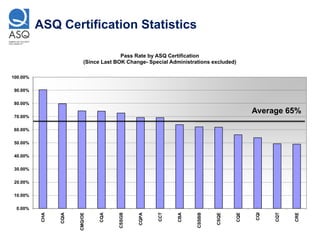
This document provides tips to avoid common mistakes when preparing for and taking ASQ certification exams. It outlines mistakes like overconfidence, poor planning, lack of experience in key areas, and not properly managing time. The document explains strategies like thoroughly reviewing the body of knowledge, using practice exams to identify weaknesses, and adhering to the 30-60-90 second rule during exams. Checklists are provided to ensure all materials are prepared and organized prior to taking an ASQ certification exam.