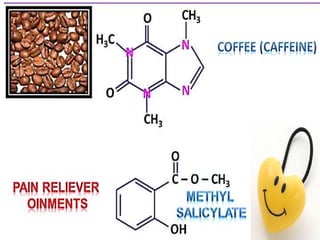
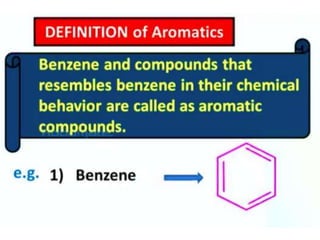
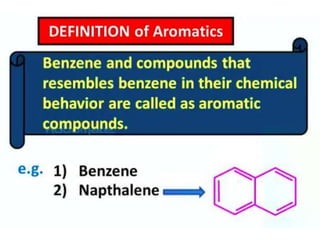
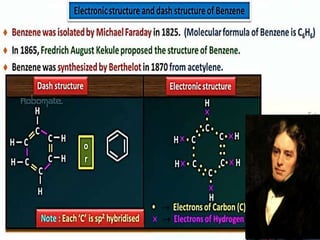
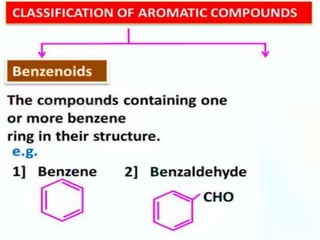
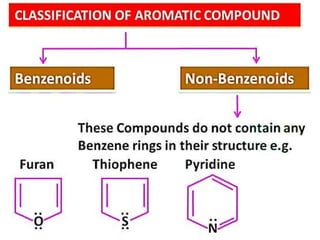
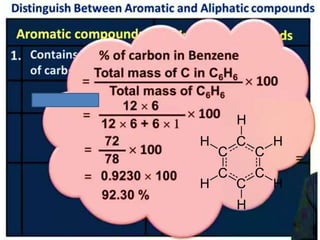
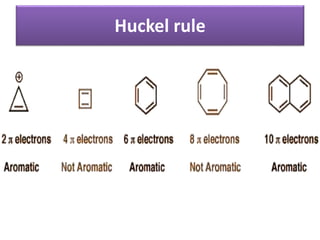
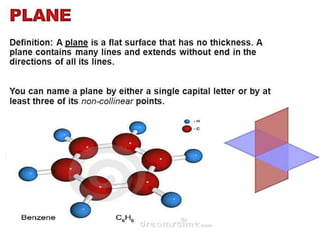
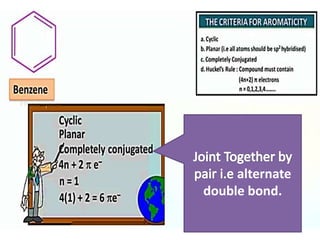
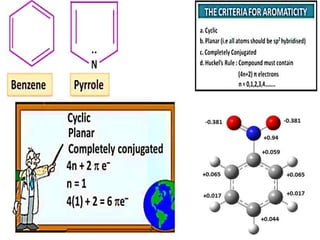
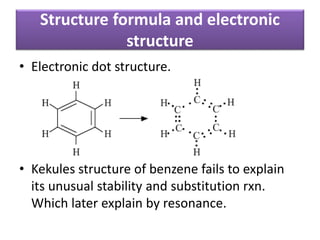
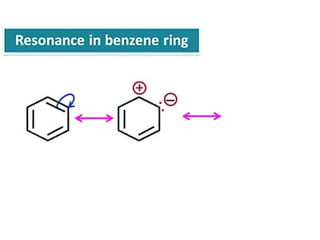
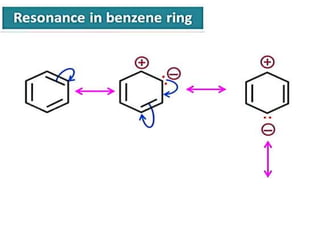
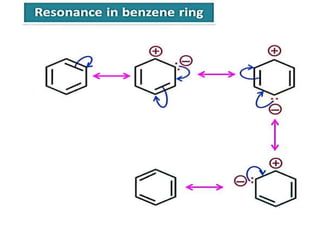
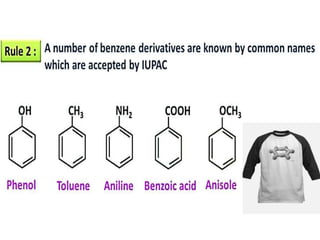
