Key Concepts Unit A Chemistry 30
•Download as PPTX, PDF•
1 like•1,064 views
A structural isomer is a chemical formula that can take on different shapes. Saturated hydrocarbons have single bonds and cannot bond further, while unsaturated hydrocarbons have double or triple bonds and can bond with other compounds. Carboxylic acids have the basic structure of R-COOH and are used in esterification reactions with alcohols. Esterification is the reaction between an alcohol and a carboxylic acid to form an ester. Combustion reactions break down hydrocarbons and oxygen to produce water and carbon dioxide. Polymers are formed through polymerization reactions that link monomers together into chains.
Report
Share
Report
Share
Recommended
Other Organic Compounds

Substituted hydrocarbons are hydrocarbons where one or more hydrogen atoms have been replaced by other atoms or groups of atoms. Examples include halogen groups replacing hydrogen to form compounds like chloromethane, and alcohols formed when a hydroxyl group replaces a hydrogen like in ethanol. Carboxylic acids are formed when a carboxyl group replaces hydrogen, and amines are formed when an amino group replaces hydrogen like in methylamine. Amino acids are formed when both an amino group and a carboxyl group replace hydrogens on the same carbon, and proteins are formed from combinations of the 20 different amino acids bonding together.
Week 5. preparation of alcohols

1. Explain methods of various alcohol preparation methods by using chemical reactions
2. Identify Fermentation
05 macromolecules

1. Carbon is the backbone of biological molecules in living organisms and can form single, double, triple, or quadruple bonds.
2. Hydrocarbons like methane, ethane and ethene are molecules made of only carbon and hydrogen. Lipids, which do not form polymers, include fats and phospholipids.
3. Carbohydrates, proteins, nucleic acids and lipids are the four major classes of macromolecules that make up living things and carry out essential functions.
26sec3

The document discusses different types of carbonyl compounds including aldehydes, ketones, carboxylic acids, and esters. It covers their structures, naming conventions, properties, examples and how they are related through oxidation-reduction reactions. Aldehydes contain a carbonyl group bonded to at least one hydrogen, while ketones have the carbonyl group bonded to two carbon groups. Carboxylic acids contain both a carbonyl and hydroxyl group. Esters are derivatives of carboxylic acids where the hydroxyl is replaced by an alkoxy group from an alcohol.
Chem final

Alcohol is an organic compound characterized by the presence of a hydroxyl (OH) functional group. Drinking alcohol leads to negative health impacts like high blood pressure, cancer, and weakening the immune system. It also causes changes in mood and behavior. Alcohols are named based on the parent hydrocarbon, with "ol" replacing the final "e". Polyalcohols contain multiple OH groups and are named with prefixes like "di", "tri" to indicate the number of groups. Alcohols have higher boiling and melting points than similar hydrocarbons due to hydrogen bonding between OH groups. They are also able to dissolve both polar and nonpolar substances.
Hydrocarbons.kamran mammadli

The document discusses hydrocarbons, which are organic compounds made of only carbon and hydrogen. It defines hydrocarbons and explains that fossil fuels like petroleum, natural gas, and coal are important non-renewable energy sources and are the primary sources of hydrocarbons. The document also categorizes hydrocarbons into aliphatic hydrocarbons like alkanes, alkenes, and alkynes which differ based on their carbon bonding structure, and aromatic hydrocarbons which contain benzene rings. Real-world examples and uses of important hydrocarbons like crude oil and natural gas are provided.
Chemistry 2

Carbon forms the backbone of most biological molecules due to its ability to form diverse covalent bonds. It can link to other carbon atoms in chains or rings with varying lengths and branches. Functional groups including hydroxyl, carbonyl, carboxyl, amino, sulfhydryl, and phosphate give organic molecules distinctive reactivity and properties. These groups, along with isomerism, underlie the vast molecular diversity essential for life.
Organic chemistry

This document discusses esters in organic chemistry. It defines esters as compounds derived from acids where a hydroxyl group is replaced by an alkoxy group. Esters are usually derived from carboxylic acids and alcohols. The document covers ester nomenclature, structure, bonding, physical properties, preparation methods such as esterification, and common reactions like hydrolysis and reduction.
Recommended
Other Organic Compounds

Substituted hydrocarbons are hydrocarbons where one or more hydrogen atoms have been replaced by other atoms or groups of atoms. Examples include halogen groups replacing hydrogen to form compounds like chloromethane, and alcohols formed when a hydroxyl group replaces a hydrogen like in ethanol. Carboxylic acids are formed when a carboxyl group replaces hydrogen, and amines are formed when an amino group replaces hydrogen like in methylamine. Amino acids are formed when both an amino group and a carboxyl group replace hydrogens on the same carbon, and proteins are formed from combinations of the 20 different amino acids bonding together.
Week 5. preparation of alcohols

1. Explain methods of various alcohol preparation methods by using chemical reactions
2. Identify Fermentation
05 macromolecules

1. Carbon is the backbone of biological molecules in living organisms and can form single, double, triple, or quadruple bonds.
2. Hydrocarbons like methane, ethane and ethene are molecules made of only carbon and hydrogen. Lipids, which do not form polymers, include fats and phospholipids.
3. Carbohydrates, proteins, nucleic acids and lipids are the four major classes of macromolecules that make up living things and carry out essential functions.
26sec3

The document discusses different types of carbonyl compounds including aldehydes, ketones, carboxylic acids, and esters. It covers their structures, naming conventions, properties, examples and how they are related through oxidation-reduction reactions. Aldehydes contain a carbonyl group bonded to at least one hydrogen, while ketones have the carbonyl group bonded to two carbon groups. Carboxylic acids contain both a carbonyl and hydroxyl group. Esters are derivatives of carboxylic acids where the hydroxyl is replaced by an alkoxy group from an alcohol.
Chem final

Alcohol is an organic compound characterized by the presence of a hydroxyl (OH) functional group. Drinking alcohol leads to negative health impacts like high blood pressure, cancer, and weakening the immune system. It also causes changes in mood and behavior. Alcohols are named based on the parent hydrocarbon, with "ol" replacing the final "e". Polyalcohols contain multiple OH groups and are named with prefixes like "di", "tri" to indicate the number of groups. Alcohols have higher boiling and melting points than similar hydrocarbons due to hydrogen bonding between OH groups. They are also able to dissolve both polar and nonpolar substances.
Hydrocarbons.kamran mammadli

