Validation of factor xa assay for enoxaparin sodium enoxaparin injection
•
0 likes•624 views
chromogenic assay intended for the quantitative determination in purified solutions by measurement of factor IIa or Xa inhibition activity. The kit can be used for 100 test reactions as per microtiter plate protocol.
Report
Share
Report
Share
Download to read offline
Recommended
Bio assay of heparin

The document describes the bioassay method used to determine the potency of heparin. The method involves comparing the clotting time of samples with that of a standard heparin preparation using plasma and calcium chloride. Calculations are done to determine the potency ratio of the test sample to the standard based on their clotting times. The bioassay helps monitor heparin therapy and ensure concentrations remain in the therapeutic range.
Schedule B.pptx

This document outlines the fee schedules for various tests and analyses conducted by central and state drug laboratories in India. Schedule B lists 9 categories of fees for tests on drugs, including animal-based assays, microbiological tests, identification tests, physical tests, assays, polymorph tests, fees for sera and vaccines, cosmetics, and homeopathic medicines. Fees range from Rs. 50-5000 depending on the type of test. Schedule B(1) provides fees specifically for tests or analyses conducted by the Pharmacopoeial Laboratory for Indian Medicine or the Government Analyst, including sterility testing, microbiological assay, physiochemical testing, and animal-based tests.
Bioassay of Heparin Sodium

The document summarizes the bioassay of heparin sodium. The bioassay involves comparing the concentration of a standard heparin preparation needed to prevent clotting of plasma to that of the test heparin solution. Prepared plasma is mixed with increasing dilutions of both the standard and test heparin solutions and the degree of clotting is observed after 1 hour and used to determine the potency of the test sample relative to the standard. Heparin is an anticoagulant drug extracted from animal tissues and used to treat and prevent blood clots and thrombosis. Its potency is typically measured using clotting-based biological assays.
Expt. 5 Study of anti ulcer activity of a drug using nsaid induced ulcer model

This document describes an experiment to study the anti-ulcer activity of drugs using a NSAID-induced ulcer model in rats. The experiment involves pretreating rats with cimetidine or ranitidine before administering ulcerogenic NSAIDs like aspirin or indomethacin. The rats are then sacrificed and their stomachs examined for ulcers. Parameters like gastric volume, acidity, and ulcer index are measured and compared between treated and untreated groups to determine the antisecretory and ulcer protective effects of cimetidine and ranitidine. The results show that cimetidine and ranitidine reduce gastric acid secretion and inhibit ulcer formation in NSAID-treated rats.
Validation of Factor IIa for heparin sodium or heparin injection

A chromogenic assay intended for the quantitative determination in purified solutions by measurement of factor IIa or Xa inhibition activity. The kit can be used for 100 test reactions as per microtiter plate protocol.
Bioassy of insulin according to Indian pharmacopoeia

This document summarizes several methods for bioassaying insulin, including preparation of standard and test solutions, rabbit, mouse, rat diaphragm, and rat epididymal fat pad methods. It also describes the radioimmunoassay method, which uses radiolabeled insulin and antibodies to determine insulin concentration in test samples by comparing to standard curves.
Tetanus antitoxin (apa)

This document discusses tetanus, the causative bacterium Clostridium tetani, symptoms of tetanus, and the use and production of tetanus antitoxin. Specifically, it notes that tetanus is caused by C. tetani bacteria entering wounds and releasing a neurotoxin. Tetanus antitoxin is a solution of proteins from immunized horse blood that neutralizes the tetanus toxin to prevent or treat symptoms like muscle spasms. The document outlines the process for determining the potency of tetanus antitoxin compared to a standard preparation through mouse experiments.
Biological test and assay of Absorbed Diphtheria vaccine

This document discusses vaccines and the biological assay of adsorbed diphtheria vaccines. It defines vaccines as substances containing harmless forms of disease-causing germs. The two main classifications of vaccines are live/attenuated vaccines, which use weakened live pathogens, and dead/inactivated vaccines, which use killed pathogens. The document then focuses on adsorbed diphtheria vaccines, describing their production, recommended dosing, and two methods for determining their potency/effectiveness - the intradermal challenge method using guinea pigs and the lethal challenge method. Both methods involve injecting vaccinated and unvaccinated guinea pigs with diphtheria toxin to compare protection levels.
Recommended
Bio assay of heparin

The document describes the bioassay method used to determine the potency of heparin. The method involves comparing the clotting time of samples with that of a standard heparin preparation using plasma and calcium chloride. Calculations are done to determine the potency ratio of the test sample to the standard based on their clotting times. The bioassay helps monitor heparin therapy and ensure concentrations remain in the therapeutic range.
Schedule B.pptx

This document outlines the fee schedules for various tests and analyses conducted by central and state drug laboratories in India. Schedule B lists 9 categories of fees for tests on drugs, including animal-based assays, microbiological tests, identification tests, physical tests, assays, polymorph tests, fees for sera and vaccines, cosmetics, and homeopathic medicines. Fees range from Rs. 50-5000 depending on the type of test. Schedule B(1) provides fees specifically for tests or analyses conducted by the Pharmacopoeial Laboratory for Indian Medicine or the Government Analyst, including sterility testing, microbiological assay, physiochemical testing, and animal-based tests.
Bioassay of Heparin Sodium

The document summarizes the bioassay of heparin sodium. The bioassay involves comparing the concentration of a standard heparin preparation needed to prevent clotting of plasma to that of the test heparin solution. Prepared plasma is mixed with increasing dilutions of both the standard and test heparin solutions and the degree of clotting is observed after 1 hour and used to determine the potency of the test sample relative to the standard. Heparin is an anticoagulant drug extracted from animal tissues and used to treat and prevent blood clots and thrombosis. Its potency is typically measured using clotting-based biological assays.
Expt. 5 Study of anti ulcer activity of a drug using nsaid induced ulcer model

This document describes an experiment to study the anti-ulcer activity of drugs using a NSAID-induced ulcer model in rats. The experiment involves pretreating rats with cimetidine or ranitidine before administering ulcerogenic NSAIDs like aspirin or indomethacin. The rats are then sacrificed and their stomachs examined for ulcers. Parameters like gastric volume, acidity, and ulcer index are measured and compared between treated and untreated groups to determine the antisecretory and ulcer protective effects of cimetidine and ranitidine. The results show that cimetidine and ranitidine reduce gastric acid secretion and inhibit ulcer formation in NSAID-treated rats.
Validation of Factor IIa for heparin sodium or heparin injection

A chromogenic assay intended for the quantitative determination in purified solutions by measurement of factor IIa or Xa inhibition activity. The kit can be used for 100 test reactions as per microtiter plate protocol.
Bioassy of insulin according to Indian pharmacopoeia

This document summarizes several methods for bioassaying insulin, including preparation of standard and test solutions, rabbit, mouse, rat diaphragm, and rat epididymal fat pad methods. It also describes the radioimmunoassay method, which uses radiolabeled insulin and antibodies to determine insulin concentration in test samples by comparing to standard curves.
Tetanus antitoxin (apa)

This document discusses tetanus, the causative bacterium Clostridium tetani, symptoms of tetanus, and the use and production of tetanus antitoxin. Specifically, it notes that tetanus is caused by C. tetani bacteria entering wounds and releasing a neurotoxin. Tetanus antitoxin is a solution of proteins from immunized horse blood that neutralizes the tetanus toxin to prevent or treat symptoms like muscle spasms. The document outlines the process for determining the potency of tetanus antitoxin compared to a standard preparation through mouse experiments.
Biological test and assay of Absorbed Diphtheria vaccine

This document discusses vaccines and the biological assay of adsorbed diphtheria vaccines. It defines vaccines as substances containing harmless forms of disease-causing germs. The two main classifications of vaccines are live/attenuated vaccines, which use weakened live pathogens, and dead/inactivated vaccines, which use killed pathogens. The document then focuses on adsorbed diphtheria vaccines, describing their production, recommended dosing, and two methods for determining their potency/effectiveness - the intradermal challenge method using guinea pigs and the lethal challenge method. Both methods involve injecting vaccinated and unvaccinated guinea pigs with diphtheria toxin to compare protection levels.
Experiment no 1

Study of anti-ulcer activity of a drug using pylorus ligand (SHAY) rat model and NSAIDS induced ulcer model
Monograph of any drug as per i.p

