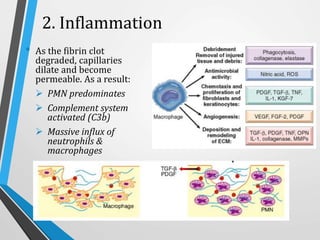
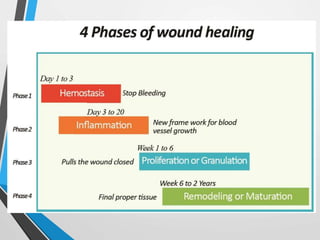
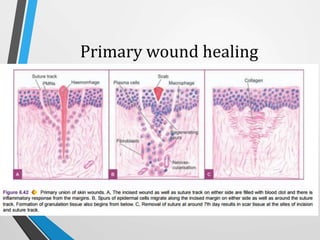
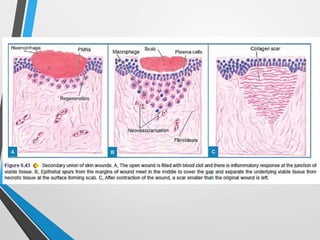
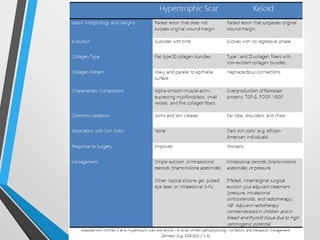
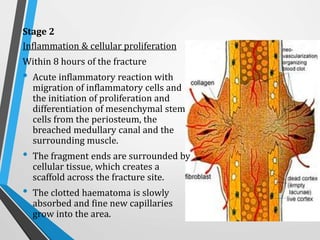
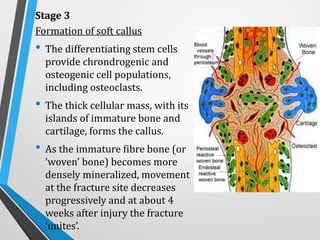
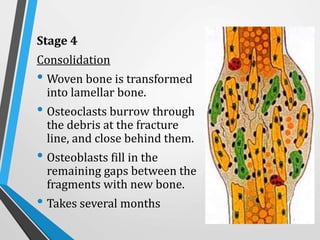
Wound and bone healing involve regeneration and repair processes. Wound healing occurs in four stages - hemostasis, inflammation, proliferation, and remodeling. There are three types of wound healing - primary, secondary, and delayed primary. Primary healing involves clean surgical wounds closed with sutures. Secondary healing involves open wounds without sutures that heal from the bottom up. Bone fracture healing similarly occurs through stages of hematoma formation, inflammation and callus formation, consolidation, and remodeling either directly or indirectly through callus formation. Factors like age, bone type, fracture pattern, and immobilization can affect bone healing.