The document discusses hydrocarbons, which are organic compounds made of only carbon and hydrogen. It defines hydrocarbons and explains that fossil fuels like petroleum, natural gas, and coal are important non-renewable energy sources and are the primary sources of hydrocarbons. The document also categorizes hydrocarbons into aliphatic hydrocarbons like alkanes, alkenes, and alkynes which differ based on their carbon bonding structure, and aromatic hydrocarbons which contain benzene rings. Real-world examples and uses of important hydrocarbons like crude oil and natural gas are provided.
Chemistry 2

Carbon forms the backbone of most biological molecules due to its ability to form diverse covalent bonds. It can link to other carbon atoms in chains or rings with varying lengths and branches. Functional groups including hydroxyl, carbonyl, carboxyl, amino, sulfhydryl, and phosphate give organic molecules distinctive reactivity and properties. These groups, along with isomerism, underlie the vast molecular diversity essential for life.
Organic chemistry

This document discusses esters in organic chemistry. It defines esters as compounds derived from acids where a hydroxyl group is replaced by an alkoxy group. Esters are usually derived from carboxylic acids and alcohols. The document covers ester nomenclature, structure, bonding, physical properties, preparation methods such as esterification, and common reactions like hydrolysis and reduction.
Xi.carboxylic acids

This document provides an introduction to carboxylic acids. It defines carboxylic acids as containing a carboxyl group, which is a carbon double-bonded to an oxygen and single-bonded to a hydroxyl group. It discusses the structure, naming conventions, physical properties including acidity, solubility, and higher boiling points of carboxylic acids compared to similar molecules due to hydrogen bonding between molecules. Examples of uses of carboxylic acids in soaps, foods, pharmaceuticals, and other industries are also provided.
Isomers & Physical properties of alcohols

The slides contains Introduction to Isomerism Topic, classification of Isomers,Physical Properties of Alcohols. It contains examples and questions.
Esters, all properties

A general view of all chemical and physical properties of esters. Ass well as their daily use and structure ;)
Esters2015

Esters are organic compounds formed through the reaction of an alcohol with a carboxylic acid. They have many applications in medicine, biology, chemistry and industry. Esters can be naturally occurring or made in the laboratory through esterification reactions. The structure of esters is such that the main chain comes from the carboxylic acid and the alkyl group comes from the alcohol used. Esters have various smells and flavors that make them useful in producing fragrances, flavors and perfumes.
Esters

Esters are polar molecules that can participate in hydrogen bonding and dipole-dipole interactions. They are more polar than ethers but less polar than alcohols. Esters have lower melting and boiling points than the corresponding acids and amides. They undergo hydrolysis under basic conditions and are relatively resistant to reduction compared to ketones and aldehydes. Esters are commonly used as solvents, plasticizers, and food flavorings.
Esters

Ethyl ethanoate is an ester with a boiling point of 77.1°C. Esters have lower boiling points than carboxylic acids with the same number of carbon atoms due to their inability to form hydrogen bonds. Esters are soluble in water, with solubility decreasing with increasing chain length. The hydrolysis of esters can occur through acid or base mechanisms, involving nucleophilic attack of the hydroxide or hydronium ion on the carbonyl carbon and the formation of an intermediate. Ethyl ethanoate is used as a solvent and in artificial flavors, glues, and cigarette production.
Chemical properties of alcohols

Explain chemical properties of alcohols by various chemical reactions
Define and explain preparation of ethers from alcohols by using chemical equations
Carbohydrates paper

Carbohydrates play an important role in living organisms. They are classified as simple or complex sugars and serve as a primary energy source. The most common simple sugars are monosaccharides like glucose, fructose, and galactose. Complex carbohydrates are made of two or more linked monosaccharides and include disaccharides like sucrose and polysaccharides like starch and cellulose. Carbohydrates have many functions in biochemistry such as energy storage, structure in plants, and determining blood types in humans.
26 - Synthetic Polymers - Wade 7th

This document discusses synthetic polymers, including their classification, production methods, properties, and applications. It covers addition polymers formed through free radical, cationic, and anionic polymerization and condensation polymers like nylons, polyesters, polycarbonates, and polyurethanes. Key topics include the structures of polymers like polypropylene, polystyrene, and natural rubber; methods for controlling properties like crystallinity and plasticity; and major industrial polymers and their uses.
Unit 6 Organic Chemistry

Organic chemistry revision notes cover the formation of fossil fuels like oil from dead marine organisms under heat and pressure. Crude oil is separated into fractions like gasoline and kerosene through fractional distillation, and combustion produces pollution like carbon monoxide and nitrogen oxides. The energy released during combustion can be measured using a calorimeter. Homologous series are families of compounds with the same functional group and general formula that differ by CH2. Main series include alkanes, alkenes, and alcohols, which are named based on their carbon chain and functional group.
Alcohols (B.Pharm 1st Year 2nd Sem.)

Alcohols, Qualitative tests for Alcohols, Structure and uses of Ethyl alcohol, chlorobutanol, Cetosterylalcohol,Benzyl alcohol, Glycerol, Propylene glycol
Synthesis via enolates intro. and eaa

Enolates are intermediates formed during base catalysed reactions of active methylene compounds with electrophilc reagents/sustrates
Esters 

Esters are organic compounds that are derived from an acid and an alcohol. They contain a carbon atom bonded to an oxygen atom and another carbon or hydrogen atom. Common esters have characteristic smells and are found in flowers, fruits, and perfumes. Esters can be synthesized by heating an alcohol and acid together with an acid catalyst, in a reaction called Fischer esterification. They have various uses including in perfumes, solvents, plastics, surfactants and explosives.
Synthesis Via ENOLATES.pptx

