Lung Drug Delivery Systems
- 2. Introduction Anatomy of lungs Advantages and disadvantages Mechanism of particle deposition in the airways Different systems used to target lungs Different dosage forms Marketed products References CPS,IST,JNTUH 12/18/2013 2
- 3. There is a continuing need for development of new treatments for lung disease. Basic scientific investigations are identifying novel targets for the development of new approaches to therapy of a range of respiratory conditions. CPS,IST,JNTUH 12/18/2013 3
- 4. The large surface area, good vascularization, immense capacity for solute exchange and ultrathinness of the alveolar epithelium are unique features of the lung that can facilitate systemic delivery via pulmonary administration of peptides and proteins. Physical and biochemical barriers, lack of optimal dosage forms and delivery devices limit the systemic delivery of biotherapeutic agents by inhalation. CPS,IST,JNTUH 12/18/2013 4
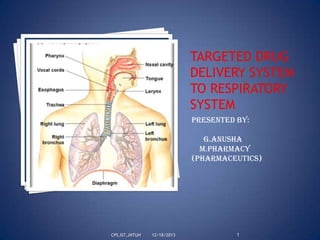
- 5. Anatomy and Physiology of the Respiratory System…: Respiratory system of man consists Upper Respiratory Tract: Consists of nose, nasal passages, paranasal sinuses, mouth, eustachian tube, the pharynx to the oesophagus, the laryinx and trachea Lower Respiratory Tract: Consists of lungs (both air passage ways and respiratory units) Lower Respiratory Tract: The Lung Human lung comprises of left and right lung, are divided into slightly unequal proportions. Each lung is supplied by a major branch of the bronchial tree. The tissue substance of a lung includes air passages, alveoli, blood vessels, connective tissues, lymphatic vessels and nerves CPS,IST,JNTUH 12/18/2013 5
- 8. Improve efficiency. Reduced unwanted systemic side effects. Large surface area for absorption. Thin alveolar epithelium permitting rapid absorption. Absence of first pass metabolism. Rapid onset of action. High availability. It requires small and fraction of oral dose and low concentration in the systemic circulation is associated with reduced systemic side effects. CPS,IST,JNTUH 12/18/2013 8
- 9. Oropharyngeal deposition gives local side effect. Patient may have difficulty using the pulmonary drug devices correctly Drug absorption may be limited by the physical barrier of the mucus layer. Various factors affect the reproducibility on drug delivery on the lungs, including physiological and pharmaceutical barrier. The lungs are not only accessible surface for drug delivery complex but also delivery devices are required to target drug delivery CPS,IST,JNTUH 12/18/2013 9
- 10. Deposition of drug/aerosol in the airways depends on four factors: i. The physico- chemical properties of drug ii. The formulation iii. The delivery device iv. The patient (breathing pattern and clinical status) The particle size of aerosol is usually standardized by calculation of its “ aerodynamic diameter ” to deliver particles to different areas of lung. CPS,IST,JNTUH 12/18/2013 10
- 11. DEFINITION: The aero dynamic diameter of a particle is the diameter of a unit density sphere (1 gm/cm 3 ) having the same terminal settling velocity (in still air) as the particle under consideration. It is derived from stoke’s law D a = ( ρ p / ρ 0 ) 1/2 D g where, ρ p is the particle density, ρ 0 is the standard particle density (1 gm/cm 3 ) D g is the geometric diameter of the particle CPS,IST,JNTUH 12/18/2013 11
- 13. Lung deposition: - Occurs mainly by 3 mechanisms 1) inertial impaction : Where a bifurcation occurs in the respiratory tract, the air stream changes direction and particles with in the air stream having, sufficiently high momentum, will impact on the airway walls rather than follow the changing air stream Particles > 5 µ m and particularly > 10 µ m are deposited by this mechanism CPS,IST,JNTUH 12/18/2013 13
- 14. 2) Gravitational sedimentation: As the remaining small particles move on to the central lung the air velocity gradually decreases to much lower values and the force of gravity becomes important Particles 1-5 um are deposited From stoke’s law particles settling under gravity will attain constant terminal settling velocity S Ut = ρ gd 2 /18 ŋ Thus, gravitational sedimentation of an Inhaled particle is dependent on its size and density in addition to its residence time in the airways 3) Brownian motion : The finest particles enter the periphery of the lung where they can contact with the walls of the airways as the result of Brownian motion (particle diffusion) smaller than 0.5 um are deposited The particle diffusion is inversly proportional to particle size. Other methods include interception and electrostatic attraction CPS,IST,JNTUH 12/18/2013 14