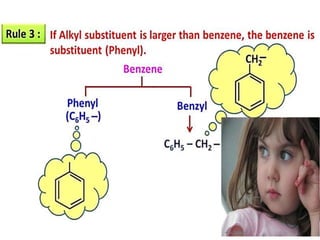
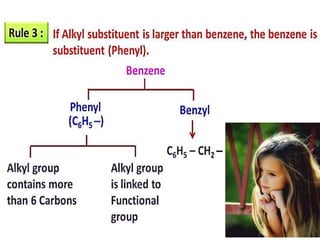
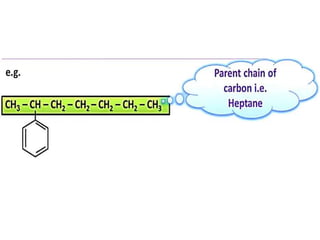
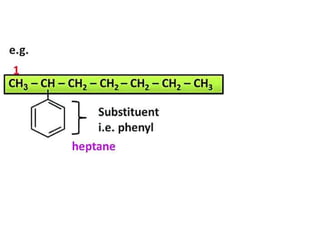
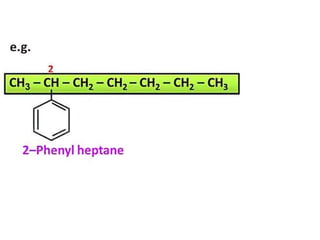
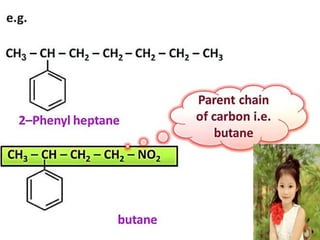
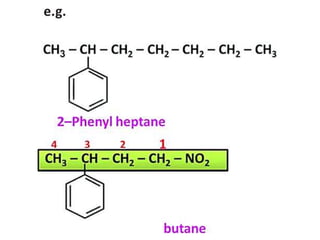
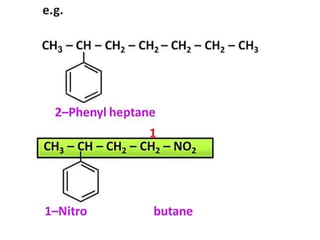
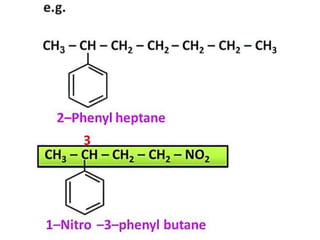
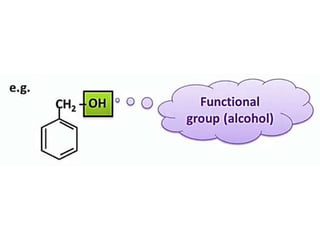
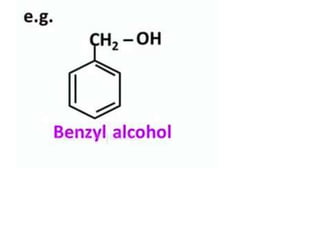
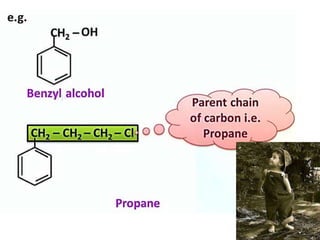
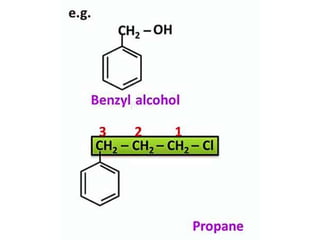
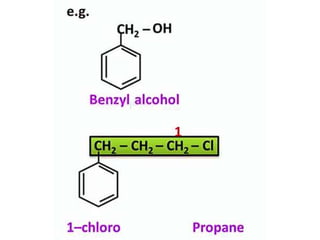
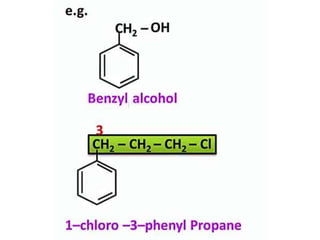
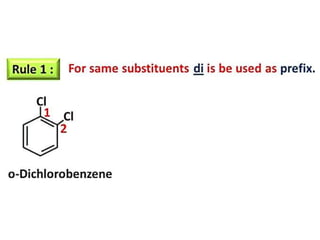