Dr. Satish S. Kola discusses organic synthesis via enolates, specifically focusing on ethyl acetoacetate. Ethyl acetoacetate is prepared through the heating of ethyl acetate with sodium ethoxide followed by acidification. It undergoes Claisen condensation, forming an alcohol and β-keto ester. Ethyl acetoacetate displays properties of both the keto and enol forms. It is acidic, forming salts when treated with bases through the ionization of the methylene hydrogen. Its acidity is enhanced by inductive effects and resonance stabilization of the carbanion. Ethyl acetoacetate is used synthetically to produce carboxylic acids, ket
Functional Groups and Biochemistry 

The document defines several types of organic molecules including ketones, carboxylic acids, esters, and amines. It then discusses important biological organic molecules such as vitamins, carbohydrates, lipids, proteins, and nucleic acids. Vitamins can be water soluble or fat soluble and aid in various metabolic processes. Carbohydrates include polysaccharides and simple sugars. Lipids contain fatty acids and cholesterols and transport nutrients. Proteins are made of amino acids and are essential for cellular processes. Nucleic acids DNA and RNA contain genetic instructions and are essential for life. DNA has a double helix structure while RNA is single stranded and contains uracil instead of thymine.
Reaction Mechanisms of Esters

Biochemistry: Reaction Mechanisms of Esters.
Esters are among the most widespread of all naturally
occurring compounds. Many simple esters are pleasant-smelling liquids that are responsible for the fragrant odors of fruits and
flowers. For example, methyl butanoate is found in
pineapple oil, and isopentyl acetate is a constituent of
banana oil. The ester linkage is also present in animal fats and in
many biologically important molecule.
Topic- Green Chemistry.ppt

The document discusses green chemistry and its principles. It begins by describing some industrial accidents involving chemical releases that resulted in deaths and health issues. It then outlines the 12 principles of green chemistry as developed by Paul Anestas, including things like waste prevention and energy efficiency. Finally, it discusses how green chemistry aims to make chemical processes safer and less polluting through various methods like designing safer chemicals and alternative synthetic pathways.
Molecular Design

The document discusses ethers, which are organic compounds that contain an oxygen atom connected to two alkyl groups or aryl groups. It provides examples of common ethers such as diethyl ether, ethoxyethane, and methoxyflurane. It discusses the IUPAC naming conventions for ethers and their properties such as being flammable, soluble in water due to polarity, and having lower boiling points than other polar compounds. Uses of some ethers mentioned include as anesthetics, recreational drugs, lubricants, and industrial solvents.
alcohol

In chemistry, alcohols are organic compounds that carry at least one hydroxyl functional group bound to their aliphatic substructure. The term alcohol originally referred to the primary alcohol ethanol, which is used as a drug and is the main alcohol present in alcoholic beverages.
Chapter 21.1 : Functional Groups and Classes of Organic Compounds

This document defines functional groups and discusses various classes of organic compounds organized by their functional groups, including alcohols, alkyl halides, ethers, aldehydes, ketones, carboxylic acids, esters, and amines. It provides the general formulas and properties of each class and describes their naming conventions based on the functional group present. Key points covered include that functional groups determine properties, common uses of each class, and relationships between structure and reactivity.
chapter 15 HC,s.pptx

This document provides an overview of hydrocarbons including:
- Hydrocarbons are organic compounds made of carbon and hydrogen. Common sources include crude oil and natural gas.
- Saturated hydrocarbons like alkanes have single carbon-carbon bonds. Unsaturated hydrocarbons like alkenes have double or triple carbon-carbon bonds.
- Alkanes undergo combustion and substitution reactions. Incomplete combustion of hydrocarbons leads to air pollution. Catalytic converters reduce harmful emissions.
Chemical properties of hydrocarbons

1. The document discusses the chemical properties of hydrocarbons including alkanes, alkenes, and alkynes. It describes different types of chemical reactions such as combustion, addition, substitution, bromination, hydrogenation, and hydration.
2. Specific reactions are discussed including the combustion of hydrocarbons producing carbon dioxide and water. Addition reactions like bromination, hydrogenation, and hydration that involve adding atoms to alkenes and alkynes are also covered.
3. Examples are provided to illustrate combustion reactions, bromination of cyclohexene, hydrogenation of sunflower oil to produce solid fats, and the hydration of symmetrical and asymmetrical alkenes following Markovnik
More Related Content
What's hot
Xi.carboxylic acids

This document provides an introduction to carboxylic acids. It defines carboxylic acids as containing a carboxyl group, which is a carbon double-bonded to an oxygen and single-bonded to a hydroxyl group. It discusses the structure, naming conventions, physical properties including acidity, solubility, and higher boiling points of carboxylic acids compared to similar molecules due to hydrogen bonding between molecules. Examples of uses of carboxylic acids in soaps, foods, pharmaceuticals, and other industries are also provided.
Isomers & Physical properties of alcohols

The slides contains Introduction to Isomerism Topic, classification of Isomers,Physical Properties of Alcohols. It contains examples and questions.
Esters, all properties

A general view of all chemical and physical properties of esters. Ass well as their daily use and structure ;)
Esters2015

Esters are organic compounds formed through the reaction of an alcohol with a carboxylic acid. They have many applications in medicine, biology, chemistry and industry. Esters can be naturally occurring or made in the laboratory through esterification reactions. The structure of esters is such that the main chain comes from the carboxylic acid and the alkyl group comes from the alcohol used. Esters have various smells and flavors that make them useful in producing fragrances, flavors and perfumes.
Esters

Esters are polar molecules that can participate in hydrogen bonding and dipole-dipole interactions. They are more polar than ethers but less polar than alcohols. Esters have lower melting and boiling points than the corresponding acids and amides. They undergo hydrolysis under basic conditions and are relatively resistant to reduction compared to ketones and aldehydes. Esters are commonly used as solvents, plasticizers, and food flavorings.
Esters

Ethyl ethanoate is an ester with a boiling point of 77.1°C. Esters have lower boiling points than carboxylic acids with the same number of carbon atoms due to their inability to form hydrogen bonds. Esters are soluble in water, with solubility decreasing with increasing chain length. The hydrolysis of esters can occur through acid or base mechanisms, involving nucleophilic attack of the hydroxide or hydronium ion on the carbonyl carbon and the formation of an intermediate. Ethyl ethanoate is used as a solvent and in artificial flavors, glues, and cigarette production.
Chemical properties of alcohols

