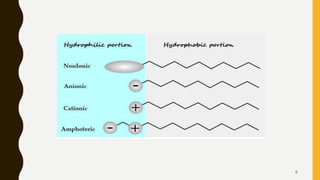
This document discusses surfactants, their classification and applications in cosmetics and cosmeceuticals. Surfactants are amphipathic molecules with a hydrophilic head and hydrophobic tail that lower surface tension. They are classified as anionic, cationic, non-ionic or amphoteric based on the nature of the hydrophilic group. Common applications include use as emulsifying agents, for foaming and cleansing purposes like in shampoos, and as wetting agents. Anionic, cationic, non-ionic and amphoteric surfactants are used for different purposes depending on their properties like detergency and foaming ability or mildness.