- 16. Controlled drug delivery to lungs: Microsphere Dendrimer Liposome Nanoparticles Gene delivery using polymeric nano-carriers Peptide-Mediated delivery CPS,IST,JNTUH 12/18/2013 16
- 17. LIPOSOMES: Liposome can also be produced with a wide range of size and will incorporate both hydrophilic and lipophilic drugs. The drug carrying capacity, release rate and deposition of liposome in the lungs is dependent upon lipid composition, size, and charge and drug/lipid ratio. Most liposomal carrier–drug systems have been investigated in aqueous systems using nebulisation. DPI liposomal formulations have been produced by lyophilisation followed by milling or spray drying. Encapsulation of drugs in liposome delivered to the lungs results in increased residence time and/or a decrease in toxic side effects. CPS,IST,JNTUH 12/18/2013 17
- 18. Microparticles: Microparticles are hollow spherical particles in which drugs encapsulated. Microparticle are in the 0.1–500μm range. Microparticles are physically and chemically more stable than liposomes , allowing higher loading of drugs. synthetic polymers - polylactic acid (PLA) -polylactic -co-glycolic acid Natural polymers - albumin, gelatin, chitosan and dextran Coating can be used to alter the properties in vivo. Such as mucoadhesive polymers - chitosan and hydroxypropylcellulose increase the residence time of peptide drug carriers in the lungs. CPS,IST,JNTUH 12/18/2013 18
- 19. Micelles: Micelles: Drugs can be trapped in the core of a micelle and transported at concentrations even greater than their intrinsic water solubility. A hydrophilic shell can form around the micelle, effectively protecting the contents. The advantages of polymeric micelles include better stability than surfactant micelles, ability to solubilize an adequate amount of hydrophobic drugs, prolonged circulation times in vivo, and capability to accumulate in the target organs. CPS,IST,JNTUH 12/18/2013 19
- 20. Nanoparticles: Nanoparticles used for dispersed liquid droplet dosage forms such as metered dose inhalers and nebulizers, and dry powder formulations. constituted of polymers or lipids and drugs bound either at the surface of the particles or encapsulated into the vector. Traditional techniques such as spray drying and grinding, and more recent advances in supercritical fluid extraction, precipitation, and solvent extraction have been employed to produce nanoparticle formulations for pulmonary delivery CPS,IST,JNTUH 12/18/2013 20
- 21. Solid lipid nanoparticles (SLN): Three possible loadings of drugs can be envisaged: ( i ) Dispersion of drugs into the particle; (ii) core-membrane model containing a membrane improved with the drug; (iii) core-membrane model containing a core improved with the drug. In vitro release studies have showed that an encapsulated drug into solid lipid nanoparticles can diffuse during a period of time ranging from 5 to 7 weeks. Drugs like prednisolone , diazepam and camptotecin have been incorporated into SLN for pulmonary applications. CPS,IST,JNTUH 12/18/2013 21
- 22. Dendrimers are polymers, which have hyperbranched structures, with layered architectures. Dendrimers are globular repeatedly branched macromolecules that exhibit controlled patterns of branching with multiple arms extending from a central core. They are used in drug delivery and imaging at a size typically ranging from 10 to 100 nm in diameter. Encapsulation of therapeutic agents, particularly peptide therapeutics. CPS,IST,JNTUH 12/18/2013 22
- 23. Microspheres : Microspheres Both targeted and sustained drug release Hydrophilic and lipophilic drugs can be entrapped or incorporated Albumin microspheres: These are biodegradable colloidal particles that can be prepared by either physical denaturation or chemical cross-linking of albumin droplets. The coating of albumin MS with surfactants could decrease the interaction with mucus layer and possibly increase the deposition in the lower airways Particle size range between 1.94 ± 1.47 and 3.42 ±1.51 μm have been prepared using a high-speed homogenization and heat denaturation process. Poly ( glycolide and/or lactide ) (PGL) microspheres: These are prepared by the polymerization of lactide and/or glycolide monomers via polyester linkages, the hydrolysis of which will result in the breakdown of the polymers to produce non-toxic substances. CPS,IST,JNTUH 12/18/2013 23