Explain chemical properties of alcohols by various chemical reactions
Define and explain preparation of ethers from alcohols by using chemical equations
Carbohydrates paper

Carbohydrates play an important role in living organisms. They are classified as simple or complex sugars and serve as a primary energy source. The most common simple sugars are monosaccharides like glucose, fructose, and galactose. Complex carbohydrates are made of two or more linked monosaccharides and include disaccharides like sucrose and polysaccharides like starch and cellulose. Carbohydrates have many functions in biochemistry such as energy storage, structure in plants, and determining blood types in humans.
26 - Synthetic Polymers - Wade 7th

This document discusses synthetic polymers, including their classification, production methods, properties, and applications. It covers addition polymers formed through free radical, cationic, and anionic polymerization and condensation polymers like nylons, polyesters, polycarbonates, and polyurethanes. Key topics include the structures of polymers like polypropylene, polystyrene, and natural rubber; methods for controlling properties like crystallinity and plasticity; and major industrial polymers and their uses.
Unit 6 Organic Chemistry

Organic chemistry revision notes cover the formation of fossil fuels like oil from dead marine organisms under heat and pressure. Crude oil is separated into fractions like gasoline and kerosene through fractional distillation, and combustion produces pollution like carbon monoxide and nitrogen oxides. The energy released during combustion can be measured using a calorimeter. Homologous series are families of compounds with the same functional group and general formula that differ by CH2. Main series include alkanes, alkenes, and alcohols, which are named based on their carbon chain and functional group.
Alcohols (B.Pharm 1st Year 2nd Sem.)

Alcohols, Qualitative tests for Alcohols, Structure and uses of Ethyl alcohol, chlorobutanol, Cetosterylalcohol,Benzyl alcohol, Glycerol, Propylene glycol
Synthesis via enolates intro. and eaa

Enolates are intermediates formed during base catalysed reactions of active methylene compounds with electrophilc reagents/sustrates
Esters 

Esters are organic compounds that are derived from an acid and an alcohol. They contain a carbon atom bonded to an oxygen atom and another carbon or hydrogen atom. Common esters have characteristic smells and are found in flowers, fruits, and perfumes. Esters can be synthesized by heating an alcohol and acid together with an acid catalyst, in a reaction called Fischer esterification. They have various uses including in perfumes, solvents, plastics, surfactants and explosives.
Synthesis Via ENOLATES.pptx

Dr. Satish S. Kola discusses organic synthesis via enolates, specifically focusing on ethyl acetoacetate. Ethyl acetoacetate is prepared through the heating of ethyl acetate with sodium ethoxide followed by acidification. It undergoes Claisen condensation, forming an alcohol and β-keto ester. Ethyl acetoacetate displays properties of both the keto and enol forms. It is acidic, forming salts when treated with bases through the ionization of the methylene hydrogen. Its acidity is enhanced by inductive effects and resonance stabilization of the carbanion. Ethyl acetoacetate is used synthetically to produce carboxylic acids, ket
Functional Groups and Biochemistry 

The document defines several types of organic molecules including ketones, carboxylic acids, esters, and amines. It then discusses important biological organic molecules such as vitamins, carbohydrates, lipids, proteins, and nucleic acids. Vitamins can be water soluble or fat soluble and aid in various metabolic processes. Carbohydrates include polysaccharides and simple sugars. Lipids contain fatty acids and cholesterols and transport nutrients. Proteins are made of amino acids and are essential for cellular processes. Nucleic acids DNA and RNA contain genetic instructions and are essential for life. DNA has a double helix structure while RNA is single stranded and contains uracil instead of thymine.
Reaction Mechanisms of Esters

Biochemistry: Reaction Mechanisms of Esters.
Esters are among the most widespread of all naturally
occurring compounds. Many simple esters are pleasant-smelling liquids that are responsible for the fragrant odors of fruits and
flowers. For example, methyl butanoate is found in
pineapple oil, and isopentyl acetate is a constituent of
banana oil. The ester linkage is also present in animal fats and in
many biologically important molecule.
Topic- Green Chemistry.ppt

The document discusses green chemistry and its principles. It begins by describing some industrial accidents involving chemical releases that resulted in deaths and health issues. It then outlines the 12 principles of green chemistry as developed by Paul Anestas, including things like waste prevention and energy efficiency. Finally, it discusses how green chemistry aims to make chemical processes safer and less polluting through various methods like designing safer chemicals and alternative synthetic pathways.
Molecular Design

The document discusses ethers, which are organic compounds that contain an oxygen atom connected to two alkyl groups or aryl groups. It provides examples of common ethers such as diethyl ether, ethoxyethane, and methoxyflurane. It discusses the IUPAC naming conventions for ethers and their properties such as being flammable, soluble in water due to polarity, and having lower boiling points than other polar compounds. Uses of some ethers mentioned include as anesthetics, recreational drugs, lubricants, and industrial solvents.
alcohol

In chemistry, alcohols are organic compounds that carry at least one hydroxyl functional group bound to their aliphatic substructure. The term alcohol originally referred to the primary alcohol ethanol, which is used as a drug and is the main alcohol present in alcoholic beverages.
Chapter 21.1 : Functional Groups and Classes of Organic Compounds

This document defines functional groups and discusses various classes of organic compounds organized by their functional groups, including alcohols, alkyl halides, ethers, aldehydes, ketones, carboxylic acids, esters, and amines. It provides the general formulas and properties of each class and describes their naming conventions based on the functional group present. Key points covered include that functional groups determine properties, common uses of each class, and relationships between structure and reactivity.
What's hot (20)
Chapter 21.1 : Functional Groups and Classes of Organic Compounds

Chapter 21.1 : Functional Groups and Classes of Organic Compounds
Similar to Key Concepts Unit A Chemistry 30
chapter 15 HC,s.pptx

This document provides an overview of hydrocarbons including:
- Hydrocarbons are organic compounds made of carbon and hydrogen. Common sources include crude oil and natural gas.
- Saturated hydrocarbons like alkanes have single carbon-carbon bonds. Unsaturated hydrocarbons like alkenes have double or triple carbon-carbon bonds.
- Alkanes undergo combustion and substitution reactions. Incomplete combustion of hydrocarbons leads to air pollution. Catalytic converters reduce harmful emissions.
Chemical properties of hydrocarbons