The document provides a monograph for Paracetamol as per the Indian Pharmacopoeia. It details the chemical and physical properties of Paracetamol including its definition, characteristics, identification tests, purity tests, assay, storage requirements, impurities, uses, and formulations. Paracetamol has the chemical name N-(4-Hydroxyphenyl)acetamide and is a white, crystalline powder that is sparingly soluble in water and soluble in alcohol. It is used as an analgesic and antipyretic drug in various preparations like tablets and oral solutions.
Biological test and assay of Antivenom

Snake venom contains various toxic proteins and enzymes that can be deadly to humans in small amounts. Antivenom is produced by injecting snake venom into other animals like horses to induce an immune response and antibody production. The purified antibodies from these animals are then used to treat snake bites. Biological testing of antivenoms involves injecting diluted venom samples into mice and observing mortality over 24 hours to determine the effective dose and confirm antivenom neutralization of venom toxins.
Bioassay of Heparin

This document summarizes the bioassay method used to test the potency of heparin preparations. The method involves comparing the clotting time of plasma incubated with the test heparin sample to the clotting time of plasma incubated with a reference standard heparin preparation. Several dilutions of both the standard and test sample are tested to identify the dilution for each that results in equivalent clotting time. The potency of the test sample can then be estimated based on its equivalent dilution relative to the standard. The bioassay follows specific procedures for obtaining and preparing plasma, making standard and test solutions, conducting the clotting time assays, and calculating potency results.
Stability indicating rp hplc method development and validation for simultaneo...

The document describes the development and validation of a stability-indicating RP-HPLC method for the simultaneous estimation of levodropropizine and chlorpheniramine maleate in bulk and pharmaceutical dosage forms. The method utilizes a C18 column with a mobile phase of 45% 0.1% orthophosphoric acid and 55% acetonitrile at a flow rate of 1 mL/min. The method was validated for parameters such as linearity, accuracy, precision, specificity, limit of detection, limit of quantification and robustness according to ICH guidelines. Forced degradation studies were also performed to demonstrate the stability-indicating ability of the developed method.
HDT Practical Exp.1

This document describes an experiment to perform preliminary phytochemical screening of crude drugs to determine their chemical constituents. The procedure involves extracting plant materials using organic solvents like ethanol, methanol and acetone as well as aqueous extraction. The various extracts are then tested for the presence of phytochemicals like proteins, carbohydrates, phenols, tannins, flavonoids, saponins, glycosides, steroids and terpenoids using standard tests involving reagents like Millon's reagent, Ninhydrin, Fehling's solution, Benedict's reagent, Molisch's reagent, ferric chloride, magnesium ribbon, sodium hydroxide, acetic acid, chloroform, sulfur
Expt. 11 Determination of acute eye irritation corrosion of a test substance

This document outlines the procedure for conducting an acute eye irritation/corrosion test of a substance using albino rabbits. The objective is to determine the degree of eye irritation or corrosion caused by a test substance. A single dose of the substance is applied to one eye of each rabbit, while the other eye serves as a control. Lesions to the conjunctiva, cornea, and iris are observed and graded at various time intervals over 72 hours. Based on the grading scores of the test eye compared to the control eye, the sensitization potential of the substance can be determined.
Antibiotic -Drugs/ Medicinal Chemistry

This document provides an overview of β-lactam antibiotics, including penicillins. It begins by classifying common antibiotics and listing their mechanisms of action. It then focuses on β-lactam antibiotics, describing their structure and major subclasses like penicillins, cephalosporins, carbapenems, and monobactams. Specific penicillins are discussed in depth, highlighting their structures, spectra of activity, stability properties, and mechanisms of resistance. β-lactamase inhibitors like clavulanic acid are also introduced.
Expt. 7 Effect of saline purgative on frog intestine

Objective
Principle
Requirements
Procedure
Observations
Observation table
Result and interpretation
Additional information
BIOASSAY OF TITANUS ANTI TIOXIN AND DIPTHERIA VACCINE

This document provides information about tetanus antitoxin and adsorbed diphtheria vaccine. It discusses the principles, preparation, and potency testing of these antitoxins. For tetanus antitoxin, the document explains that it contains antibodies that neutralize toxins produced by Clostridium tetani. It also describes the mouse protection test used to determine potency relative to a reference standard. For adsorbed diphtheria vaccine, the document states that it contains antibodies against diphtheria toxin from Corynebacterium diphtheriae. It further discusses the guinea pig erythema test used to assess potency by comparing to reference toxin dilutions.
Isolation, identification & estimation by Pooja Khanpara 

This document discusses the isolation, identification, and analysis of phytoconstituents from various plant sources. It provides details on four methods for isolating diosgenin from fenugreek, including alcoholic extraction, acid hydrolysis, fermentation with acid hydrolysis, and incubation with acid hydrolysis. It also discusses the isolation of atropine from Atropa belladonna through ethanol extraction and acid-base partitioning. Methods for isolating reserpine from Rauwolfia serpentina roots include benzene extraction, acid-base partitioning, and chromatography. Identification tests and analytical techniques like TLC, HPTLC, and GC-MS are also summarized.
Expt. 2 Bioassay of acetylcholine using rat ileum by four point bioassay

Objective
Principle
Requirements
Experimental specifications (conditions)
Preparation of ACh stock and standard solutions
Preparation of Tyrode solution (PSS)
Procedure
Kymograph recording of contractions
Observation table
Calculation
Result and interpretation
Anti-inflammatory activity of drugs using carrageenan induced paw-edema model

This document describes a study to evaluate the anti-inflammatory activity of drugs using a carrageenan-induced paw edema model in rats. The study involves injecting carrageenan into the paws of rats to cause inflammation and edema, which are measured over time using a plethysmometer. Rats are divided into control and test groups, with the test group receiving indomethacin prior to carrageenan injection. Paw volumes are measured and compared between groups to determine the ability of indomethacin to reduce edema caused by carrageenan. The results show that indomethacin demonstrated anti-inflammatory effects by inhibiting the paw edema induced by carrageenan.
To synthesize and submit sulphanilamide

This document provides instructions for synthesizing sulphanilamide from acetanilide in 3 steps:
1. Acetanilide is treated with chlorosulphonic acid to form the intermediate p-acetamidobenzenesulphonyl chloride.
2. p-acetamidobenzenesulphonyl chloride is reacted with ammonia to form p-acetamidobenzenesulphonamide.
3. p-acetamidobenzenesulphonamide undergoes acid hydrolysis with hydrochloric acid to remove the acetamide group and yield the final product sulphanilamide hydrochloride.
The percentage yield of sulphan
Expt. 1 Bioassay of serotonin using rat fundus strip by three point bioassay

Objective
Principle
Requirements
Experimental specifications (conditions)
Preparation of serotonin stock solution
Preparation of Krebs solution (PSS)
Procedure
Kymograph recording of contractions
Observation table
Calculation
Result and interpretation
Expt. 1 Bioassay of serotonin using rat fundus strip by three point bioassay

This document describes an experiment to determine the unknown concentration of serotonin using a three-point bioassay with an isolated rat fundus strip preparation. The experiment involves constructing dose-response curves for a serotonin standard and test sample, selecting doses that elicit submaximal responses in a 1:2 ratio, and determining the test concentration using the measured responses. Rat fundus tissue is sensitive to serotonin and contracts in a concentration-dependent manner when exposed to increasing doses of the drug. The experiment aims to precisely and reliably estimate the concentration of an unknown serotonin sample through this validated bioassay method.
Micro biological assay of vaccines

This document describes microbiological assays for various vaccines including diphtheria, rabies, and tetanus vaccines. It discusses the principles, procedures, test animals, preparations, and interpretations for biological assays of these vaccines. The assays involve comparing the potency of a test vaccine to a reference vaccine by determining the dose needed to protect animals from challenges with toxins or viruses. The document provides technical details on how to conduct the assays and evaluate results to validate the tests and determine if a vaccine meets potency requirements.
Compendial methods for evalution of crude drug and herbal formulation