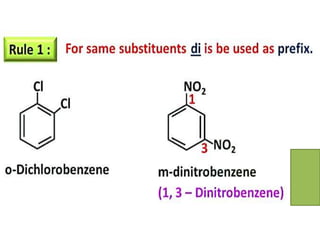
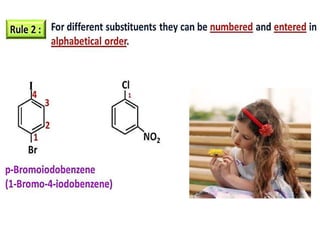
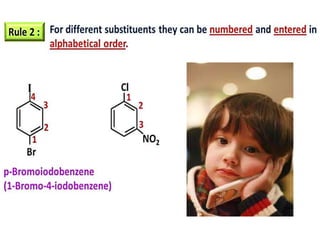
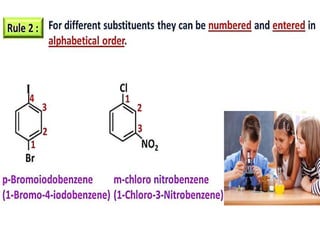
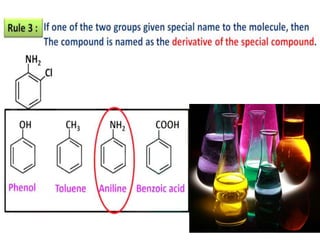
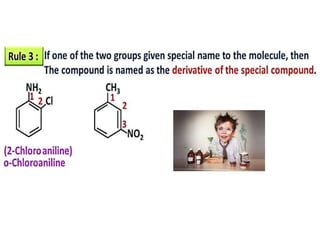
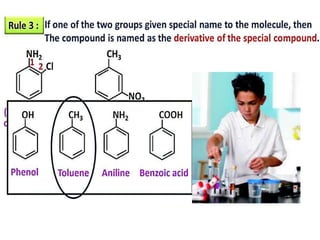
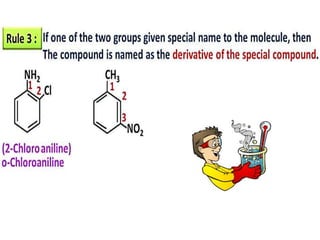
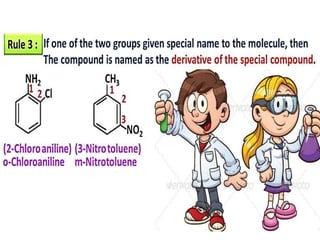
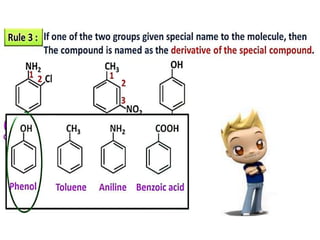
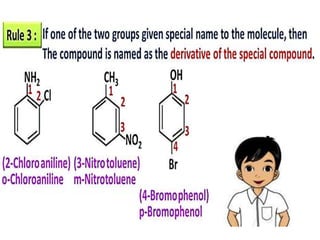
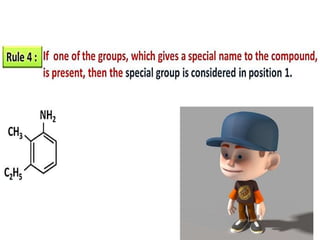
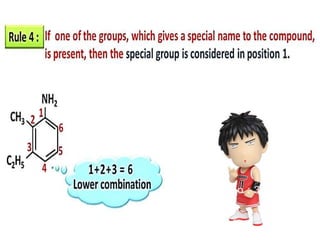
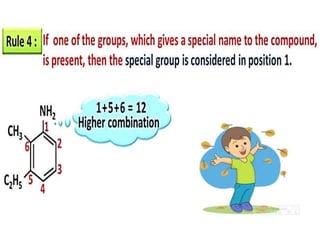
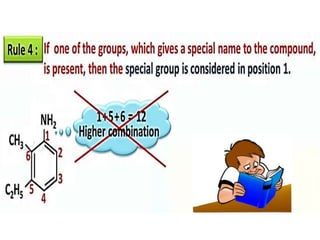
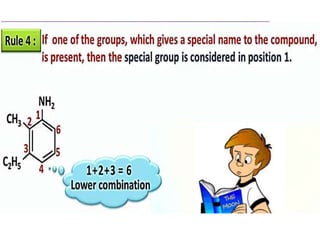
