SMDM 2015
•
0 likes•121 views
1. The document presents a conceptual framework for pharmacoeconomic modeling of biosimilars in the US healthcare system. 2. Key challenges in modeling include price uncertainty, potential differences in indications from reference biologics, and uncertainty around interchangeability. 3. The framework recommends modeling a 35% average discount for biosimilars relative to reference biologics, biosimilar market share growth up to 60% over 10 years, and conducting sensitivity analyses to reflect uncertainty and the potential for factors and their interactions to change over time.
Report
Share
Report
Share
Download to read offline
Recommended
Carol Propper: Is competition a force for good in hospitals?

This document summarizes evidence on whether competition increases efficiency in healthcare. It finds that in the US, competition increases quality when prices are regulated, but the effects are less clear when prices are not regulated. UK evidence shows that recent healthcare reforms increased competition and led to higher quality care without increased spending. Competition was also found to improve hospital management practices. However, some negative effects were found when prices were determined by markets rather than regulated. Overall, the rules and degree of competition matter greatly for outcomes.
Training slides format

This document provides an overview of a molecule or product, including its history, mechanism of action, medical uses, effects on medical conditions, and comparisons to other molecules or products. It also discusses the molecule or product's market opportunities and outlines promotion strategies to target customers and analyze competitors.
Surya Singh: "From Development to Delivery in the US"

Surya Singh: "From Development to Delivery in the US"The Petrie-Flom Center for Health Law Policy, Biotechnology, and Bioethics
PLEASE NOTE: THESE SLIDES MAY NOT DISPLAY PROPERLY ONLINE, BUT THEY ARE READABLE IF DOWNLOADED.
October 28, 2018
This one-day conference explored the current pharmaceutical pricing landscape by bringing together leaders from the pharmaceutical industry, policymakers, legal practitioners, and scholars to engage in novel, interdisciplinary discussions to better understand current challenges and articulate best practices to address these issues. Participants assessed the current challenges presented in drug pricing policy, from development to delivery, in both the United States and international context. We also explored and articulated best practices to expand access to medicines and worked toward developing a plan for disseminating these practices more widely.Ameet Sarpatwari, "Drug Pricing and Cost"

Ameet Sarpatwari, "Drug Pricing and Cost"The Petrie-Flom Center for Health Law Policy, Biotechnology, and Bioethics
January 23, 2017
The Fifth Annual Health Law Year in P/Review symposium featured leading experts discussing major developments during 2016 and what to watch out for in 2017. The discussion at this day-long event covered hot topics in such areas as health policy under the new administration, regulatory issues in clinical research, law at the end-of-life, patient rights and advocacy, pharmaceutical policy, reproductive health, and public health law.
The Fifth Annual Health Law Year in P/Review was sponsored by the Petrie-Flom Center for Health Law Policy, Biotechnology, and Bioethics at Harvard Law School, Harvard Health Publications at Harvard Medical School, Health Affairs, the Hastings Center, the Program On Regulation, Therapeutics, And Law (PORTAL) in the Division of Pharmacoepidemiology and Pharmacoeconomics at Brigham and Women’s Hospital, and the Center for Bioethics at Harvard Medical School, with support from the Oswald DeN. Cammann Fund.
Learn more on our website: http://petrieflom.law.harvard.edu/events/details/5th-annual-health-law-year-in-p-reviewArticle

The document summarizes a study that used a microsimulation model to analyze the impacts of state policies on health outcomes and costs for people living with HIV/AIDS. The study used national data to estimate relationships between insurance coverage, health status, employment, treatment and medical costs. The model allowed researchers to simulate the effects of more generous state policies on economic outcomes. The researchers found that more generous policies, like increasing Medicaid eligibility, could improve health outcomes while increasing short-term costs for treatment but decreasing long-term hospitalization costs. However, the savings may not fully benefit the programs paying for increased treatment.
HawkPartners Perspective on Payer Research

If done right, payer research can offer important insights for your enterprise.
To learn more about HawkPartners, visit hawkpartners.com
Jerry Avorn, "Drug Regulation, Promotion, and Advocacy in 2017"