1. The document discusses various parameters for evaluating crude drugs and herbal formulations, including physical, chemical, biological, and toxicological parameters.
2. Key physical parameters include viscosity, moisture content, density, and ash value. Chemical parameters involve detecting compounds like alkaloids, carbohydrates, fixed oils, and phenolic compounds.
3. Biological/toxicological parameters include determining pesticide residues, arsenic, heavy metals, and radioactive contamination using techniques like gas chromatography and colorimetric analysis.
Industrial Production, Estimation and Utilization of the Phytoconstituents

Forskolin, Sennoside, Artemisinin, Diosgenin, Digoxin, Atropine, Podophyllotoxin, Caffeine, Taxol, Vincristine and Vinblastine
Expt. 9 Test for pyrogens ( rabbit method)

What is pyrogens?
Sources of pyrogens and its elimination methods
Tests for pyrogens-
1. In Vitro Test / LAL Test
2. In Vivo Test / Rabbit Test.
Objective
Principle
Requirements
Procedure
Observation table
Result and interpretation
Validation of factor xa assay for nadroparin calcium nadroparin injection

This document describes a chromogenic assay used to quantify the anticoagulant Nadroparin in samples by measuring its inhibitory effect on factor Xa. The assay involves incubating Nadroparin samples with factor Xa and anti-thrombin III, then adding a chromogenic substrate and measuring the absorbance. The absorbance is inversely proportional to the Nadroparin concentration as it inhibits factor Xa activity. The assay kit and procedure are validated for use in quantifying Nadroparin levels in bioequivalence studies.
Validation of factor xa assay for heparin sodium heparin injection

This document summarizes the procedures for using a chromogenic assay kit to quantify heparin levels in samples by measuring heparin's anti-factor Xa activity. The kit utilizes heparin's ability to catalyze the reaction between factor Xa and antithrombin III, which allows the residual factor Xa activity to be measured and used to determine heparin concentration in unknown samples. The document outlines the materials provided, reconstitution procedures, standard and sample preparation steps, assay procedure, and methods for data analysis and interpretation of results.
More Related Content
What's hot
Experiment no 1

Study of anti-ulcer activity of a drug using pylorus ligand (SHAY) rat model and NSAIDS induced ulcer model
Monograph of any drug as per i.p

The document provides a monograph for Paracetamol as per the Indian Pharmacopoeia. It details the chemical and physical properties of Paracetamol including its definition, characteristics, identification tests, purity tests, assay, storage requirements, impurities, uses, and formulations. Paracetamol has the chemical name N-(4-Hydroxyphenyl)acetamide and is a white, crystalline powder that is sparingly soluble in water and soluble in alcohol. It is used as an analgesic and antipyretic drug in various preparations like tablets and oral solutions.
Biological test and assay of Antivenom

Snake venom contains various toxic proteins and enzymes that can be deadly to humans in small amounts. Antivenom is produced by injecting snake venom into other animals like horses to induce an immune response and antibody production. The purified antibodies from these animals are then used to treat snake bites. Biological testing of antivenoms involves injecting diluted venom samples into mice and observing mortality over 24 hours to determine the effective dose and confirm antivenom neutralization of venom toxins.
Bioassay of Heparin

This document summarizes the bioassay method used to test the potency of heparin preparations. The method involves comparing the clotting time of plasma incubated with the test heparin sample to the clotting time of plasma incubated with a reference standard heparin preparation. Several dilutions of both the standard and test sample are tested to identify the dilution for each that results in equivalent clotting time. The potency of the test sample can then be estimated based on its equivalent dilution relative to the standard. The bioassay follows specific procedures for obtaining and preparing plasma, making standard and test solutions, conducting the clotting time assays, and calculating potency results.
Stability indicating rp hplc method development and validation for simultaneo...

The document describes the development and validation of a stability-indicating RP-HPLC method for the simultaneous estimation of levodropropizine and chlorpheniramine maleate in bulk and pharmaceutical dosage forms. The method utilizes a C18 column with a mobile phase of 45% 0.1% orthophosphoric acid and 55% acetonitrile at a flow rate of 1 mL/min. The method was validated for parameters such as linearity, accuracy, precision, specificity, limit of detection, limit of quantification and robustness according to ICH guidelines. Forced degradation studies were also performed to demonstrate the stability-indicating ability of the developed method.
HDT Practical Exp.1

This document describes an experiment to perform preliminary phytochemical screening of crude drugs to determine their chemical constituents. The procedure involves extracting plant materials using organic solvents like ethanol, methanol and acetone as well as aqueous extraction. The various extracts are then tested for the presence of phytochemicals like proteins, carbohydrates, phenols, tannins, flavonoids, saponins, glycosides, steroids and terpenoids using standard tests involving reagents like Millon's reagent, Ninhydrin, Fehling's solution, Benedict's reagent, Molisch's reagent, ferric chloride, magnesium ribbon, sodium hydroxide, acetic acid, chloroform, sulfur
Expt. 11 Determination of acute eye irritation corrosion of a test substance

This document outlines the procedure for conducting an acute eye irritation/corrosion test of a substance using albino rabbits. The objective is to determine the degree of eye irritation or corrosion caused by a test substance. A single dose of the substance is applied to one eye of each rabbit, while the other eye serves as a control. Lesions to the conjunctiva, cornea, and iris are observed and graded at various time intervals over 72 hours. Based on the grading scores of the test eye compared to the control eye, the sensitization potential of the substance can be determined.
Antibiotic -Drugs/ Medicinal Chemistry

This document provides an overview of β-lactam antibiotics, including penicillins. It begins by classifying common antibiotics and listing their mechanisms of action. It then focuses on β-lactam antibiotics, describing their structure and major subclasses like penicillins, cephalosporins, carbapenems, and monobactams. Specific penicillins are discussed in depth, highlighting their structures, spectra of activity, stability properties, and mechanisms of resistance. β-lactamase inhibitors like clavulanic acid are also introduced.
Expt. 7 Effect of saline purgative on frog intestine

Objective
Principle
Requirements
Procedure
Observations
Observation table
Result and interpretation
Additional information
BIOASSAY OF TITANUS ANTI TIOXIN AND DIPTHERIA VACCINE

This document provides information about tetanus antitoxin and adsorbed diphtheria vaccine. It discusses the principles, preparation, and potency testing of these antitoxins. For tetanus antitoxin, the document explains that it contains antibodies that neutralize toxins produced by Clostridium tetani. It also describes the mouse protection test used to determine potency relative to a reference standard. For adsorbed diphtheria vaccine, the document states that it contains antibodies against diphtheria toxin from Corynebacterium diphtheriae. It further discusses the guinea pig erythema test used to assess potency by comparing to reference toxin dilutions.
Isolation, identification & estimation by Pooja Khanpara 

This document discusses the isolation, identification, and analysis of phytoconstituents from various plant sources. It provides details on four methods for isolating diosgenin from fenugreek, including alcoholic extraction, acid hydrolysis, fermentation with acid hydrolysis, and incubation with acid hydrolysis. It also discusses the isolation of atropine from Atropa belladonna through ethanol extraction and acid-base partitioning. Methods for isolating reserpine from Rauwolfia serpentina roots include benzene extraction, acid-base partitioning, and chromatography. Identification tests and analytical techniques like TLC, HPTLC, and GC-MS are also summarized.
Expt. 2 Bioassay of acetylcholine using rat ileum by four point bioassay

Objective
Principle
Requirements
Experimental specifications (conditions)
Preparation of ACh stock and standard solutions
Preparation of Tyrode solution (PSS)
Procedure
Kymograph recording of contractions
Observation table
Calculation
Result and interpretation
Anti-inflammatory activity of drugs using carrageenan induced paw-edema model

This document describes a study to evaluate the anti-inflammatory activity of drugs using a carrageenan-induced paw edema model in rats. The study involves injecting carrageenan into the paws of rats to cause inflammation and edema, which are measured over time using a plethysmometer. Rats are divided into control and test groups, with the test group receiving indomethacin prior to carrageenan injection. Paw volumes are measured and compared between groups to determine the ability of indomethacin to reduce edema caused by carrageenan. The results show that indomethacin demonstrated anti-inflammatory effects by inhibiting the paw edema induced by carrageenan.
To synthesize and submit sulphanilamide