1. The document discusses the chemical properties of hydrocarbons including alkanes, alkenes, and alkynes. It describes different types of chemical reactions such as combustion, addition, substitution, bromination, hydrogenation, and hydration.
2. Specific reactions are discussed including the combustion of hydrocarbons producing carbon dioxide and water. Addition reactions like bromination, hydrogenation, and hydration that involve adding atoms to alkenes and alkynes are also covered.
3. Examples are provided to illustrate combustion reactions, bromination of cyclohexene, hydrogenation of sunflower oil to produce solid fats, and the hydration of symmetrical and asymmetrical alkenes following Markovnik
Organic Chemistry.ppt

This document provides an overview of organic chemistry concepts. It defines organic compounds as hydrocarbons and related compounds containing carbon. Carbon atoms can bond together in chains, allowing for a large number of organic compounds. Saturated compounds have single carbon bonds while unsaturated have double or triple bonds. Common hydrocarbons include alkanes (single bonds), alkenes (double bonds), and alkynes (triple bonds). The document also discusses the structure and reactions of important organic compound classes like alkenes, alcohols, acids, and polymers.
CLASS X CHEMSITRY

1. Carbon forms covalent bonds by sharing electrons, allowing it to form large molecules through catenation. It can form single, double, and triple bonds.
2. Homologous series are groups of compounds with the same functional group substituting for hydrogen in a carbon chain. Saturated hydrocarbons burn with a clean flame while unsaturated ones burn with a yellow flame.
3. Soaps form micelles that help dissolve dirt in water, but hard water prevents this and causes scum. Detergents avoid this issue and work in hard water.
Types of organic_chemical_reactions_1

Organic chemical reactions can be categorized as substitution, addition, condensation/elimination, hydrolysis, oxidation, combustion, or acid/base reactions. Substitution reactions involve replacing one atom in an organic molecule with another atom. Addition reactions add new atoms or groups to unsaturated organic molecules containing double or triple bonds. Condensation and elimination reactions combine or dehydrate organic compounds containing functional groups like alcohols or carboxylic acids to form new bonds and products.
Organic molecules

Organic chemistry deals with carbon-containing compounds called organic molecules. Carbon atoms can bond to many other atoms, often forming long chain structures. Organic compounds can be represented using molecular formulas or structural formulas. They contain functional groups that give them certain chemical properties and can be classified based on these groups. Organic reactions include addition, elimination, and substitution. Polymers are large molecules formed by combining repeating structural units (monomers) and include both natural and synthetic varieties.
Chapter-Organic-Chemistry-class-10-important-Questions.pdf

This document provides answers to questions about organic chemistry concepts. It defines key terms like catenation, isomerism, alkyl groups, functional groups, alkanes, and alkyl radicals. It also lists major commercial sources of alkanes, describes isomers and functional groups for several compounds, and provides structural formulas for alkanes and alkynes. The document aims to clarify fundamental organic chemistry concepts and distinguish between related terms.
Review on Organic Chemical Reactions (1).ppt

I apologize, upon further review I do not have enough information to determine the products of the reaction you asked about. The document provided describes various types of organic chemical reactions but does not include any specific reactions to analyze.
Práctica 4 reacciones típicas de química orgánica

This document provides instructions for a virtual laboratory simulation. It explains that users can select different buttons and interactive elements throughout the virtual lab to access theoretical background information, laboratory materials and reagents, and to perform simulated experiments. The document provides examples of interactive elements that can be clicked, such as books to access theoretical bases, drawers to view laboratory instruments, and flasks to see reagents. It also explains that all rooms in the virtual lab contain a button to return to the main room and buttons to move forward or backward within the content. The goal is for users to feel like they are conducting a real laboratory experiment by navigating between the different sections and interactive elements.
Organic chemistry

Crude oil is formed from the remains of ancient organisms over millions of years. It is separated into fractions by fractional distillation based on boiling points. The lighter fractions such as liquefied petroleum gas and gasoline are used as fuels, while heavier fractions such as lubricating oils and bitumen have other industrial uses. "Cracking" converts heavier fractions into smaller, more useful molecules like alkenes needed for plastics production. Alkenes polymerize to form large plastic polymer molecules. However, plastics cause waste and pollution problems as they do not biodegrade.
Alcohols:Organic Chemistry MANIK

2. Chemistry of Aliphatic Compounds: Introduction, methods of preparation, physical and chemical properties and pharmaceutical applications of alcohols, aldehydes, ketones, hydrocarbons, ester, ethers, amines, amides and carboxylic acids.
organic (alkanes).ppt

This document covers organic chemistry concepts including alkanes. It begins with an introduction to organic chemistry, noting that it involves carbon-containing compounds that often also contain hydrogen, oxygen, or other elements. Most organic compounds occur naturally, while many others are made artificially. Section 3 specifically discusses alkanes, the simplest hydrocarbons. It defines alkanes as saturated hydrocarbons that form a homologous series with the general formula CnH2n+2. Examples of alkanes and their properties like physical state are provided. Combustion reactions of alkanes and substitution reactions are also summarized.
Cmc chapter 22

The document is a chapter menu for organic chemistry covering substituted hydrocarbons and their reactions. It outlines 5 main sections that discuss alkyl and aryl halides, alcohols/ethers/amines, carbonyl compounds, other organic reactions, and polymers. Each section defines functional groups, draws structures, and discusses properties and reactions for different compound classes.
c15hydrocarbons-161113170003.pptx

This document outlines learning outcomes and content about hydrocarbons. It discusses the bonding ability of carbon and different types of organic compounds. It introduces the alkane and alkene homologous series, including their structures, properties, and reactions. Specific topics covered include structural formulas of alkanes and alkenes, combustion reactions, substitution reactions with halogens, and uses of alkanes and alkenes. Polyunsaturated foods are also mentioned.
Organic chemistry

This document provides an overview of organic chemistry concepts, including hydrocarbons, organic compounds, and their derivatives. It discusses the structure and properties of carbon-based molecules like alkanes, alkenes, alkynes, aromatics, and polymers. It also covers the four major macromolecules that make up living organisms: proteins, carbohydrates, lipids, and nucleic acids. Finally, it briefly mentions how petroleum and coal can be used to produce synthetic polymers.
Application of organic chemistry ok1294986436