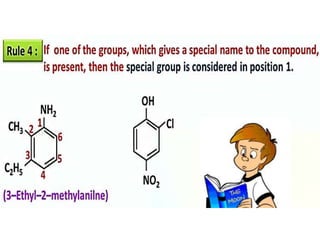
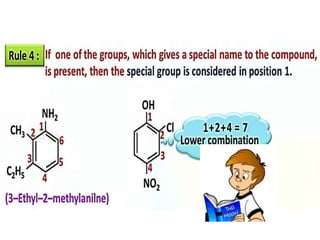
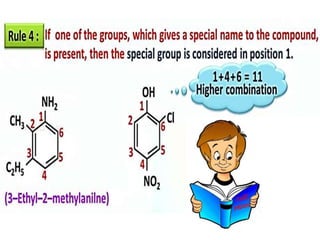
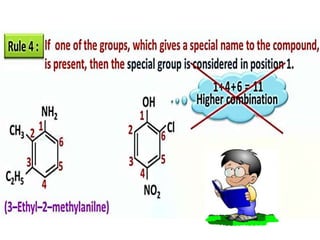
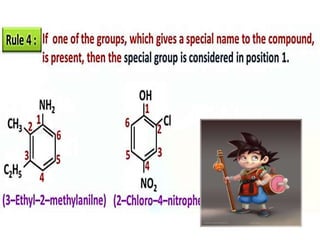
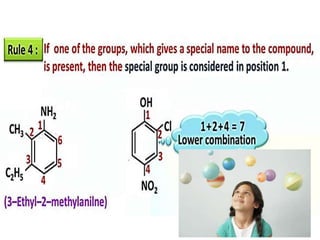
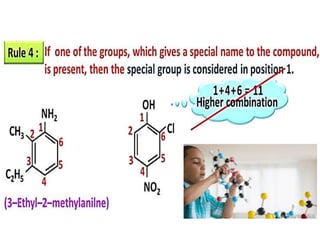
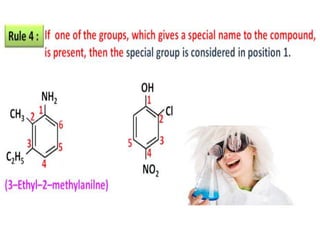
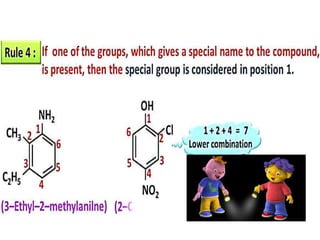
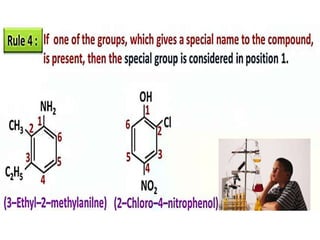
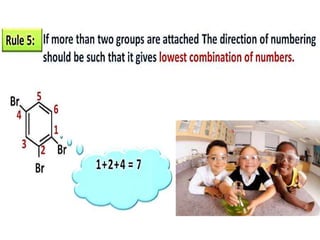
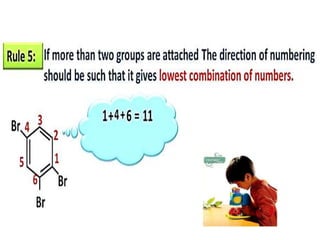
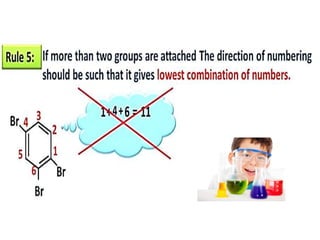
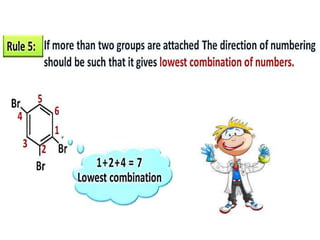
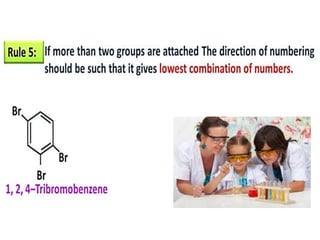
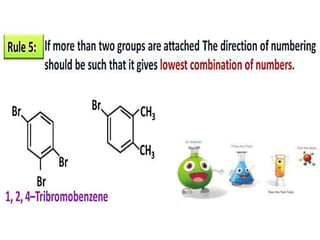