Jerry Avorn, "Drug Regulation, Promotion, and Advocacy in 2017"The Petrie-Flom Center for Health Law Policy, Biotechnology, and Bioethics
January 23, 2017
The Fifth Annual Health Law Year in P/Review symposium featured leading experts discussing major developments during 2016 and what to watch out for in 2017. The discussion at this day-long event covered hot topics in such areas as health policy under the new administration, regulatory issues in clinical research, law at the end-of-life, patient rights and advocacy, pharmaceutical policy, reproductive health, and public health law.
The Fifth Annual Health Law Year in P/Review was sponsored by the Petrie-Flom Center for Health Law Policy, Biotechnology, and Bioethics at Harvard Law School, Harvard Health Publications at Harvard Medical School, Health Affairs, the Hastings Center, the Program On Regulation, Therapeutics, And Law (PORTAL) in the Division of Pharmacoepidemiology and Pharmacoeconomics at Brigham and Women’s Hospital, and the Center for Bioethics at Harvard Medical School, with support from the Oswald DeN. Cammann Fund.
Learn more on our website: http://petrieflom.law.harvard.edu/events/details/5th-annual-health-law-year-in-p-reviewNational Association of Attorneys Generals’ Presidential Initiative Summit Ap...

Remarks by Thomas M. Moriarty, Executive Vice President, Chief Strategy Officer and General Counsel at the National Association of Attorneys Generals’ April 2017 Summit.
For more on the event, please visit: https://cvshealth.com/thought-leadership/making-progress-how-public-and-private-stakeholders-are-collaborating-to-improve-care
Recommended
Carol Propper: Is competition a force for good in hospitals?

This document summarizes evidence on whether competition increases efficiency in healthcare. It finds that in the US, competition increases quality when prices are regulated, but the effects are less clear when prices are not regulated. UK evidence shows that recent healthcare reforms increased competition and led to higher quality care without increased spending. Competition was also found to improve hospital management practices. However, some negative effects were found when prices were determined by markets rather than regulated. Overall, the rules and degree of competition matter greatly for outcomes.
Training slides format

This document provides an overview of a molecule or product, including its history, mechanism of action, medical uses, effects on medical conditions, and comparisons to other molecules or products. It also discusses the molecule or product's market opportunities and outlines promotion strategies to target customers and analyze competitors.
Surya Singh: "From Development to Delivery in the US"

Surya Singh: "From Development to Delivery in the US"The Petrie-Flom Center for Health Law Policy, Biotechnology, and Bioethics
PLEASE NOTE: THESE SLIDES MAY NOT DISPLAY PROPERLY ONLINE, BUT THEY ARE READABLE IF DOWNLOADED.
October 28, 2018
This one-day conference explored the current pharmaceutical pricing landscape by bringing together leaders from the pharmaceutical industry, policymakers, legal practitioners, and scholars to engage in novel, interdisciplinary discussions to better understand current challenges and articulate best practices to address these issues. Participants assessed the current challenges presented in drug pricing policy, from development to delivery, in both the United States and international context. We also explored and articulated best practices to expand access to medicines and worked toward developing a plan for disseminating these practices more widely.Ameet Sarpatwari, "Drug Pricing and Cost"

Ameet Sarpatwari, "Drug Pricing and Cost"The Petrie-Flom Center for Health Law Policy, Biotechnology, and Bioethics
January 23, 2017
The Fifth Annual Health Law Year in P/Review symposium featured leading experts discussing major developments during 2016 and what to watch out for in 2017. The discussion at this day-long event covered hot topics in such areas as health policy under the new administration, regulatory issues in clinical research, law at the end-of-life, patient rights and advocacy, pharmaceutical policy, reproductive health, and public health law.
The Fifth Annual Health Law Year in P/Review was sponsored by the Petrie-Flom Center for Health Law Policy, Biotechnology, and Bioethics at Harvard Law School, Harvard Health Publications at Harvard Medical School, Health Affairs, the Hastings Center, the Program On Regulation, Therapeutics, And Law (PORTAL) in the Division of Pharmacoepidemiology and Pharmacoeconomics at Brigham and Women’s Hospital, and the Center for Bioethics at Harvard Medical School, with support from the Oswald DeN. Cammann Fund.
Learn more on our website: http://petrieflom.law.harvard.edu/events/details/5th-annual-health-law-year-in-p-reviewArticle