This document provides instructions for synthesizing sulphanilamide from acetanilide in 3 steps:
1. Acetanilide is treated with chlorosulphonic acid to form the intermediate p-acetamidobenzenesulphonyl chloride.
2. p-acetamidobenzenesulphonyl chloride is reacted with ammonia to form p-acetamidobenzenesulphonamide.
3. p-acetamidobenzenesulphonamide undergoes acid hydrolysis with hydrochloric acid to remove the acetamide group and yield the final product sulphanilamide hydrochloride.
The percentage yield of sulphan
Expt. 1 Bioassay of serotonin using rat fundus strip by three point bioassay

Objective
Principle
Requirements
Experimental specifications (conditions)
Preparation of serotonin stock solution
Preparation of Krebs solution (PSS)
Procedure
Kymograph recording of contractions
Observation table
Calculation
Result and interpretation
Expt. 1 Bioassay of serotonin using rat fundus strip by three point bioassay

This document describes an experiment to determine the unknown concentration of serotonin using a three-point bioassay with an isolated rat fundus strip preparation. The experiment involves constructing dose-response curves for a serotonin standard and test sample, selecting doses that elicit submaximal responses in a 1:2 ratio, and determining the test concentration using the measured responses. Rat fundus tissue is sensitive to serotonin and contracts in a concentration-dependent manner when exposed to increasing doses of the drug. The experiment aims to precisely and reliably estimate the concentration of an unknown serotonin sample through this validated bioassay method.
Micro biological assay of vaccines

This document describes microbiological assays for various vaccines including diphtheria, rabies, and tetanus vaccines. It discusses the principles, procedures, test animals, preparations, and interpretations for biological assays of these vaccines. The assays involve comparing the potency of a test vaccine to a reference vaccine by determining the dose needed to protect animals from challenges with toxins or viruses. The document provides technical details on how to conduct the assays and evaluate results to validate the tests and determine if a vaccine meets potency requirements.
Compendial methods for evalution of crude drug and herbal formulation

1. The document discusses various parameters for evaluating crude drugs and herbal formulations, including physical, chemical, biological, and toxicological parameters.
2. Key physical parameters include viscosity, moisture content, density, and ash value. Chemical parameters involve detecting compounds like alkaloids, carbohydrates, fixed oils, and phenolic compounds.
3. Biological/toxicological parameters include determining pesticide residues, arsenic, heavy metals, and radioactive contamination using techniques like gas chromatography and colorimetric analysis.
Industrial Production, Estimation and Utilization of the Phytoconstituents

Forskolin, Sennoside, Artemisinin, Diosgenin, Digoxin, Atropine, Podophyllotoxin, Caffeine, Taxol, Vincristine and Vinblastine
Expt. 9 Test for pyrogens ( rabbit method)

What is pyrogens?
Sources of pyrogens and its elimination methods
Tests for pyrogens-
1. In Vitro Test / LAL Test
2. In Vivo Test / Rabbit Test.
Objective
Principle
Requirements
Procedure
Observation table
Result and interpretation
What's hot (20)
Stability indicating rp hplc method development and validation for simultaneo...

Stability indicating rp hplc method development and validation for simultaneo...
Expt. 11 Determination of acute eye irritation corrosion of a test substance

Expt. 11 Determination of acute eye irritation corrosion of a test substance
Expt. 7 Effect of saline purgative on frog intestine

Expt. 7 Effect of saline purgative on frog intestine
BIOASSAY OF TITANUS ANTI TIOXIN AND DIPTHERIA VACCINE

BIOASSAY OF TITANUS ANTI TIOXIN AND DIPTHERIA VACCINE
Isolation, identification & estimation by Pooja Khanpara 

Isolation, identification & estimation by Pooja Khanpara
Expt. 2 Bioassay of acetylcholine using rat ileum by four point bioassay

Expt. 2 Bioassay of acetylcholine using rat ileum by four point bioassay
Anti-inflammatory activity of drugs using carrageenan induced paw-edema model

Anti-inflammatory activity of drugs using carrageenan induced paw-edema model
Expt. 1 Bioassay of serotonin using rat fundus strip by three point bioassay

Expt. 1 Bioassay of serotonin using rat fundus strip by three point bioassay
Expt. 1 Bioassay of serotonin using rat fundus strip by three point bioassay

Expt. 1 Bioassay of serotonin using rat fundus strip by three point bioassay
Compendial methods for evalution of crude drug and herbal formulation

Compendial methods for evalution of crude drug and herbal formulation
Industrial Production, Estimation and Utilization of the Phytoconstituents

Industrial Production, Estimation and Utilization of the Phytoconstituents
Similar to Validation of factor xa assay for enoxaparin sodium enoxaparin injection
Validation of factor xa assay for nadroparin calcium nadroparin injection

This document describes a chromogenic assay used to quantify the anticoagulant Nadroparin in samples by measuring its inhibitory effect on factor Xa. The assay involves incubating Nadroparin samples with factor Xa and anti-thrombin III, then adding a chromogenic substrate and measuring the absorbance. The absorbance is inversely proportional to the Nadroparin concentration as it inhibits factor Xa activity. The assay kit and procedure are validated for use in quantifying Nadroparin levels in bioequivalence studies.
Validation of factor xa assay for heparin sodium heparin injection

This document summarizes the procedures for using a chromogenic assay kit to quantify heparin levels in samples by measuring heparin's anti-factor Xa activity. The kit utilizes heparin's ability to catalyze the reaction between factor Xa and antithrombin III, which allows the residual factor Xa activity to be measured and used to determine heparin concentration in unknown samples. The document outlines the materials provided, reconstitution procedures, standard and sample preparation steps, assay procedure, and methods for data analysis and interpretation of results.
Validation of factor xa assay for tinzaparin sodium tinzaparin injection

chromogenic assay intended for the quantitative determination in purified solutions by measurement of factor IIa or Xa inhibition activity. The kit can be used for 100 test reactions as per microtiter plate protocol.
Validation of factor xa assay for dalteparin sodium dalteparin injection

chromogenic assay intended for the quantitative determination in purified solutions by measurement of factor IIa or Xa inhibition activity. The kit can be used for 100 test reactions as per microtiter plate protocol.
Validation of Factor IIa assay for enoxaparin sodium or enoxaparin injection

A chromogenic assay intended for the quantitative determination in purified solutions by measurement of factor IIa or Xa inhibition activity. The kit can be used for 100 test reactions as per microtiter plate protocol.
Validation of Factor IIa assay for nadroparin calcium or nadroparin injection

A chromogenic assay intended for the quantitative determination in purified solutions by measurement of factor IIa or Xa inhibition activity. The kit can be used for 100 test reactions as per microtiter plate protocol.
Validation of factor i ia assay for tinzaparin sodium-tinzaparin injection

chromogenic assay intended for the quantitative determination in purified solutions by measurement of factor IIa or Xa inhibition activity. The kit can be used for 100 test reactions as per microtiter plate protocol.
Validation of Factor II and Xa Chromogenic Assays for Dalteparin, Enoxaparin,...

A chromogenic assay intended for the quantitative determination in purified solutions by measurement of factor IIa or Xa inhibition activity. The kit can be used for 100 test reactions as per microtiter plate protocol.
Analysis of analgesics and antipyretics.indu

The document summarizes analytical methods for several analgesics and antipyretics. It discusses classification of analgesics and antipyretics and their mechanisms of action. Specific analytical methods covered include titrimetric, spectrophotometric, chromatographic and colorimetric assays for drugs like aspirin, diclofenac sodium, aceclofenac, ibuprofen, paracetamol, analgin and antipyrine. Gravimetric, colorimetric and polarographic methods are described for the analysis of antipyrine.
How to find antioxidant properties from selected plants

How to find antioxidant properties from selected plantsGovt. Holkar Science College , Indore, M.P., India
This document outlines a study to screen and analyze selected plant species for their antioxidant properties. The objectives are to:
1. Screen 3-4 plant species from forest regions for antioxidant properties.
2. Identify primary and secondary metabolites in plant extracts.
3. Isolate and quantify bioactive antioxidant compounds and determine medicinal value.
4. Compare antioxidant profiles between plant species.
Plants will be extracted using solvent extraction. Phytochemical analysis will test for compounds like alkaloids, flavonoids, tannins. Total phenolic content and flavonoid content will be determined colorimetrically. Antioxidant capacity will be evaluated using DPPH, ABTS, hydroxyMethod development and validation for the estimation of Atorvastatin, Ezitimi...

