This document discusses viral hepatitis, focusing on hepatitis A, B, C, D, and E. It provides details on the definition, causes, epidemiology, transmission, signs and symptoms, diagnosis, treatment and prevention of each type of viral hepatitis. Key points include that hepatitis A, B, C, D and E viruses are the main causes of viral hepatitis in humans. They differ in their transmission routes, clinical presentations, risk of chronic infection and availability of treatment. Vaccination is an effective way to prevent hepatitis A and B infection.


![3
http://rxpharmedico.in/
Laboratory tests
1. Positive serum immunoglobulin M anti–hepatitis A virus
2. Mild elevations of serum bilirubin, γ-globulin, and hepatic transaminase
(ALT [alanine transaminase] and aspartate transaminase [AST]) values to
about twice normal in acute anicteric disease
3. Elevations of alkaline phosphatase, γ-glutamyl transferase, and total
bilirubin in patients with cholestatic illness
Prevention
1. Prevention of hepatitis A infection can be achieved through
immunoprophylactic measures.
2. Immuno prophylaxis may be passive, active, or a combination of both. In
passive immunization, temporary protective antibody in the form of
immunoglobulin is administered.
3. In active immunization, a vaccine is administered to induce the formation
of protective antibody.
4. Prophylaxis can be administered before (pre-exposure prophylaxis) or
after exposure (postexposureprophylaxis).
Pre-Exposure Prophylaxis
Immunoglobulin
1. Before hepatitis A vaccine was available, the sole therapy for pre
exposure prophylaxis of hepatitis A infection was immunoglobulin.
2. Although passive immunization with immunoglobulin alone is highly
effective in preventing HAV infection,
3. The duration of protection is short.
4. When used for pre-exposure prophylaxis
5. A dose of 0.02 mL/kg of immunoglobulin administered intramuscularly
(IM) confers protection for less than 3 months, and an IM dose of 0.06
mL/kg confers protection for 5 months or longer for travellers.
Vaccine
1. Active immunization with hepatitis A vaccine has largely supplanted the
use of immunoglobulin for pre-exposure prophylaxis of infection caused
by HAV.](https://image.slidesharecdn.com/viralhepatitisbynomnaik-210303023703/85/VIRAL-HEPATITIS-by-NOM-KUMAR-NAIK-3-320.jpg)

![5
http://rxpharmedico.in/
Any person18 years of age or older having an indication for both
hepatitis A and hepatitis B vaccine can be given Twinrix, including
patients with chronic liver disease, users of illicit injectable drugs, men
who have sex with men, and persons with clotting factor disorders who
receive therapeutic blood products.
For international travel, hepatitis A vaccine is recommended; hepatitis B
vaccine is recommended for travelers to areas of high or intermediate
hepatitis B endemicity who plan to stay for longer than 6 months and
have frequent close contactwith the local population.
At 1 month after completion of the three-dose series, seroconversion for
anti-HAV (titer>20 milli-international units/mL) was elicited in 99.9% of
vaccines, and protective antibodies against HBsAg (anti-HBs>10 milli-
international units/mL) were elicited in 98.5% of vaccinees.
The persistence of anti-HAV and antibody to HBsAg (anti-HBs) after
administration is similar to that after single-antigen hepatitis A and B
vaccine administration at 4-year follow-up.
Observed adverse effects were generally similar in type and frequency to
those reported after vaccination with monovalent hepatitis A and B
vaccines.
VIRUS HAV HBV HCV HDV HEV
Genome RNA DNA RNA RNA RNA
Family Picornavirus Hepadnavirus Flavivirus Satellite Hepeviridae
Size (nm) 27 42 30-60 40 32
Incubation (days)
[mean]
15–50 [30] 45–180[80] 15–160 21–140[35] 15–65 [42]
Transmission
Oral
Percutaneous
Sexual
Perinatal
Common
Rare
No
No
Rare
Common
Common
Common
Rare
Common
Common
Rare
No
Common
Common
Common
Yes, common
Unknown
No
Rare](https://image.slidesharecdn.com/viralhepatitisbynomnaik-210303023703/85/VIRAL-HEPATITIS-by-NOM-KUMAR-NAIK-5-320.jpg)
![7
http://rxpharmedico.in/
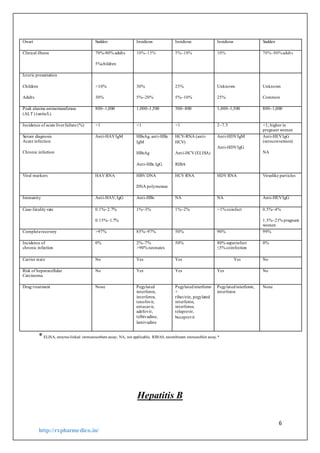
Aetiology
a. Caused by hepatitis B virus (HBY), belonging to the group of
hepadna viruses.
