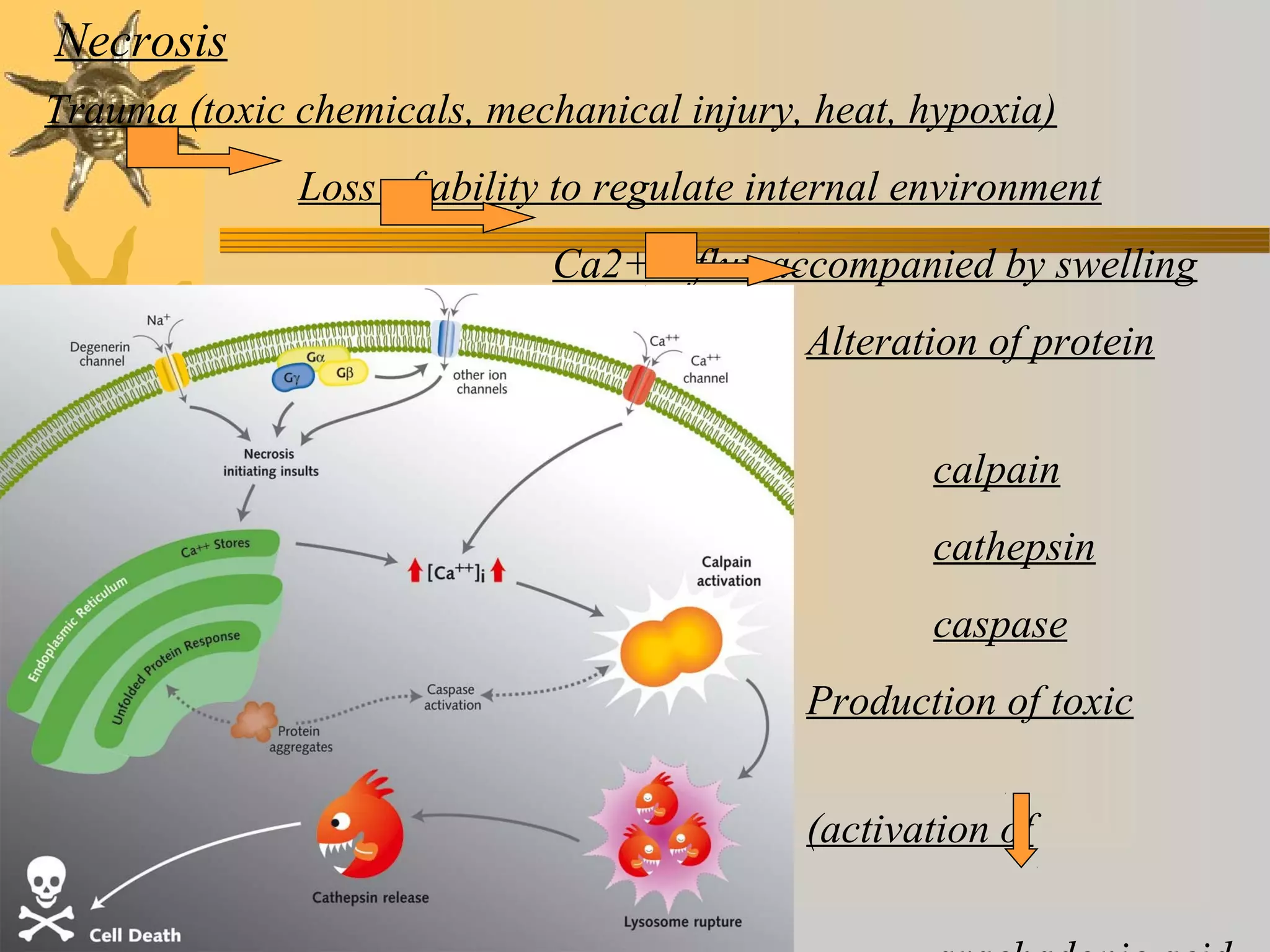
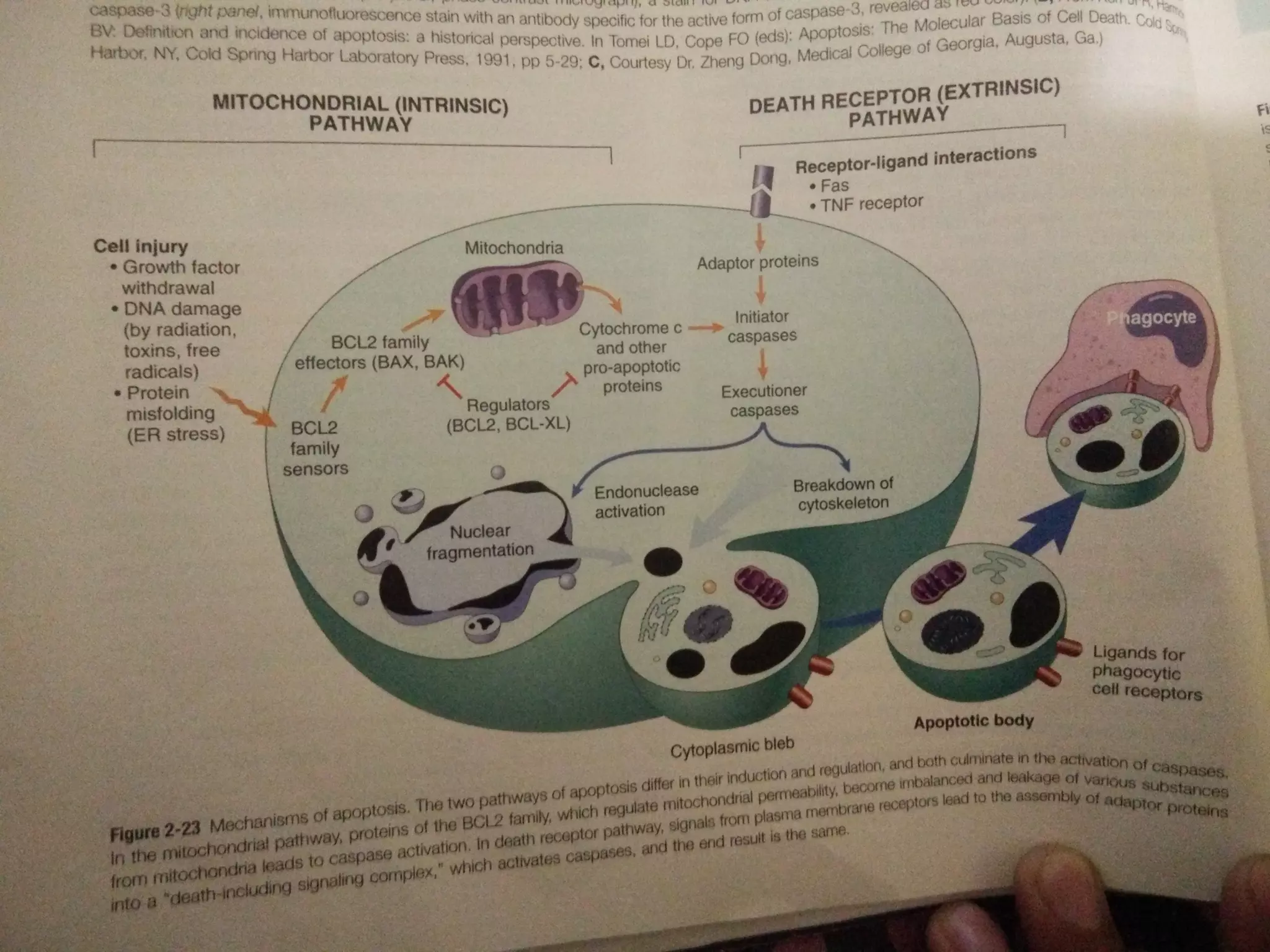
Oxidative stress occurs due to an accumulation of reactive oxygen species (ROS) generated during normal metabolism. ROS can damage cells by reacting with and modifying proteins, lipids, DNA, and other molecules. Theories of aging propose that oxidative stress and the resulting damage contributed to aging over time. While cells have antioxidant defenses against ROS, these defenses decline with age. Emerging evidence suggests oxidative stress plays a role in aging and age-related diseases such as Alzheimer's disease.