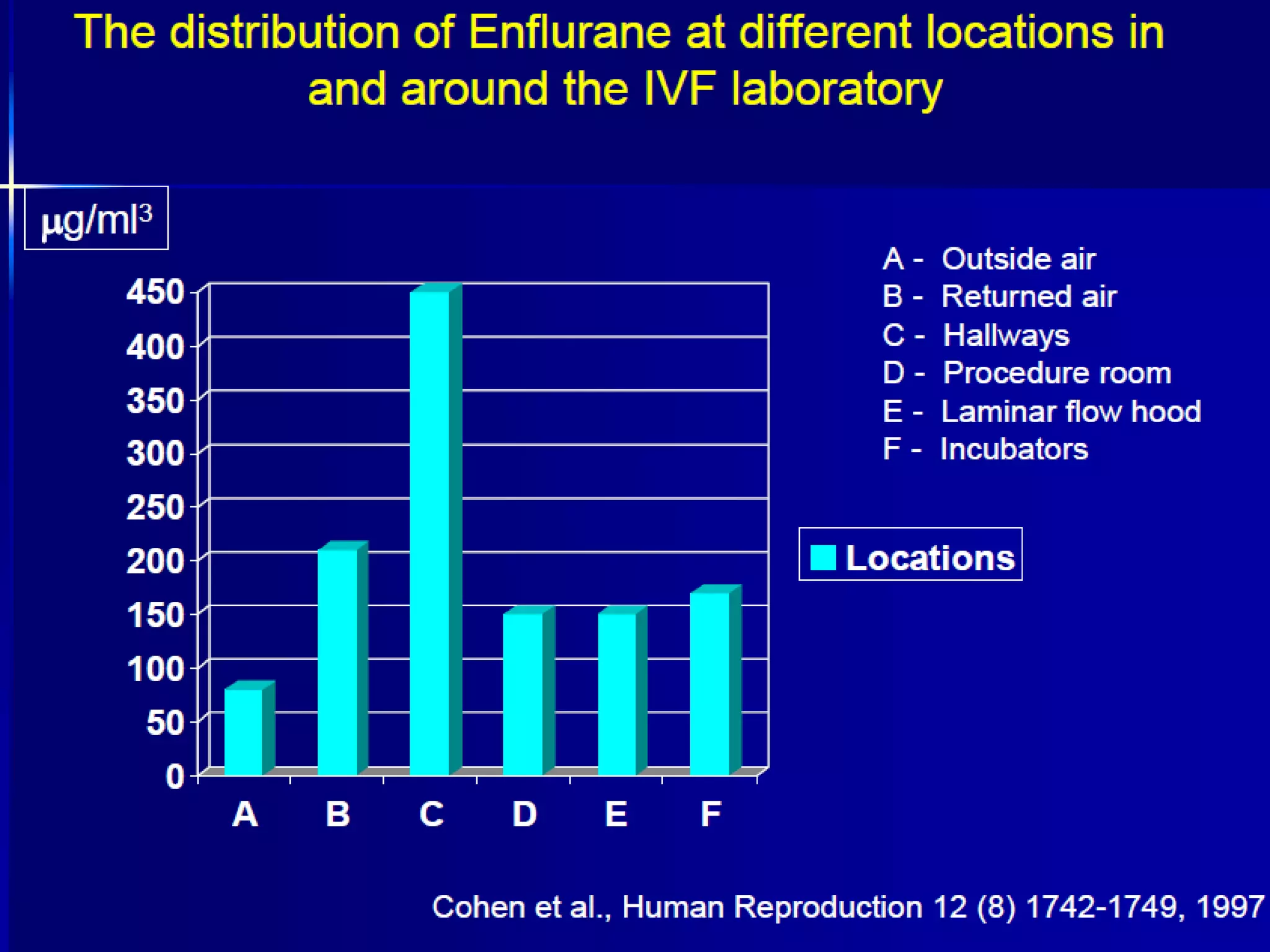
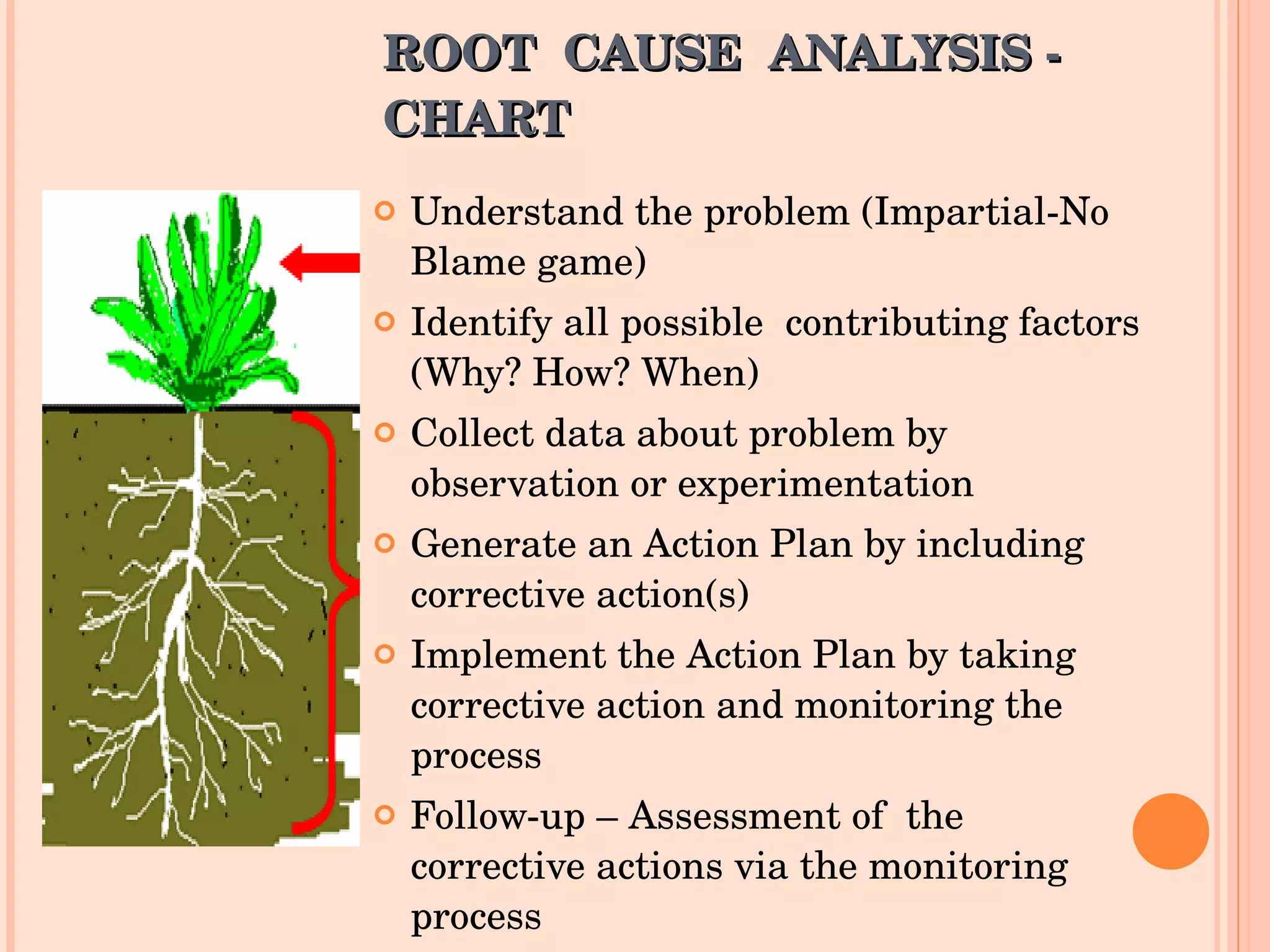
The document discusses the challenges and troubleshooting in IVF clinics, emphasizing the importance of quality control to prevent failures. It outlines various trouble areas such as equipment failures, human resource issues, and environmental factors that can lead to inconsistent success rates. The conclusion highlights the need for well-trained personnel, proper procedures, and systematic corrective actions to maintain high standards in IVF practices.