b. HBY comprises a surface envelope [antigen expressed on it is
called hepatitis B surface antigen (HBsAg)] and a nucleocapsid
core containing DNA [antigen expressed on its surface is called
hepatitis B core antigen (HBcAg), while another soluble antigen in
the nucleocapsid is called hepatitis Be antigen (HBeAg)].
Nucleocapsid core also bas DNA polymerase enzyme.
c. The correspondingantibodies are:
Anti-HBs
Anti-HBc
Anti-HBe.
d. HBsAg-positive serum containing HBeAg is more likely to be
highly infectious than HBeAg-negative or anti-HBe positive
serum.
Epidemiology
1. Incubation period is about 90 days (50--150 days). Hepatitis B is one of
the most common causes of chronic liver disease and hepatocellular
carcinoma worldwide.
2. Approximately one-third of the world’s population have serological
evidence of past or current infection with hepatitis B and approximately
350–400 million people are chronic HBsAg carriers.
3. It is estimated that 1.25 million people in the United States are
chronically infected with hepatitis B virus.
4. Approximately 78,000 people per year becomeacutely infected with the
hepatitis B virus in the United States.
5. Of those infected, 70% become symptomatic; 26% are hospitalized; 90%
of infants infected at birth, 30% of children infected at age 1 to 5 years,
and 6% of those infected over 5 years of age develop chronic infections;
and 1% from acute disease and 15% to 25% from chronic disease die
each year.
6. 14 Patients with cirrhosis are at a much greater risk for developing liver
cancer (12 to 300 times normal risk).
Transmission](https://image.slidesharecdn.com/viralhepatitisbynomnaik-210303023703/85/VIRAL-HEPATITIS-by-NOM-KUMAR-NAIK-7-320.jpg)
![8
http://rxpharmedico.in/
Transmitted through parenteral but occasionally non-parenteral
Transfusion of infected blood or blood products, injections with
contaminated needles, intravenous drug use with needle sharing, tattooing
and acupuncture.
Non-parenteral means of transmission include spread through bodyfluids
like saliva, urine, semen and vaginal secretions. However, this requires
close personal contact, sexual intercourse and male homosexuality.
Mother-to-child spread (perinatal transmission) is also common. It could
be either transplacental transmission or transmission at or soonafter birth.
Signsand symptoms
• Easy fatigability, anxiety, anorexia, and malaise
• Ascites, jaundice, variceal bleeding, and hepatic encephalopathy can
manifest with liver decompensation
• Hepatic encephalopathy is associated with hyperexcitability, impaired
mentation, confusion, obtundation, and eventually coma
• Vomiting and seizures
Physical examination
• Icteric sclera, skin, and secretions
• Decreased bowel sounds, increased abdominal girth, and detectable fluid
wave
• Asterixis
• Spider angiomata
Laboratory tests
• Presence of hepatitis B surface antigen for >6 months
• Intermittent elevations of hepatic transaminase (alanine transaminase [ALT]
and aspartate transaminase [AST]) and hepatitis B virus DNA >20,000 IU/mL
(105 copies/mL or 108 copies/L)
• Liver biopsies for pathologic classification aschronic persistent hepatitis,
chronic active hepatitis, or cirrhosis](https://image.slidesharecdn.com/viralhepatitisbynomnaik-210303023703/85/VIRAL-HEPATITIS-by-NOM-KUMAR-NAIK-8-320.jpg)