- 24. Metered Dry dose inhalers powder inhalers Nebulizers. CPS,IST,JNTUH 12/18/2013 24
- 25. Metered dose inhalers…: In MDIs, drug is either dissolved or suspended in a liquid propellent mixture together with other excipients, including surfactants, lubricants for the valve mechanism and cosolvents A predetermined dose is released as a spray on actuatation metering valve After actuating the canister, expansion of the propellent when exposed to atmospheric pressure aerosolizes the drug. This active process consumes heat and thus cools both aerosol and canister As it travels through the air, the aerosol warms and the propellent evaporates, reducing the particle size to the desirable range The high speed gas flow helps to break up the liquid into a fine spray of droplets CPS,IST,JNTUH 12/18/2013 25
- 26. 1) Containers: Aluminium container with a capacity of 10-30ml 2) Propellents: Chloro Fluoro Carbons (CFCs): Trichlorofluoro methane (CFC-11), dichloro difluro methane (CFC-12) and dichloro tetrafluro methane (CFC-114) Hydrofluoro Alkanes(HFAs) :Trifluro Monofluro Ethane (HFA-134a) & Hectafluoro Propane (HFA-227) These are poor solvents for the surfactants. Ethonal is used to dissolve the surfactants CPS,IST,JNTUH 12/18/2013 26
- 27. Metering valve: Reproducible delivery of small volume (25-100 μ l) of product Metering valve in MDIs are used in the inverted position Depression of the valve stem allows the contents of the metering chamber to be discharged through the orifice in the valve stem and made available to the patient After actuation the metering chamber refills with liquid from the bulk and is ready to dispense the next dose To prevent undesirable layering of medication the canister should be shaken between each puff CPS,IST,JNTUH 12/18/2013 27
- 28. Spacers and breath actuated MDIs: Spacers are positioned between the MDIs and the patient The dose from an MDI is discharged directly into the reservoir prior to the inhalation Disadvantage of spacers is they may cumbersome The breath actuated device overcomes the coordination problem of conventional MDI with out adding bulk to the device However a substantial inspiratory flow rate is required for its operation CPS,IST,JNTUH 12/18/2013 28
- 29. Types of medicines are available as MDIs, including: bronchodilators (ProAir HFS, Proventil HFA, Ventolin HFA, Maxair, or Alupent) inhaled steroids (Azmacort, Flovent, Pulmicort, Qvar) combination of long-acting bronchodilator and inhaled steroid ( ADVAIR HFA, Symbicort) cromolyn (Intal) nedocromil (Tilade) ipratropium bromide (Atrovent). CPS,IST,JNTUH 12/18/2013 29
- 30. A number of metered dose inhalers,including AERX, AERODOSE,RESPIMAT has been developed. A.AERE B.RESPIMAT C.AERODOSE CPS,IST,JNTUH 12/18/2013 30
- 31. Dry Powder Inhaler…: The drug is inhaled as cloud of fine particles The drug is either preloaded in an inhalation device or filled into hard gelatin capsules or foil glister discs, which are loaded into a device prior to use DPIs have several advantages over MDIs propellant free and do not contain excipients, other than carrier they are breath actuated, avoiding the problems of inhalation/actuation coordination encountered with MDIs. Less potential for formulation problems CPS,IST,JNTUH 12/18/2013 31
- 32. Disadvantages of DPIs: Liberation of the powder from the device and the deaggregation of the particles are limited by the patients ability to inhale, which in the case of respiratory disease may be impaired An increase in turbulent airflow created by an increase in inhaled air velocity increases the de aggregation of emerging particles, but also increases the potential for inertial impaction in the upper airways and throat, so a compromise has to be found DPIs are exposed to ambient atmospheric conditions, which may reduce formulation stability DPIs are generally less efficient at drug delivery than MDIs CPS,IST,JNTUH 12/18/2013 32
