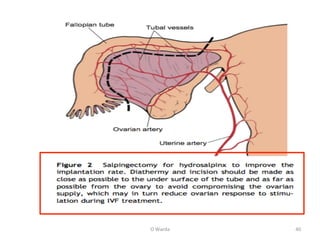
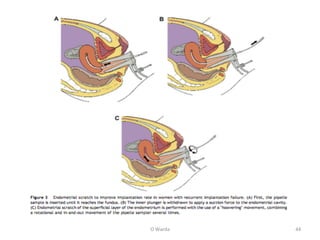
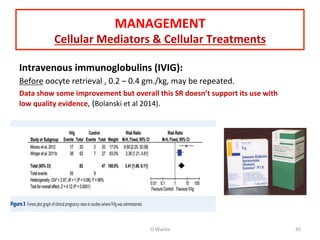
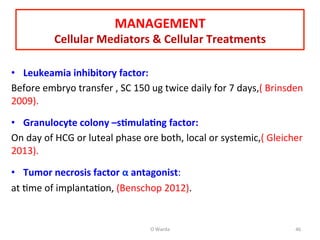
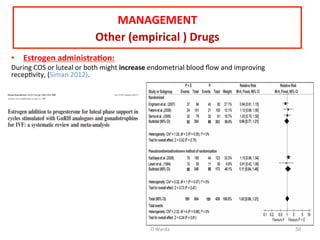
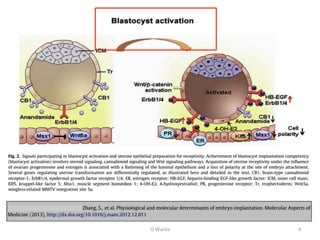
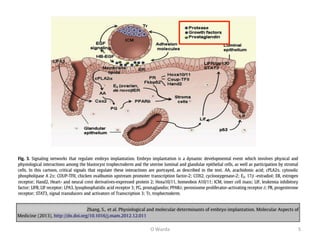
The document discusses repeated implantation failure (RIF) in the context of assisted conception, outlining the complex interaction between the receptive endometrium and healthy embryos. It details the definitions, causes, investigations, and management strategies for RIF, emphasizing the need for a multidisciplinary approach and the importance of various factors such as gamete quality, uterine pathology, and lifestyle modifications. Additionally, it highlights the significance of embryo selection techniques and the potential impact of treatments like assisted hatching and pre-implantation genetic diagnosis.
















![MANAGEMENT
LIFESTYLE
CHANGES
-‐
lifestyle
changes
which
could
improve
the
likelihood
of
treatment
success.
1.
Smoking;
stop
2.
Body
mass
index
:
should
be
[
between
≥
19kg/m2
and
≤
29kg/m2
]
3. Alcohol
consumpAon
should
be
decreased
or
stopped.
O
Warda
19](https://image.slidesharecdn.com/rif-190215144641/85/Repeated-implantation-failure-warda-full-19-320.jpg)