The document summarizes a study that used a microsimulation model to analyze the impacts of state policies on health outcomes and costs for people living with HIV/AIDS. The study used national data to estimate relationships between insurance coverage, health status, employment, treatment and medical costs. The model allowed researchers to simulate the effects of more generous state policies on economic outcomes. The researchers found that more generous policies, like increasing Medicaid eligibility, could improve health outcomes while increasing short-term costs for treatment but decreasing long-term hospitalization costs. However, the savings may not fully benefit the programs paying for increased treatment.
HawkPartners Perspective on Payer Research

If done right, payer research can offer important insights for your enterprise.
To learn more about HawkPartners, visit hawkpartners.com
Jerry Avorn, "Drug Regulation, Promotion, and Advocacy in 2017"

Jerry Avorn, "Drug Regulation, Promotion, and Advocacy in 2017"The Petrie-Flom Center for Health Law Policy, Biotechnology, and Bioethics
January 23, 2017
The Fifth Annual Health Law Year in P/Review symposium featured leading experts discussing major developments during 2016 and what to watch out for in 2017. The discussion at this day-long event covered hot topics in such areas as health policy under the new administration, regulatory issues in clinical research, law at the end-of-life, patient rights and advocacy, pharmaceutical policy, reproductive health, and public health law.
The Fifth Annual Health Law Year in P/Review was sponsored by the Petrie-Flom Center for Health Law Policy, Biotechnology, and Bioethics at Harvard Law School, Harvard Health Publications at Harvard Medical School, Health Affairs, the Hastings Center, the Program On Regulation, Therapeutics, And Law (PORTAL) in the Division of Pharmacoepidemiology and Pharmacoeconomics at Brigham and Women’s Hospital, and the Center for Bioethics at Harvard Medical School, with support from the Oswald DeN. Cammann Fund.
Learn more on our website: http://petrieflom.law.harvard.edu/events/details/5th-annual-health-law-year-in-p-reviewNational Association of Attorneys Generals’ Presidential Initiative Summit Ap...

Remarks by Thomas M. Moriarty, Executive Vice President, Chief Strategy Officer and General Counsel at the National Association of Attorneys Generals’ April 2017 Summit.
For more on the event, please visit: https://cvshealth.com/thought-leadership/making-progress-how-public-and-private-stakeholders-are-collaborating-to-improve-care
Antibody drug conjugate market opportunity analysis

“Antibody Drug Conjugate Market Opportunity Analysis” Report Highlights:
Global ADC Market Insight
ADC Patent Analysis
Orphan Status & ADC
Favorable Market Drivers & Key Issues to be Discussed
ADC Clinical Trial Insight by Phase & Target Indications
ADC Profiles in Report: 201
Majority of ADC in Preclinical Phase: 83
Marketed ADC Clinical Profiles
SOA 2009 - New developments in Predictive Modeling and Risk Adjustment

This document summarizes challenges in selecting a risk adjustment system for a new public health program. It discusses whether to use diagnosis-based or pharmacy-based models and whether to adjust for all members or a subset. It also addresses recalibrating existing models for new programs and ensuring proper weighting for members with partial eligibility periods. The document notes that while risk adjustment is not perfect, the benefits include encouraging market participation and competition to potentially lower costs and improve quality of care. It suggests risk-based contracting between health plans and providers may be a future direction for risk adjustment.
Comparitive Assessment of Medical Devices and Product Choice by Orthodedic Su...

Comparitive Assessment of Medical Devices and Product Choice by Orthodedic Su...Leonard Davis Institute of Health Economics
The document summarizes a study that examined the criteria orthopedic surgeons use when selecting a vendor for hip and knee implants. It found that surgeons reported being most influenced by improved patient outcomes, implant longevity and design, and scientific evidence. They also considered the sales representative's level of service important. While vendor relationships and payments did not impact criteria, surgeons demonstrated loyalty to vendors they had trained with or been exposed to historically. The study concludes that comparative effectiveness research could help influence product choice and that minimal differentiation among vendors may help hospitals in purchasing decisions.David Dranove

This document analyzes market responses to obstetricians' negligence through a study of Florida malpractice and patient data. It finds that "high quality" obstetricians who face malpractice lawsuits lose privately insured patients but gain Medicaid patients, while "low quality" obstetricians lose patients overall. The financial impacts of negligence on obstetricians' earnings can last 5 years or more. While negligence does impose costs, the study questions how much the tort system alone deters negligence given markets also respond and the inefficiency of courts.
David Mitchell: "Runaway Train: America’s Drug Price Problem"

