eθ Potassium – High ∆H change - Less energetically favourable -∆H = spontaneous/favourable -∆H = spontaneous/favourable ↓ K → K+ + e +Eθ Sodium – Highest∆H change - Least energeticallyfavourable +∆H = NON spontaneous/favourable +∆H = NON spontaneous/favourable ↓ Na → Na+ + e +Eθ ReactivitySeries Potassium > Sodium > Lithium Lithium > Potassium > Sodium vs Reactivityvs ElectrochemicalSeries Lithium is above Potassium in electrochemicalseries ↓ Lithium is below Potassium in ReactivitySeries ↓ Due to kinetics factors/activationenergy, Rxn is slower Potassium K Sodium Na Lithium Li