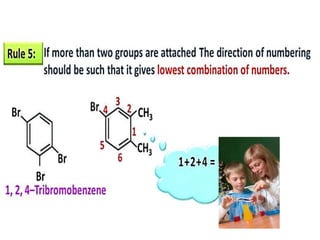
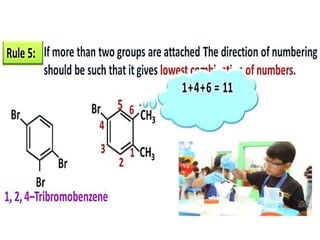
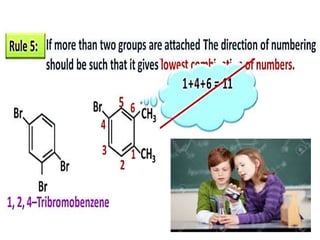
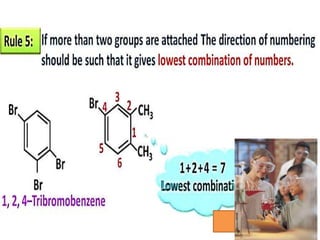
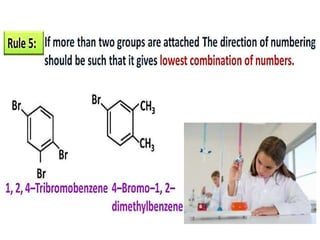
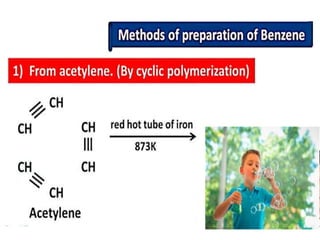
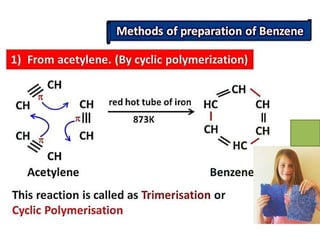
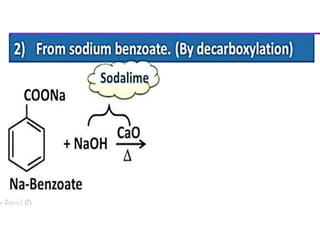
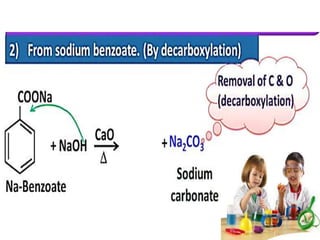
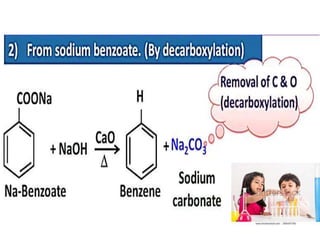
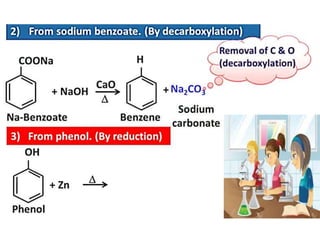
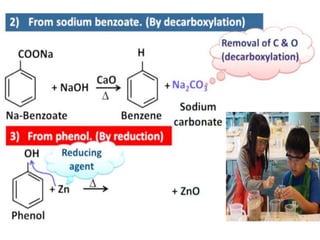
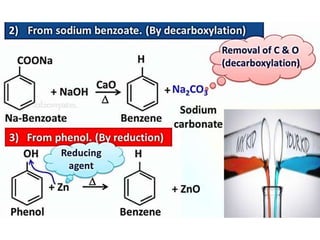
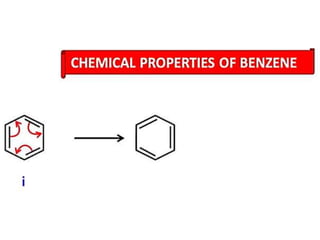
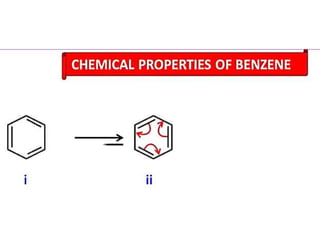
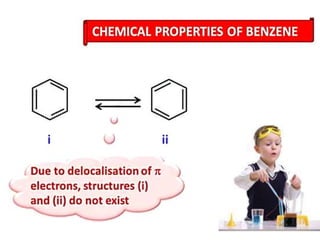