Method development and validation for the estimation of Atorvastatin, Ezitimibe and Fenofibrate in bulk and pharmaceutical dosage forms by RP-HPLC method
In vitro conservation of Centella asiatica (Linn.)Urban. and Bacopa monnieri ...

In vitro conservation of Centella asiatica (Linn.)Urban. and Bacopa monnieri ...Sugandika Weerasinghe
1) The document describes an in vitro study of Centella asiatica and Bacopa monnieri, two medicinal plants known to have antioxidant properties.
2) Explants from both plants were cultured on MS medium and multiple shoots were successfully induced from nodal explants and leaves.
3) Extracts from Centella asiatica were evaluated for antioxidant activity using DPPH and FRAP assays, and for antifungal activity against Aspergillus niger. The extracts showed free radical scavenging and ferric ion reducing abilities.Method development and validation for the estimation of Allopurinol and alpha...

Method development and validation for the estimation of Allopurinol and alpha lipoic acid by RP-HPLC method
New RP HPLC method for the simultaneous estimation of terbutaline and theophy...

New RP HPLC method for the simultaneous estimation of terbutaline and theophylline in pharmaceutical dosage form
Extraction of Secondary Metabolites from Roots of Acanthus Ilicifolius L and ...

The root extracts of Acanthus ilicifolius L finds a prominent place in folk medicine. In this study, we
extracted alkaloid, flavonoid, tannin and total phenols in benzene, ethyl acetate, acetone, methanol and
ethanol, their antibacterial activity and antioxidant activity was evaluated. The antioxidant activity is executed
by FRAP assay and agar well diffusion method is done to study the antibacterial activity against Enterobacter
aerogenes, Enterobacter cloacae, Escherichia coli, Bacillus subtilis, Staphylococcus aureus and Streptococcus
pyogenes. The antibacterial activity of all the extracts was compared with standard antibiotic gentamicin.
The Minimum Inhibitory Concentration [MIC] was determined by serial dilution method. Alkaloids are rich in
acetone and Flavonoids are high in methanol extracts. The acetone extract showed higher antioxidant activity,
while benzene extract was identified to contain lower antioxidant activity. The extent of inhibition by the root
extracts diverge between the solvents used, among them ethanol extracts exhibited higher level of inhibition
against the gram positive test cultures compared to gram negative test cultures employed. Whereas, the acetone
extracts efficacy is more on gram negative test cultures than the gram positive cultures. The MIC was found to
be between 1mg/100µl to 5mg/100µl. This study gives the source for purification and characterization of
bioactive principles that possess antioxidant and antibacterial action from the root of Acanthus ilicifolius.
Analytical method development and validation for the estimation of aspirin an...

A simple and selective LC method is described for the determination of Aspirin and Omeprazole in tablet dosage forms. Chromatographic separation was achieved on a c18 column using mobile phase consisting of a mixture of 30 volumes of ammonium acetate buffer, 40 volumes of acetonitrile and 30 volumes of Methanol with detection of 233 nm. Linearity was observed in the range 18-42 µg/ml for Aspirin (r2 =0.983) and 6-14 µg /ml for Omeprazole (r2 =0.970) for the amount of drugs estimated by the proposed methods was in good agreement with the label claim. The proposed methods were validated. The accuracy of the methods was assessed by recovery studies at three different levels. Recovery experiments indicated the absence of interference from commonly encountered pharmaceutical additives. The method was found to be precise as indicated by the repeatability analysis, showing %RSD less than 2. All statistical data proves validity of the methods and can be used for routine analysis of pharmaceutical dosage form.
Screening of anticholinesterases

This document describes various in vitro and in vivo screening methods for evaluating anticholinesterase agents. It outlines assays measuring acetylcholinesterase and butyrylcholinesterase inhibition in rat and human tissues. In vivo methods include inhibitory avoidance tests in rodents using step-down, step-through, and uphill avoidance tasks. Other tests involve active avoidance, discrimination learning, and conditioned response tasks to assess effects on memory and learning. The document provides detailed procedures, reagents, and evaluations for each screening method.
In vitro screening methods ach, ne and 5-HT

This document describes in vitro screening methods for acetylcholine, norepinephrine, and serotonin. It discusses methods to estimate acetylcholine-esterase and butyrylcholine-esterase activity. It also describes releasing radioactive acetylcholine from brain slices and measuring its stimulation and inhibition. A novel method is presented for screening treatments for eye strain using rabbit ciliary muscles in a tissue bath.
New RP HPLC method for the simultaneous estimation of rosuvastatin and aspiri...

A simple and selective LC method is described for the determination of Rosuvastatin and Aspirin tablet dosage forms. Chromatographic separation was achieved on a C18 column using mobile phase consisting of A mixture of 60 volumes of 20mM Phosphate buffer pH 3.5: 20 volumes of Acetonitrile and 20 voulmes of Methanol. With detection of 232 nm. Linearity was observed in the range 12-28 µg /ml for Rosuvastatin (r2 =0.9977) and 90-210 µg /ml for Aspirin (r2 =0.9953) for the amount of drugs estimated by the proposed methods was in good agreement with the label claim. The proposed methods were validated. The accuracy of the methods was assessed by recovery studies at three different levels. Recovery experiments indicated the absence of interference from commonly encountered pharmaceutical additives. The method was found to be precise as indicated by the repeatability analysis, showing % RSD less than 2. All statistical data proves validity of the methods and can be used for routine analysis of pharmaceutical dosage form.
Validated RP-HPLC Method for the Determination of Nelaribine in Bulk and Tabl...

A novel, simple and economic reverse phase high performance liquid chromatography (RP-HPLC) method has been developed for the estimation of Nelaribine in bulk and tablet dosage form with greater precision and accuracy. Separation was achieved on Cosmiscil C18 column (150X4.6mm i.d.,5-µm) in isocratic mode using Triflouro acetic acid PH-3.6 buffer and Acetonitrile in the ratio of 90:10(v/v) as mobile phase, pumped in to the column at flow rate of 1.0 mL min-1and the detection of eluent from the column was carried out using variable wavelength UV detector at 248 nm. The total run time was 15 min and the column was maintained at ambient temperature. The retention time of Nelaribine was 4.003 min. The standard curves were linear over the concentration range of 25-150 -µg/ml with R2 0.999 and the LOD and LOQ values for Nelaribine were 0.04 -µg/ml and 0.12 -µg/ml , respectively. The percentage recovery was found to be 101.76 “ 98.72 %, the % RSD was found to be 0.43. The percentage amount of a marketed tablet formulation of Nelaribine was found to be 101.2 %. The method was validated as per ICH guidelines. Validation studies demonstrated that the proposed RP-HPLC method is simple, specific, rapid, reliable and reproducible. Hence the proposed method can be applied for the routine quality control analysis of Nelaribine in bulk and tablet dosage forms. Mrs.P.D.Chaithanya Sudha | Prof.D.Gowri Sankar"Validated RP-HPLC Method for the Determination of Nelaribine in Bulk and Tablet Dosage Form" Published in International Journal of Trend in Scientific Research and Development (ijtsrd), ISSN: 2456-6470, Volume-1 | Issue-4 , June 2017, URL: http://www.ijtsrd.com/papers/ijtsrd181.pdf http://www.ijtsrd.com/pharmacy/analytical-chemistry/181/validated-rp-hplc-method-for-the-determination-of-nelaribine-in-bulk-and-tablet-dosage-form/mrspdchaithanya-sudha
Similar to Validation of factor xa assay for enoxaparin sodium enoxaparin injection (20)
Validation of factor xa assay for nadroparin calcium nadroparin injection

Validation of factor xa assay for nadroparin calcium nadroparin injection
Validation of factor xa assay for heparin sodium heparin injection

Validation of factor xa assay for heparin sodium heparin injection
Validation of factor xa assay for tinzaparin sodium tinzaparin injection

Validation of factor xa assay for tinzaparin sodium tinzaparin injection
Validation of factor xa assay for dalteparin sodium dalteparin injection

Validation of factor xa assay for dalteparin sodium dalteparin injection
Validation of Factor IIa assay for enoxaparin sodium or enoxaparin injection

Validation of Factor IIa assay for enoxaparin sodium or enoxaparin injection
Validation of Factor IIa assay for nadroparin calcium or nadroparin injection

Validation of Factor IIa assay for nadroparin calcium or nadroparin injection
Validation of factor i ia assay for tinzaparin sodium-tinzaparin injection

Validation of factor i ia assay for tinzaparin sodium-tinzaparin injection
Validation of Factor II and Xa Chromogenic Assays for Dalteparin, Enoxaparin,...