- 33. Formulating DPI : To produce particles of suitable size, drug powders are usually micronized The high energy powders produced have poor flow properties The flowability of powder affected by physical properties To improve the flow property, poorly flowing drug particles are generally mixed with large carrier particles (usually 30-50um) of an excipient, usually lactose Once liberated from the device, the turbulent airflow generated with in the inhalation device should be sufficient for the de aggregation of the drug or carrier aggregates CPS,IST,JNTUH 12/18/2013 33
- 34. Unit Dose Devices with drug in hard gelatin capsule The first DPI device developed was the spinhaler for the delivery of sodium chromoglycate. Each dose, contained in a hard gelatin capsules, is loaded individually into the device. The capsule, placed in a loose fitting rotor, is pierced by two metal needles an either side of capsule. Inhaled air flow through the device cause a turbo vibratory air pattern as the rotor rotates rapidly, resulting in the powder being dispersed through the capsule walls and out through the perforation into the air. A minimum air flow rate of 3540ltrs per minute through the device is required to produce adequate vibrations through the rotor. CPS,IST,JNTUH spinhaler 12/18/2013 34
- 35. ROTAHALER: Another unit dose DPI is the rota haler , which is a simple two piece device The gelatin capsule is inserted into an orifice at the rear of the device and when the two sections are rotated a fin on the inner barrel pulls the two halves of the capsule apart During inhalation, the freed half of the capsule spins, dispersing its contents, which are inhaled through the mouth piece Others include the aerohaler and cyclohaler CPS,IST,JNTUH 12/18/2013 35
- 36. Turbuhaler , has overcome the need for both a carrier and loading of individual doses The device contains a large number of doses of undiluted, loosely aggregated micronized drug, which is stored in reservoir from which it flows onto rotating disc in the dosing unit The fine holes in the disc are filled and excess drug is removed by scrapers As the rotating disc is turned by moving a turning grip back and forth, one metered dose is presented to the inhalation channel A dose indicator is incorporated CPS,IST,JNTUH 12/18/2013 36
- 37. Nebulizers These are applied to aerosolize drug solutions or suspension Two types of nebulizers are there i. Jet nebulizer ii. ultrasonic nebulizer CPS,IST,JNTUH 12/18/2013 37
- 38. JET NEBULIZER: unctions by the Bernoulli principle Compressed gas (air or oxygen) passes through a narrow orifice creating an area of low pressure at the outlet of the adjacent liquid feed tube This results in drug solution being drawn up from the fluid reservoir and shattered into droplets in the gas CPS,IST,JNTUH 12/18/2013 38
- 39. Ultra sonic nebulizer: It uses a piezoelectric crystal vibrating at the high frequency usually 1-3 MHz To generate a fountain of liquid in the Nebulizer chamber The higher the frequency, the smaller the droplet produced disadvantages: Time consuming Inefficient Large amount of drug wastage, CPS,IST,JNTUH 12/18/2013 39
- 40. Formulation of Nebulizer fluids: These are formulated in water, occasionally with the addition of co solvents and with the addition of surfactants for suspension formulations Stabilizers such as anti oxidants and preservatives may also be included The physical property of drug formulations may have an effect on nebulization rates and particle size The viscosity, ionic strength, osmolarity, pH and surface tension may prevent the nebulization of some formulations CPS,IST,JNTUH 12/18/2013 40
- 41. Application of pulmonary drug delivery in Asthma and COPD. Recent role pulmonary delivery in Patients on ventilators Pulmonary delivery in cystic fibrosis In migraine Angina pectoris Recent use of pulmonary drug delivery in Transplantation In emphysema In Pulmonary arterial hypertension In acute lung injury CPS,IST,JNTUH 12/18/2013 41
- 43. REFERENCES: •Wiley VCH verlag GmbH. Gilbert S. Banker and Christopher T. Rhodes, Modern Pharmaceutics, Second edition, Volume 40, •Leon Lachman, Herbert A. Lieber Man, Juseph L. Kanig, M.E. Aulton, Pharmaceutics, The science of Dossage form design. Second edition, 2002. •Remington, The Science and Practice of Pharmacy, 21st edition, •Volume – I, published by Wolters Kluwer health (India) Pvt. Ltd. New Delhi. S.P. Vyas and Roop K. Khar, • Yie W. Chien, Kenneth S.E. Su, Shyi – Feu Chang. Nasal Systemic Drug Delivery, Marcel Dekker, INC Volume •The controlled delivery of drugs to the lung, International journal of pharmaceutics 124(1995) 149-164. CPS,IST,JNTUH 12/18/2013 43