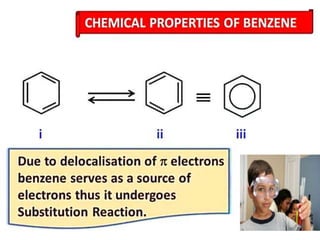
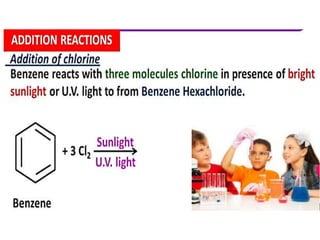
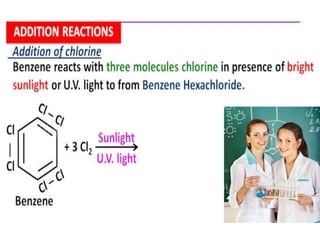
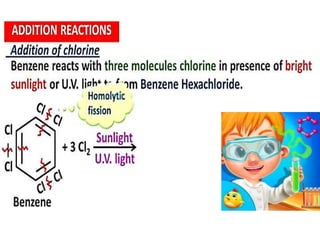
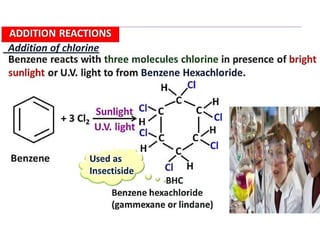
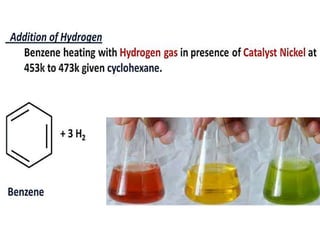
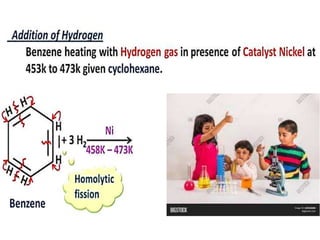
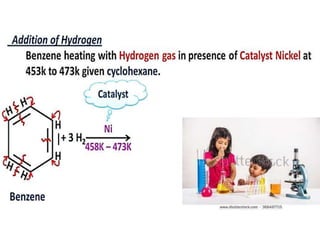
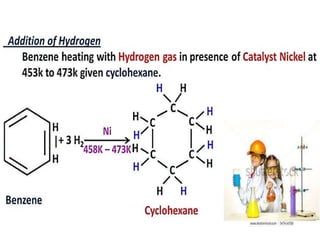
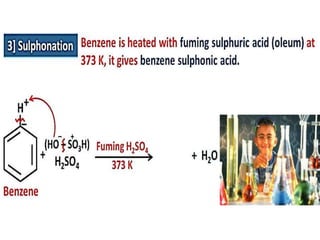
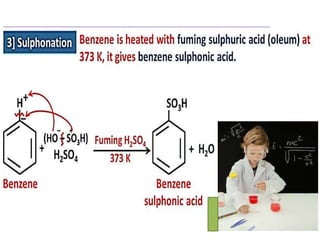
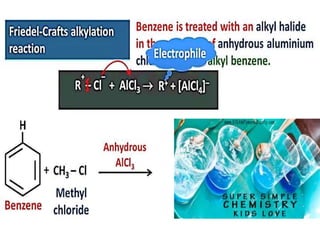
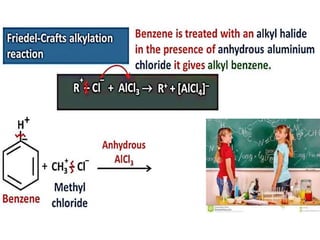
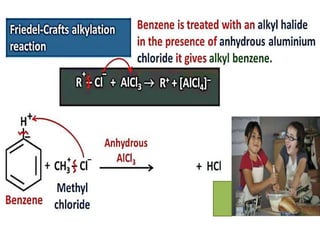
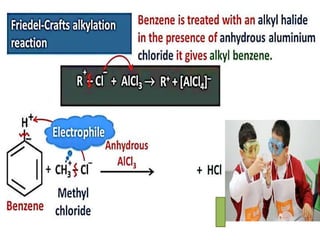
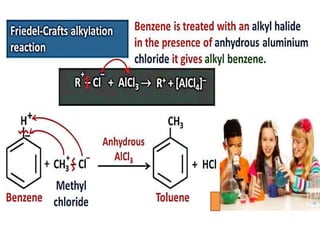