Validation of Factor II and Xa Chromogenic Assays for Dalteparin, Enoxaparin,...
How to find antioxidant properties from selected plants

How to find antioxidant properties from selected plants
Method development and validation for the estimation of Atorvastatin, Ezitimi...

Method development and validation for the estimation of Atorvastatin, Ezitimi...
In vitro conservation of Centella asiatica (Linn.)Urban. and Bacopa monnieri ...

In vitro conservation of Centella asiatica (Linn.)Urban. and Bacopa monnieri ...
Method development and validation for the estimation of Allopurinol and alpha...

Method development and validation for the estimation of Allopurinol and alpha...
New RP HPLC method for the simultaneous estimation of terbutaline and theophy...

New RP HPLC method for the simultaneous estimation of terbutaline and theophy...
Extraction of Secondary Metabolites from Roots of Acanthus Ilicifolius L and ...

Extraction of Secondary Metabolites from Roots of Acanthus Ilicifolius L and ...
Analytical method development and validation for the estimation of aspirin an...

Analytical method development and validation for the estimation of aspirin an...
New RP HPLC method for the simultaneous estimation of rosuvastatin and aspiri...

New RP HPLC method for the simultaneous estimation of rosuvastatin and aspiri...
Validated RP-HPLC Method for the Determination of Nelaribine in Bulk and Tabl...

Validated RP-HPLC Method for the Determination of Nelaribine in Bulk and Tabl...
More from krishgen
KRIBIOLISA Drug Monitoring ELISA Trastuzumab

Kribiolisa ELISA kits for drug monitoring of therapeutic drugs like Adalimumab, Trastuzumab, Atezolizumab, Eculizumab, Ranibizumab. Sensitive assays for drug monitoring and immunogenicity. CE marked.
KRIBIOLISA Drug Monitoring ELISA Ranibizumab

Kribiolisa ELISA kits for drug monitoring of therapeutic drugs like Adalimumab, Trastuzumab, Atezolizumab, Eculizumab, Ranibizumab. Sensitive assays for drug monitoring and immunogenicity. CE marked.
KRIBIOLISA Drug Monitoring ELISA Eculizumab

Kribiolisa ELISA kits for drug monitoring of therapeutic drugs like Adalimumab, Trastuzumab, Atezolizumab, Eculizumab, Ranibizumab. Sensitive assays for drug monitoring and immunogenicity. CE marked.
KRIBIOLISA Drug Monitoring ELISA Atezolizumab

Kribiolisa ELISA kits for drug monitoring of therapeutic drugs like Adalimumab, Trastuzumab, Atezolizumab, Eculizumab, Ranibizumab. Sensitive assays for drug monitoring and immunogenicity. CE marked.
KRIBIOLISA Drug Monitoring ELISA 

This document describes KRIBIOLISA ELISA kits for measuring drug and anti-drug antibody levels, including their KRIBIOLISA ADALIMUMAB ELISA and KRIBIOLISA ANTI-ADALIMUMAB ELISA kits. It provides details on the kit catalog numbers, assay type, sample matrix, calibration range, regulatory status, and validation methodology. It also summarizes a study that established a therapeutic drug level range of 3.51-7.00 mg/L for adalimumab treatment of plaque psoriasis.
Validation of anti niv igm capture elisa version#1

NiV is a negative-sense, non-segmented RNA virus that was first isolated from cerebrospinal fluid of human patients and classified in the family Paramyxoviridae under the new genus
Henipavirus. Its genome encodes six structural proteins: the nucleocapsid (N) protein,
phosphoprotein (P), matrix (M) protein, fusion (F) protein, glycoprotein (G), and large (L)
protein.
Nipah virus glycoprotein G has a globular head domain formed of a six-bladed beta sheet propeller, connected via a flexible stalk domain to a transmembrane anchor. The G binds to the cellular receptors ephrin B2 are ephrin B3, mediating viral attachment. Following attachment Nipah Virus glycoprotein G undergoes a conformational change that leads to triggering of glycoprotein F which leads to membrane fusion (Biering et al, 2012).
The Nipah virus glycoprotein G is a recombinant protein expressed in mammalian HEK293 cells. It is presented as a fusion protein with a mouse Fc tag linked to the C-terminus of glycoprotein G, amino acids 71-602.
We established preliminary specifications defining acceptable ranges for the parameters indicated herein below for our Anti Nipah Virus IgM Capture ELISA kit. These parameters were tracked day-to-day, run-to-run, and operator-to-operator, over a schedule defined inhouse.
Recommended assay characteristics included absorbance of a zero concentration standard; factors which describe the calibration for each standard and statistical description of the calibration curve such as coefficient of correlation, slope and/or intercept; and recovery of results on control samples. It is important to be able to relate the specifications for a parameter to expected reliability of the result. Our in-house standard defined was r=0.990.
Validation of bevacizumab elisa ich q2 ver3,0 dt14.03

This document presents a discussion of the characteristics of our KRIBIOLISA™
BEVACIZUMAB ELISA kit considered by us during the validation of this kit in accordance
with ICH Q2 (R1) guidelines. The document is prepared based on tests run in our laboratory
and does not necessarily seek to cover the testing that may be required at user’s end for
registration in, or regulatory submissions. The objective of this validation is to demonstrate
that it is suitable for its intended purpose – detection of Bevacizumab (Avastin)
White Paper : Need for Standardization of Tests for Plant Viruses

The document discusses the need to standardize virus testing procedures for tissue culture plants in India. Currently, each accredited testing laboratory uses its own in-house assays and methods, leading to inconsistent results. The author proposes that the Department of Biotechnology accredit standardized virus testing kits from commercial Indian manufacturers to be used uniformly across all testing laboratories. This would ensure a transparent, reproducible certification process and boost confidence among stakeholders in the tissue culture industry.
rat cytokines general flyer

KRISHGEN is leading manufacturer of ELISA kits for measuring markers in rat serum or plasma. Citations are available for majority of the products and can be viewed on the website.
Fluorescent Assays - KRISHGEN

KRISHGEN is a leading manufacturer of fluroescent based assay kits which can be used on plate readers. With numerous citations for their products, the company is a leading assay and kits manufacturer.
About Krishgen

Krishgen is an Indian biotech company established in 2003 that focuses on developing tools for the life sciences and agriculture industries. It has experienced strong growth, with revenues reaching $5 million in 2011. Krishgen provides products and services related to medical diagnostics, drug discovery research, biopharmaceuticals, and plant/food sciences. It aims to enable breakthroughs in biological research and disease treatment.
About Krishgen

Krishgen is an Indian biotech company established in 2003 that focuses on developing tools for the life sciences and agri industries. It has experienced strong growth, with revenues reaching $5 million in 2011. The company provides products and services related to medical diagnostics, drug discovery research, biopharmaceuticals, and plant/food sciences.
More from krishgen (12)
Validation of anti niv igm capture elisa version#1

Validation of anti niv igm capture elisa version#1
Validation of bevacizumab elisa ich q2 ver3,0 dt14.03

Validation of bevacizumab elisa ich q2 ver3,0 dt14.03
White Paper : Need for Standardization of Tests for Plant Viruses

White Paper : Need for Standardization of Tests for Plant Viruses
Recently uploaded
Integrating Ayurveda into Parkinson’s Management: A Holistic Approach

Explore the benefits of combining Ayurveda with conventional Parkinson's treatments. Learn how a holistic approach can manage symptoms, enhance well-being, and balance body energies. Discover the steps to safely integrate Ayurvedic practices into your Parkinson’s care plan, including expert guidance on diet, herbal remedies, and lifestyle modifications.
8 Surprising Reasons To Meditate 40 Minutes A Day That Can Change Your Life.pptx

8 Surprising Reasons To Meditate 40 Minutes A Day That Can Change Your Life.pptxHolistified Wellness
We’re talking about Vedic Meditation, a form of meditation that has been around for at least 5,000 years. Back then, the people who lived in the Indus Valley, now known as India and Pakistan, practised meditation as a fundamental part of daily life. This knowledge that has given us yoga and Ayurveda, was known as Veda, hence the name Vedic. And though there are some written records, the practice has been passed down verbally from generation to generation.Promoting Wellbeing - Applied Social Psychology - Psychology SuperNotes

A proprietary approach developed by bringing together the best of learning theories from Psychology, design principles from the world of visualization, and pedagogical methods from over a decade of training experience, that enables you to: Learn better, faster!
Phone Us ❤8107221448❤ #ℂall #gIRLS In Dehradun By Dehradun @ℂall @Girls Hotel...