Organic chemistry studies hydrocarbons like those found in crude oil. Hydrocarbons are compounds made of only carbon and hydrogen. They can have single, double, or triple bonds between carbon atoms. Crude oil is pumped from the ground as a complex mixture of hydrocarbons and then separated through distillation into usable products like gasoline and kerosene. Functional groups can be added to hydrocarbons through reactions, producing compounds like alcohols, ethers, aldehydes and ketones.
Introductiontoorganicchemistry 090518040648 Phpapp02

1. The document discusses organic chemistry nomenclature and isomerism, as well as petroleum and several classes of organic compounds including alkanes, alkenes, haloalkanes, and alcohols.
2. Naming allows identification of isomers, while petroleum provides many hydrocarbons as fuel and feedstocks, but combustion can cause pollution.
3. Alkenes are more reactive than alkanes due to weaker double bonds, and haloalkanes react via nucleophilic substitution on their polar bonds. The reactivity of alcohols depends on whether they are primary, secondary, or tertiary.
alcohol phenols ethers class 12 notes pdf

1) Alcohols and phenols are formed when a hydrogen atom in a hydrocarbon is replaced by an -OH group, attached to either a sp3 carbon (alcohols) or sp2 carbon (phenols).
2) Ethers are formed when a hydrogen atom in a hydrocarbon is replaced by an alkoxy or aryloxy group.
3) Alcohols and phenols can be classified based on the number of hydroxyl groups as mono-, di-, tri-, or polyhydric compounds. Ethers can be classified as symmetrical or unsymmetrical.
Organic chemistry

This document provides an overview of organic chemistry concepts, including:
1) Hydrocarbons are compounds made of only carbon and hydrogen, and can have single, double, or triple bonds between carbons.
2) Organic compounds derived from hydrocarbons include alcohols, ethers, aldehydes, ketones, acids, and esters which are formed by replacing hydrogen with other functional groups.
3) Macromolecules like proteins, carbohydrates, and nucleic acids are essential to life and control cellular activities and growth.
alcohols phenols ethers-.pdf

This document summarizes key information about alcohols and phenols. It defines alcohols as compounds containing hydroxyl groups bonded to carbon atoms, and phenols as having hydroxyl groups bonded to aromatic carbons. Alcohols and phenols are classified based on the number and position of hydroxyl groups. The document also discusses the structures, properties, nomenclature and reactions of alcohols, phenols, and ethers.
Similar to Key Concepts Unit A Chemistry 30 (20)
Chapter-Organic-Chemistry-class-10-important-Questions.pdf

Chapter-Organic-Chemistry-class-10-important-Questions.pdf
Introductiontoorganicchemistry 090518040648 Phpapp02

Introductiontoorganicchemistry 090518040648 Phpapp02
Recently uploaded
National Security Agency - NSA mobile device best practices

Threats to mobile devices are more prevalent and increasing in scope and complexity. Users of mobile devices desire to take full advantage of the features
available on those devices, but many of the features provide convenience and capability but sacrifice security. This best practices guide outlines steps the users can take to better protect personal devices and information.
Salesforce Integration for Bonterra Impact Management (fka Social Solutions A...

Sidekick Solutions uses Bonterra Impact Management (fka Social Solutions Apricot) and automation solutions to integrate data for business workflows.
We believe integration and automation are essential to user experience and the promise of efficient work through technology. Automation is the critical ingredient to realizing that full vision. We develop integration products and services for Bonterra Case Management software to support the deployment of automations for a variety of use cases.
This video focuses on integration of Salesforce with Bonterra Impact Management.
Interested in deploying an integration with Salesforce for Bonterra Impact Management? Contact us at sales@sidekicksolutionsllc.com to discuss next steps.
Unlock the Future of Search with MongoDB Atlas_ Vector Search Unleashed.pdf

Discover how MongoDB Atlas and vector search technology can revolutionize your application's search capabilities. This comprehensive presentation covers:
* What is Vector Search?
* Importance and benefits of vector search
* Practical use cases across various industries
* Step-by-step implementation guide
* Live demos with code snippets
* Enhancing LLM capabilities with vector search
* Best practices and optimization strategies
Perfect for developers, AI enthusiasts, and tech leaders. Learn how to leverage MongoDB Atlas to deliver highly relevant, context-aware search results, transforming your data retrieval process. Stay ahead in tech innovation and maximize the potential of your applications.
#MongoDB #VectorSearch #AI #SemanticSearch #TechInnovation #DataScience #LLM #MachineLearning #SearchTechnology
How to Get CNIC Information System with Paksim Ga.pptx

Pakdata Cf is a groundbreaking system designed to streamline and facilitate access to CNIC information. This innovative platform leverages advanced technology to provide users with efficient and secure access to their CNIC details.
AI 101: An Introduction to the Basics and Impact of Artificial Intelligence

Imagine a world where machines not only perform tasks but also learn, adapt, and make decisions. This is the promise of Artificial Intelligence (AI), a technology that's not just enhancing our lives but revolutionizing entire industries.
Main news related to the CCS TSI 2023 (2023/1695)

An English 🇬🇧 translation of a presentation to the speech I gave about the main changes brought by CCS TSI 2023 at the biggest Czech conference on Communications and signalling systems on Railways, which was held in Clarion Hotel Olomouc from 7th to 9th November 2023 (konferenceszt.cz). Attended by around 500 participants and 200 on-line followers.
The original Czech 🇨🇿 version of the presentation can be found here: https://www.slideshare.net/slideshow/hlavni-novinky-souvisejici-s-ccs-tsi-2023-2023-1695/269688092 .
The videorecording (in Czech) from the presentation is available here: https://youtu.be/WzjJWm4IyPk?si=SImb06tuXGb30BEH .
Digital Marketing Trends in 2024 | Guide for Staying Ahead

https://www.wask.co/ebooks/digital-marketing-trends-in-2024
Feeling lost in the digital marketing whirlwind of 2024? Technology is changing, consumer habits are evolving, and staying ahead of the curve feels like a never-ending pursuit. This e-book is your compass. Dive into actionable insights to handle the complexities of modern marketing. From hyper-personalization to the power of user-generated content, learn how to build long-term relationships with your audience and unlock the secrets to success in the ever-shifting digital landscape.
Project Management Semester Long Project - Acuity

Acuity is an innovative learning app designed to transform the way you engage with knowledge. Powered by AI technology, Acuity takes complex topics and distills them into concise, interactive summaries that are easy to read & understand. Whether you're exploring the depths of quantum mechanics or seeking insight into historical events, Acuity provides the key information you need without the burden of lengthy texts.
Cosa hanno in comune un mattoncino Lego e la backdoor XZ?