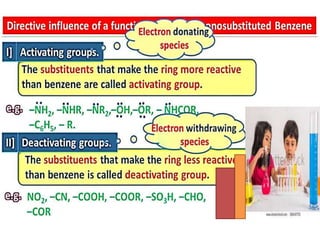
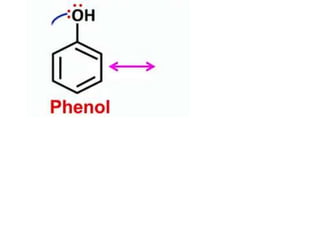
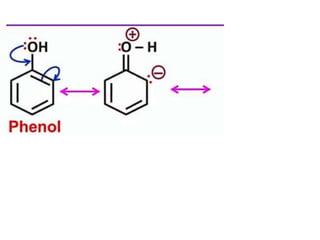
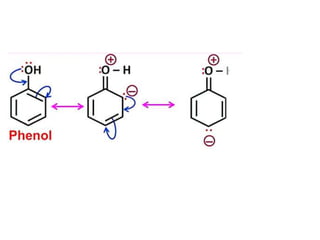
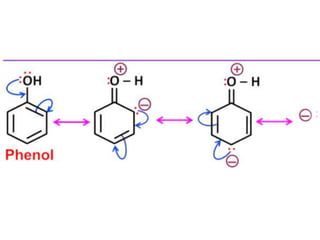
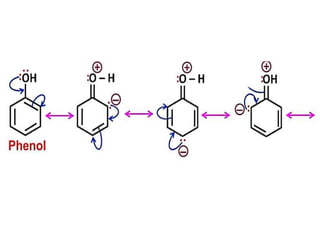
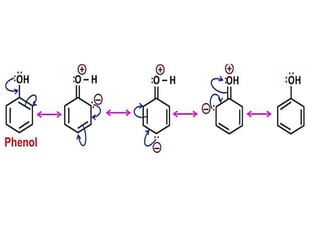
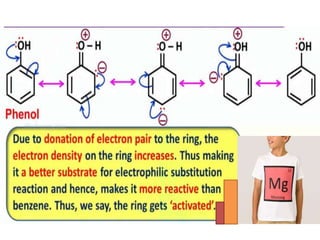
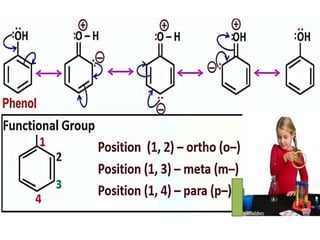
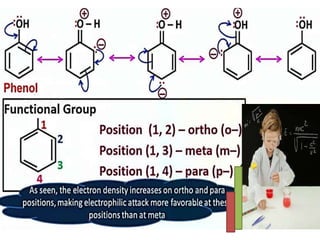
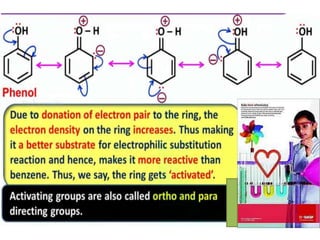
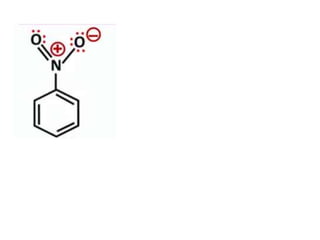
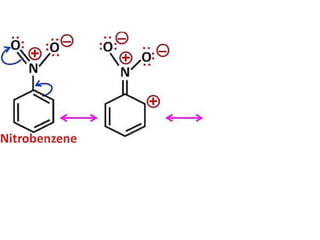
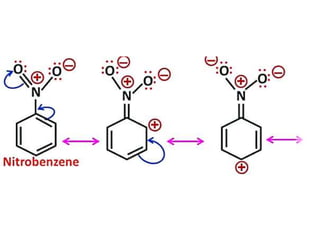
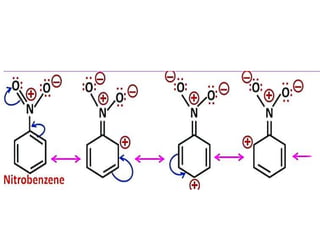
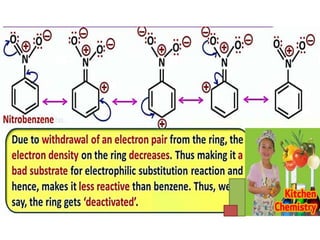
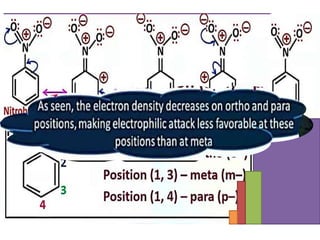
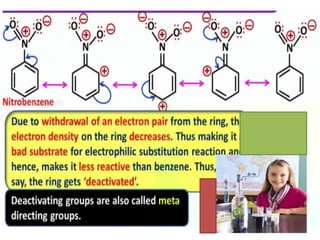
Aromatic compounds are characterized by planar, conjugated ring structures with delocalized pi-electrons. Benzene, with its cyclic structure of six carbon atoms each bonded to one hydrogen atom, is the simplest aromatic compound. It exhibits atypical stability and undergoes substitution rather than addition reactions. While some aromatic compounds have pleasant smells, aroma is not a requirement - anthracene is aromatic but odorless. The Hückel rule defines a compound as aromatic if it has (4n+2) pi electrons in its conjugated ring system, where n is an integer.