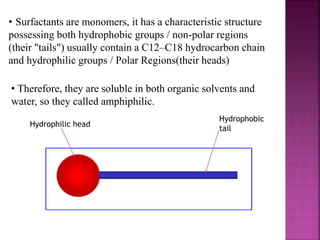
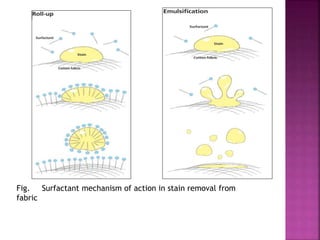
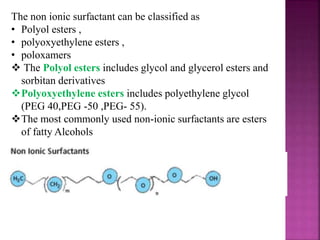
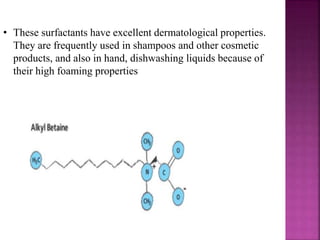
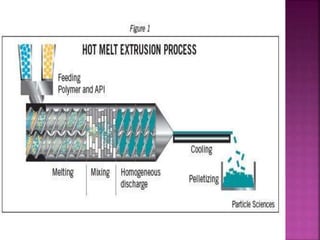
This document discusses various techniques for enhancing drug solubility. It begins with an introduction to factors affecting drug solubility and processes of solubilization. Then it describes techniques such as co-solvency, use of surfactants, complexation, and solid state manipulation. Co-solvency uses water-miscible solvents to improve drug solubility. Surfactants form micelles above the critical micelle concentration that can solubilize drugs. Complexation with cyclodextrin can enhance aqueous solubility. Manipulating a drug's solid state, such as forming polymorphs, can also increase solubility. The document provides examples and mechanisms for each solubility enhancement technique.