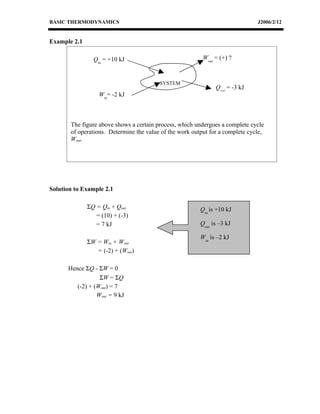
The document provides an overview of basic thermodynamics concepts including:
- Defining systems, boundaries, surroundings, open and closed systems
- Explaining properties, states, and processes
- Stating the first law of thermodynamics that total energy is conserved
- Describing the differences between work and heat transfer
- Defining internal energy as the sum of all energy stored within a system