Phone Us ❤8107221448❤ #ℂall #gIRLS In Dehradun By Dehradun @ℂall @Girls Hotel...chandankumarsmartiso
Phone Us ❤8107221448❤ #ℂall #gIRLS In Dehradun By Dehradun @ℂall @Girls Hotel With 100% Satisfaction
#cALL# #gIRLS# In Dehradun ꧁❤8107221448❤꧂#cALL# #gIRLS# Service In Dehradun W...

#cALL# #gIRLS# In Dehradun ꧁❤8107221448❤꧂#cALL# #gIRLS# Service In Dehradun W...chandankumarsmartiso
#cALL# #gIRLS# In Dehradun ꧁❤8107221448❤꧂#cALL# #gIRLS# Service In Dehradun Women Seeking Service
Dehradun #ℂall #gIRLS Oyo Hotel 8107221448 #ℂall #gIRL in Dehradun

Dehradun #ℂall #gIRLS Oyo Hotel 8107221448 #ℂall #gIRL in Dehradun
Top Effective Soaps for Fungal Skin Infections in India

Swisschem Dermacare has mentioned the List of The Best Antifungal Soap In India 2022. All of these soaps are trusted by various Dermatology Experts.
Novas diretrizes da OMS para os cuidados perinatais de mais qualidade

Novas diretrizes da OMS para os cuidados perinatais de mais qualidadeProf. Marcus Renato de Carvalho
Recomendações da OMS sobre cuidados maternos e neonatais para uma experiência pós-natal positiva.
Em consonância com os ODS – Objetivos do Desenvolvimento Sustentável e a Estratégia Global para a Saúde das Mulheres, Crianças e Adolescentes, e aplicando uma abordagem baseada nos direitos humanos, os esforços de cuidados pós-natais devem expandir-se para além da cobertura e da simples sobrevivência, de modo a incluir cuidados de qualidade.
Estas diretrizes visam melhorar a qualidade dos cuidados pós-natais essenciais e de rotina prestados às mulheres e aos recém-nascidos, com o objetivo final de melhorar a saúde e o bem-estar materno e neonatal.
Uma “experiência pós-natal positiva” é um resultado importante para todas as mulheres que dão à luz e para os seus recém-nascidos, estabelecendo as bases para a melhoria da saúde e do bem-estar a curto e longo prazo. Uma experiência pós-natal positiva é definida como aquela em que as mulheres, pessoas que gestam, os recém-nascidos, os casais, os pais, os cuidadores e as famílias recebem informação consistente, garantia e apoio de profissionais de saúde motivados; e onde um sistema de saúde flexível e com recursos reconheça as necessidades das mulheres e dos bebês e respeite o seu contexto cultural.
Estas diretrizes consolidadas apresentam algumas recomendações novas e já bem fundamentadas sobre cuidados pós-natais de rotina para mulheres e neonatos que recebem cuidados no pós-parto em unidades de saúde ou na comunidade, independentemente dos recursos disponíveis.
É fornecido um conjunto abrangente de recomendações para cuidados durante o período puerperal, com ênfase nos cuidados essenciais que todas as mulheres e recém-nascidos devem receber, e com a devida atenção à qualidade dos cuidados; isto é, a entrega e a experiência do cuidado recebido. Estas diretrizes atualizam e ampliam as recomendações da OMS de 2014 sobre cuidados pós-natais da mãe e do recém-nascido e complementam as atuais diretrizes da OMS sobre a gestão de complicações pós-natais.
O estabelecimento da amamentação e o manejo das principais intercorrências é contemplada.
Recomendamos muito.
Vamos discutir essas recomendações no nosso curso de pós-graduação em Aleitamento no Instituto Ciclos.
Esta publicação só está disponível em inglês até o momento.
Prof. Marcus Renato de Carvalho
www.agostodourado.com
How STIs Influence the Development of Pelvic Inflammatory Disease.pptx

STIs may cause PID. For the both disease, herbal medicine Fuyan Pill can be a solution.
REGULATION FOR COMBINATION PRODUCTS AND MEDICAL DEVICES.pptx

It includes regulation of combination products and medical devices. FDA and industry liaisons.
Identification and nursing management of congenital malformations .pptx

Identification and nursing management of congenital malformations .pptxMGM SCHOOL/COLLEGE OF NURSING
Identification and nursing management of congenital malformations Best Ayurvedic medicine for Gas and Indigestion

Here is the updated list of Top Best Ayurvedic medicine for Gas and Indigestion and those are Gas-O-Go Syp for Dyspepsia | Lavizyme Syrup for Acidity | Yumzyme Hepatoprotective Capsules etc
Cell Therapy Expansion and Challenges in Autoimmune Disease

There is increasing confidence that cell therapies will soon play a role in the treatment of autoimmune disorders, but the extent of this impact remains to be seen. Early readouts on autologous CAR-Ts in lupus are encouraging, but manufacturing and cost limitations are likely to restrict access to highly refractory patients. Allogeneic CAR-Ts have the potential to broaden access to earlier lines of treatment due to their inherent cost benefits, however they will need to demonstrate comparable or improved efficacy to established modalities.
In addition to infrastructure and capacity constraints, CAR-Ts face a very different risk-benefit dynamic in autoimmune compared to oncology, highlighting the need for tolerable therapies with low adverse event risk. CAR-NK and Treg-based therapies are also being developed in certain autoimmune disorders and may demonstrate favorable safety profiles. Several novel non-cell therapies such as bispecific antibodies, nanobodies, and RNAi drugs, may also offer future alternative competitive solutions with variable value propositions.
Widespread adoption of cell therapies will not only require strong efficacy and safety data, but also adapted pricing and access strategies. At oncology-based price points, CAR-Ts are unlikely to achieve broad market access in autoimmune disorders, with eligible patient populations that are potentially orders of magnitude greater than the number of currently addressable cancer patients. Developers have made strides towards reducing cell therapy COGS while improving manufacturing efficiency, but payors will inevitably restrict access until more sustainable pricing is achieved.
Despite these headwinds, industry leaders and investors remain confident that cell therapies are poised to address significant unmet need in patients suffering from autoimmune disorders. However, the extent of this impact on the treatment landscape remains to be seen, as the industry rapidly approaches an inflection point.
Local Advanced Lung Cancer: Artificial Intelligence, Synergetics, Complex Sys...

Overall life span (LS) was 1671.7±1721.6 days and cumulative 5YS reached 62.4%, 10 years – 50.4%, 20 years – 44.6%. 94 LCP lived more than 5 years without cancer (LS=2958.6±1723.6 days), 22 – more than 10 years (LS=5571±1841.8 days). 67 LCP died because of LC (LS=471.9±344 days). AT significantly improved 5YS (68% vs. 53.7%) (P=0.028 by log-rank test). Cox modeling displayed that 5YS of LCP significantly depended on: N0-N12, T3-4, blood cell circuit, cell ratio factors (ratio between cancer cells-CC and blood cells subpopulations), LC cell dynamics, recalcification time, heparin tolerance, prothrombin index, protein, AT, procedure type (P=0.000-0.031). Neural networks, genetic algorithm selection and bootstrap simulation revealed relationships between 5YS and N0-12 (rank=1), thrombocytes/CC (rank=2), segmented neutrophils/CC (3), eosinophils/CC (4), erythrocytes/CC (5), healthy cells/CC (6), lymphocytes/CC (7), stick neutrophils/CC (8), leucocytes/CC (9), monocytes/CC (10). Correct prediction of 5YS was 100% by neural networks computing (error=0.000; area under ROC curve=1.0).
The Best Ayurvedic Antacid Tablets in India

Treat the symptoms of indigestion, heartburn and stomach reflux with the 10 Best Ayurvedic Antacid Tablets in India.
Recently uploaded (20)
Integrating Ayurveda into Parkinson’s Management: A Holistic Approach

Integrating Ayurveda into Parkinson’s Management: A Holistic Approach
8 Surprising Reasons To Meditate 40 Minutes A Day That Can Change Your Life.pptx

8 Surprising Reasons To Meditate 40 Minutes A Day That Can Change Your Life.pptx
Ear and its clinical correlations By Dr. Rabia Inam Gandapore.pptx

Ear and its clinical correlations By Dr. Rabia Inam Gandapore.pptx
Promoting Wellbeing - Applied Social Psychology - Psychology SuperNotes

Promoting Wellbeing - Applied Social Psychology - Psychology SuperNotes
Phone Us ❤8107221448❤ #ℂall #gIRLS In Dehradun By Dehradun @ℂall @Girls Hotel...