ABSTRACT: A prima vista, un mattoncino Lego e la backdoor XZ potrebbero avere in comune il fatto di essere entrambi blocchi di costruzione, o dipendenze di progetti creativi e software. La realtà è che un mattoncino Lego e il caso della backdoor XZ hanno molto di più di tutto ciò in comune.
Partecipate alla presentazione per immergervi in una storia di interoperabilità, standard e formati aperti, per poi discutere del ruolo importante che i contributori hanno in una comunità open source sostenibile.
BIO: Sostenitrice del software libero e dei formati standard e aperti. È stata un membro attivo dei progetti Fedora e openSUSE e ha co-fondato l'Associazione LibreItalia dove è stata coinvolta in diversi eventi, migrazioni e formazione relativi a LibreOffice. In precedenza ha lavorato a migrazioni e corsi di formazione su LibreOffice per diverse amministrazioni pubbliche e privati. Da gennaio 2020 lavora in SUSE come Software Release Engineer per Uyuni e SUSE Manager e quando non segue la sua passione per i computer e per Geeko coltiva la sua curiosità per l'astronomia (da cui deriva il suo nickname deneb_alpha).
みなさんこんにちはこれ何文字まで入るの?40文字以下不可とか本当に意味わからないけどこれ限界文字数書いてないからマジでやばい文字数いけるんじゃないの?えこ...

ここ3000字までしか入らないけどタイトルの方がたくさん文字入ると思います。
Let's Integrate MuleSoft RPA, COMPOSER, APM with AWS IDP along with Slack

Discover the seamless integration of RPA (Robotic Process Automation), COMPOSER, and APM with AWS IDP enhanced with Slack notifications. Explore how these technologies converge to streamline workflows, optimize performance, and ensure secure access, all while leveraging the power of AWS IDP and real-time communication via Slack notifications.
20240607 QFM018 Elixir Reading List May 2024

Everything I found interesting about the Elixir programming ecosystem in May 2024
Choosing The Best AWS Service For Your Website + API.pptx

Have you ever been confused by the myriad of choices offered by AWS for hosting a website or an API?
Lambda, Elastic Beanstalk, Lightsail, Amplify, S3 (and more!) can each host websites + APIs. But which one should we choose?
Which one is cheapest? Which one is fastest? Which one will scale to meet our needs?
Join me in this session as we dive into each AWS hosting service to determine which one is best for your scenario and explain why!
“Building and Scaling AI Applications with the Nx AI Manager,” a Presentation...

“Building and Scaling AI Applications with the Nx AI Manager,” a Presentation...Edge AI and Vision Alliance
For the full video of this presentation, please visit: https://www.edge-ai-vision.com/2024/06/building-and-scaling-ai-applications-with-the-nx-ai-manager-a-presentation-from-network-optix/
Robin van Emden, Senior Director of Data Science at Network Optix, presents the “Building and Scaling AI Applications with the Nx AI Manager,” tutorial at the May 2024 Embedded Vision Summit.
In this presentation, van Emden covers the basics of scaling edge AI solutions using the Nx tool kit. He emphasizes the process of developing AI models and deploying them globally. He also showcases the conversion of AI models and the creation of effective edge AI pipelines, with a focus on pre-processing, model conversion, selecting the appropriate inference engine for the target hardware and post-processing.
van Emden shows how Nx can simplify the developer’s life and facilitate a rapid transition from concept to production-ready applications.He provides valuable insights into developing scalable and efficient edge AI solutions, with a strong focus on practical implementation.Monitoring and Managing Anomaly Detection on OpenShift.pdf

Monitoring and Managing Anomaly Detection on OpenShift
Overview
Dive into the world of anomaly detection on edge devices with our comprehensive hands-on tutorial. This SlideShare presentation will guide you through the entire process, from data collection and model training to edge deployment and real-time monitoring. Perfect for those looking to implement robust anomaly detection systems on resource-constrained IoT/edge devices.
Key Topics Covered
1. Introduction to Anomaly Detection
- Understand the fundamentals of anomaly detection and its importance in identifying unusual behavior or failures in systems.
2. Understanding Edge (IoT)
- Learn about edge computing and IoT, and how they enable real-time data processing and decision-making at the source.
3. What is ArgoCD?
- Discover ArgoCD, a declarative, GitOps continuous delivery tool for Kubernetes, and its role in deploying applications on edge devices.
4. Deployment Using ArgoCD for Edge Devices
- Step-by-step guide on deploying anomaly detection models on edge devices using ArgoCD.
5. Introduction to Apache Kafka and S3
- Explore Apache Kafka for real-time data streaming and Amazon S3 for scalable storage solutions.
6. Viewing Kafka Messages in the Data Lake
- Learn how to view and analyze Kafka messages stored in a data lake for better insights.
7. What is Prometheus?
- Get to know Prometheus, an open-source monitoring and alerting toolkit, and its application in monitoring edge devices.
8. Monitoring Application Metrics with Prometheus
- Detailed instructions on setting up Prometheus to monitor the performance and health of your anomaly detection system.
9. What is Camel K?
- Introduction to Camel K, a lightweight integration framework built on Apache Camel, designed for Kubernetes.
10. Configuring Camel K Integrations for Data Pipelines
- Learn how to configure Camel K for seamless data pipeline integrations in your anomaly detection workflow.
11. What is a Jupyter Notebook?
- Overview of Jupyter Notebooks, an open-source web application for creating and sharing documents with live code, equations, visualizations, and narrative text.
12. Jupyter Notebooks with Code Examples
- Hands-on examples and code snippets in Jupyter Notebooks to help you implement and test anomaly detection models.
Ocean lotus Threat actors project by John Sitima 2024 (1).pptx

Ocean Lotus cyber threat actors represent a sophisticated, persistent, and politically motivated group that poses a significant risk to organizations and individuals in the Southeast Asian region. Their continuous evolution and adaptability underscore the need for robust cybersecurity measures and international cooperation to identify and mitigate the threats posed by such advanced persistent threat groups.
Presentation of the OECD Artificial Intelligence Review of Germany

Consult the full report at https://www.oecd.org/digital/oecd-artificial-intelligence-review-of-germany-609808d6-en.htm
Recently uploaded (20)
National Security Agency - NSA mobile device best practices

National Security Agency - NSA mobile device best practices
Salesforce Integration for Bonterra Impact Management (fka Social Solutions A...

Salesforce Integration for Bonterra Impact Management (fka Social Solutions A...
Unlock the Future of Search with MongoDB Atlas_ Vector Search Unleashed.pdf

Unlock the Future of Search with MongoDB Atlas_ Vector Search Unleashed.pdf
How to Get CNIC Information System with Paksim Ga.pptx

How to Get CNIC Information System with Paksim Ga.pptx
AI 101: An Introduction to the Basics and Impact of Artificial Intelligence

AI 101: An Introduction to the Basics and Impact of Artificial Intelligence
Digital Marketing Trends in 2024 | Guide for Staying Ahead

Digital Marketing Trends in 2024 | Guide for Staying Ahead
Cosa hanno in comune un mattoncino Lego e la backdoor XZ?

Cosa hanno in comune un mattoncino Lego e la backdoor XZ?
みなさんこんにちはこれ何文字まで入るの?40文字以下不可とか本当に意味わからないけどこれ限界文字数書いてないからマジでやばい文字数いけるんじゃないの?えこ...

みなさんこんにちはこれ何文字まで入るの?40文字以下不可とか本当に意味わからないけどこれ限界文字数書いてないからマジでやばい文字数いけるんじゃないの?えこ...
Let's Integrate MuleSoft RPA, COMPOSER, APM with AWS IDP along with Slack

Let's Integrate MuleSoft RPA, COMPOSER, APM with AWS IDP along with Slack
Choosing The Best AWS Service For Your Website + API.pptx

Choosing The Best AWS Service For Your Website + API.pptx
“Building and Scaling AI Applications with the Nx AI Manager,” a Presentation...

“Building and Scaling AI Applications with the Nx AI Manager,” a Presentation...
Monitoring and Managing Anomaly Detection on OpenShift.pdf

Monitoring and Managing Anomaly Detection on OpenShift.pdf
Ocean lotus Threat actors project by John Sitima 2024 (1).pptx

Ocean lotus Threat actors project by John Sitima 2024 (1).pptx
Presentation of the OECD Artificial Intelligence Review of Germany

Presentation of the OECD Artificial Intelligence Review of Germany
Key Concepts Unit A Chemistry 30
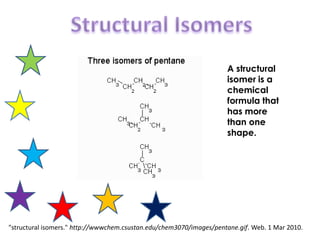
- 1. Structural Isomers A structural isomer is a chemical formula that has more than one shape. "structural isomers." http://wwwchem.csustan.edu/chem3070/images/pentane.gif. Web. 1 Mar 2010.
- 2. Saturated/Unsaturated Hydrocarbons Saturated hydrocarbons have single bonds and cannot bond with compounds anymore. Unsaturated hydrocarbons have double or triple bonds and can bond with compounds.
- 3. Halogenated Hydrocarbons Are produced When a Hydrocarbon has one or more hydrogen group replaced with a Halogen(group 17) Can help create a saturated hydrocarbon by breaking double or triple bonds. http://www.youtube.com/watch?v=1qGPWdm2MlI (watch to 1:50)
- 4. Carboxylic Acids The Basic Structure is R-COOH (R can be replaced by a hydrogen) They are used in an esterfication reaction with an alcohol When naming you change the ending to “oic” acid (methane would be methanoic acid) They have hydrogen bonds increasing their boiling points.
- 5. Structuralformulas The structural formula of a chemical compound is a graphical representation showing how the atoms are arranged. This is a structural formula where atoms are arranged forming a compound Structural formulas rock my world!!! http://en.wikipedia.org/wiki/Structural_formula http://www.1-formula.com/upl/Image/800px-Acetone-structural.png
- 6. Esterification http://en.wikipedia.org/wiki/Esterification This is an Esterification reaction I love Esterification reactions because they smell good Esterification is the general name for a chemical reaction in which two reactants (typically an alcohol and a carboxylic acid) form an ester as the reaction product.
- 7. Addition Reaction An addition reaction is the reaction between two or more unsaturated hydrocarbons to produce one large molecule.
- 8. Substitution Reaction When a carbon-hydrogen bond breaking occurs in an alkane or aromatic organic reaction An example is the reaction in which the chlorine atom in chloromethane is displaced by the hydroxide ion to form methanol CH3Cl + -OH CH3OH + Cl-
- 9. Addition and substitution reaction Video http://www.youtube.com/watch?v=nVDUB4mWJ8M
- 11. A Complete Combustion reaction is when the reactant will burn in oxygen producing a limited number of products.
- 12. An incomplete combustion reaction is when there isn’t enough oxygen to react with.C?H? + O2 CO2 + H2O C?H? + O2 CO + H2O "combustion reactions.". http://upload.wikimedia.org/wikipedia/commons/3/3c/Et_baal.jpg. Web. 1 Mar 2010
- 13. Monomers monomer - a simple compound whose molecules can join together to form polymers.
- 14. Polymers Polymers are created when two or more monomers are linked together in a chain like pattern
- 15. Polymerization Polymerization - The chemical process, normally needs the aid of a catalyst, to form a polymer by bonding together monomers. http://www.youtube.com/watch?v=LJ5hjUeZt7U&feature=related
- 16. Polymers are made from Monomers http://www.youtube.com/watch?v=Jpv2RUjNFjo