Phone Us ❤8107221448❤ #ℂall #gIRLS In Dehradun By Dehradun @ℂall @Girls Hotel...
#cALL# #gIRLS# In Dehradun ꧁❤8107221448❤꧂#cALL# #gIRLS# Service In Dehradun W...

#cALL# #gIRLS# In Dehradun ꧁❤8107221448❤꧂#cALL# #gIRLS# Service In Dehradun W...
Dehradun #ℂall #gIRLS Oyo Hotel 8107221448 #ℂall #gIRL in Dehradun

Dehradun #ℂall #gIRLS Oyo Hotel 8107221448 #ℂall #gIRL in Dehradun
Top Effective Soaps for Fungal Skin Infections in India

Top Effective Soaps for Fungal Skin Infections in India
Novas diretrizes da OMS para os cuidados perinatais de mais qualidade

Novas diretrizes da OMS para os cuidados perinatais de mais qualidade
How STIs Influence the Development of Pelvic Inflammatory Disease.pptx

How STIs Influence the Development of Pelvic Inflammatory Disease.pptx
Vestibulocochlear Nerve by Dr. Rabia Inam Gandapore.pptx

Vestibulocochlear Nerve by Dr. Rabia Inam Gandapore.pptx
Muscles of Mastication by Dr. Rabia Inam Gandapore.pptx

Muscles of Mastication by Dr. Rabia Inam Gandapore.pptx
REGULATION FOR COMBINATION PRODUCTS AND MEDICAL DEVICES.pptx

REGULATION FOR COMBINATION PRODUCTS AND MEDICAL DEVICES.pptx
Identification and nursing management of congenital malformations .pptx

Identification and nursing management of congenital malformations .pptx
Cell Therapy Expansion and Challenges in Autoimmune Disease

Cell Therapy Expansion and Challenges in Autoimmune Disease
Local Advanced Lung Cancer: Artificial Intelligence, Synergetics, Complex Sys...

Local Advanced Lung Cancer: Artificial Intelligence, Synergetics, Complex Sys...
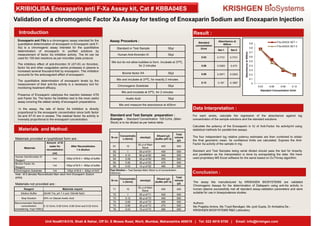
Validation of factor xa assay for enoxaparin sodium enoxaparin injection
- 1. Enoxaparin anti FXa is a chromogenic assay intended for the quantitative determination of enoxaparin in Enoxaparin (anti F- Xa) is a chromogenic assay intended for the quantitative determination of enoxaparin in purified solutions by measurement of factor Xa inhibition activity. The kit can be used for 100 test reactions as per microtiter plate protocol. The inhibitory effect of anti-thrombin III (AT-III) on thrombin, factor Xa and other coagulation serine proteases in plasma is increased several thousand-fold by enoxaparin. This inhibition accounts for the anticoagulant effect of enoxaparin. The quantitative determination of enoxaparin levels by the measurement of their anti-Xa activity is a necessary tool for monitoring treatment efficacy. Presence of Enoxaparin catalyzes the reaction between ATlll and Factor Xa. The factor Xa inhibition test is the most useful assay covering the widest variety of enoxaparin preparations. In the assay, the rate of factor Xa inhibition is directly proportional to the enoxaparin concentration since both factor Xa and AT-III are in excess. The residual factor Xa activity is inversely proportional to the enoxaparin concentration. Introduction Materials and Method: Result : Data Interpretation : Unit Nos#318/319, Shah & Nahar, Off Dr. E Moses Road, Worli, Mumbai, Maharashtra 400018 | Tel: 022 4919 8700 | Email: info@krishgen.com Materials not provided are : Reagent Materials require Dilution Buffer 20mM Tris, pH 7.4 and 150mM NaCl Stop Solution 20% v/v Glacial Acetic Acid Recommended Standard concentration (considering 1mg=100IU) 0.12 IU/ml, 0.09 IU/ml, 0.06 IU/ml and 0.03 IU/ml. Assay Procedure : Standard or Test Sample 50µl Human Anti-thrombin III 50µl Mix but do not allow bubbles to form. Incubate at 37⁰C, for 2 minutes Bovine factor XA 50µl Mix and incubate at 37⁰C, for exactly 2 minutes Chromogenic Substrate 50µl Mix and incubate at 37⁰C, for 2 minutes Acetic Acid 50µl Mix and measure the absorbance at 405nm For each series, calculate the regression of the absorbance against log concentration of the sample solutions and the standard solutions. Calculate the potency of the Enoxaparin in IU of Anti-Factor Xa activity/ml using statistical methods for parallel-line assays. The four independent log relative potency estimates are then combined to obtain the final geometric mean. Its confidence limits are calculated. Express the Anti- Factor Xa activity of the sample in mg. Standard and Test Samples being serial diluted should pass the test for linearity and parallelism as the interpretation is done by extrapolating the data. We have used proprietary MS Excel software for the same based on DJ Finney algorithm. Conclusion : The assay kits manufactured by KRISHGEN BIOSYSTEMS are validated Chromogenic Assays for the determination of Dalteparin using anti-Xa activity in human plasma successfully met all standard assay-validation parameters and were suitable for use in bioequivalence studies. Authors: Ms Prajakta Ambre, Ms Trupti Bendigeri, Ms Jyoti Gupta, Dr Amitabha De - KRISHGEN BIOSYSTEMS R&D Laboratory Materials provided in lyophilized form are : Materials Amount of DI water for reconstitution (ml) After Reconstitution 1:4 dilution Human Anti-thrombin III Reagent 1ml 100µl of M.S + 400µl of buffer Bovine Factor Xa Reagent 1ml 100µl of M.S + 400µl of buffer Chromogenic Substrate 1ml 100µl of M.S + 400µl of H2O Note : M.S denotes Reconstituted Main stock from Enoxaparin Sodium EPRS Sr.no Concentratio n (IU/ml) stock(µl) Diluent (µl) (buffer pH7.4) Total volume (µl) S1 10 50 µl of Main Stock 450 500 S2 1 50 µl of S1 450 500 S3 0.12 60 µl of S2 440 500 S4 0.09 45 µl of S2 455 500 S5 0.06 30 µl of S2 470 500 S6 0.03 15 µl of S2 485 500 Test Dilution – Test Sample Main Stock is of concentration 100IU/mL Sr.no Concentratio n (IU/ml) stock(µl) Diluent (µl) (buffer pH7.4) Total volume (µl) T1 10 50 µl of Main Stock 450 500 T2 1 50 µl of T1 450 500 T3 0.12 60 µl of T2 440 500 T4 0.09 45 µl of T2 455 500 T5 0.06 30 µl of T2 470 500 T6 0.03 15 µl of T2 485 500 Standard and Test Sample preparation : Example - Standard Concentration 100 IU/mL (Main Stock) is to be diluted as per below table: Standard concentration IU/ml Absorbance at 405nm Set-1 Set-2 0.03 0.7731 0.7731 0.06 0.5363 0.473 0.09 0.2971 0.3303 0.12 0.197 0.1897 0 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 0.03 0.06 0.09 0.12 Absorbanceat405nm Standard Concentration IU/ml FXa-ENOX SET-1 FXa-ENOX SET-2 KRIBIOLISA Enoxaparin anti F-Xa Assay kit, Cat # KBBA04ES