David Mitchell's slides from the Center for Health Journalism webinar, "Runaway Train: America’s Drug Price Problem," 5.15.18
More info: https://www.centerforhealthjournalism.org/content/runaway-train-americas-drug-price-problem
High Drug Costs

Americans pay significantly more for prescription drugs than people in other Western nations. In 2000, Americans paid 60% more on average for brand name drugs compared to seven other Western nations. By 2003, Americans paid 81% more on average. For many patented drugs in 2003, Americans paid 58-118% more than people in Switzerland, Britain, Germany, Canada, Sweden, France, and Italy. High drug costs in the US are driven by the pharmaceutical industry spending billions on marketing to doctors, creating influence over prescribing decisions and loyalty to brand name drugs. Marketing accounts for 30% of drug costs while research and development accounts for only 10-15%. Expensive new drugs are heavily marketed, including for off-label uses, driving up overall
Biosimilars: Regulatory and Clinical Considerations

Other considerations for clinical studies are that the regulations are written with some flexibility, and although clinical studies have been required thus far, they are not mandated by the regulations. Perception from sponsors is that innovators make process changes all the time that impact structure and no clinical study is done because of good analytical characterization so why are biosimilars different? With solid analytical and functional data, we should continue to challenge regulators on the need for clinical studies.
Minnesota Efforts to Promote Nursing Home Quality

Minnesota has tried various strategies to promote quality in nursing homes, including pay-for-performance, quality bonus payments, and nursing home report cards. A pay-for-performance program provided payments based on quality scores, with nursing homes keeping more of their actual costs depending on their quality tier. Minnesota has also developed quality profile elements and quality scores based on factors like staffing and resident satisfaction. A proposed plan tied case mix payments to quality scores but was not passed by the legislature.
PharmaDiplomacy Dialogue

This document summarizes the PharmaDiplomacy Dialogue initiative, which aims to improve relations between health systems and the pharmaceutical industry through diplomatic discussions. It brings together executives from healthcare payers, pharmaceutical companies, patient groups, and experts. The process involves five phases to develop a checklist to guide discussions at key points regarding clinical trials, drug approval, and on-market pricing reviews. The goal is to establish more trust and arrive at mutually acceptable drug prices that improve outcomes while supporting innovation. For this collaborative approach to work requires enabling factors like regulatory adjustments, leadership, policy support, and a neutral convener.
HCF 2019 Panel 3: Elena Montani

The document discusses measuring the performance of the EU chemicals acquis. It assessed effectiveness, efficiency, coherence, relevance, and EU added value. The assessment involved scientific evidence, citizens, stakeholders, and experts from several EU commission services and agencies. Challenges included assessing a broad framework with no baseline, data gaps, attribution issues, and diverging views. Next steps include collecting more data, evidence, and stakeholder input to conduct additional research and develop better indicators for assessing specific legislation.
Increasing the Likelihood of Biosimilar Success with Chemistry, Manufacturing...

A streamlined approach to biosimilar development to reduce timelines using insights gained from work on hundreds of biosimilar programs.
Projecting Medicines Expenditures in the English NHS Mestre-Ferrandiz AES 2013

Projecting Medicines Expenditures in the English NHS Mestre-Ferrandiz AES 2013Office of Health Economics
This document summarizes a study projecting UK National Health Service (NHS) expenditures on medicines from 2011-2015. The study used a bottom-up model incorporating historical trends, product lifecycles, research pipelines, and expert input. Key factors included the impact of new drug launches and increasing generic competition. Projections found total expenditures could grow 3.1-4.1% annually, with brands growing 0.5-1.8% and generics 10-11%. The model accounted for complex factors like varying price and volume erosion for products losing exclusivity in primary and secondary care settings.PharmaDiplomacy: Principles for Collaborative Mutually Acceptable Drug Prici...

PharmaDiplomacy: Principles for Collaborative Mutually Acceptable Drug Prici...Canadian Organization for Rare Disorders
This document summarizes the work of the PharmaDiplomacy group, which aims to rebuild trust between the pharmaceutical industry and health systems. The group identified pricing as a key source of mistrust. It developed principles for dialogue at different stages - from drug development through approval and ongoing review. The principles emphasize value, accommodate differences between systems, and rank issues by difficulty. Next steps include distributing the principles, applying them to new products, and engaging regulators to help rebuild trust through open communication.Dimasi 2007s

The document discusses trends in new drug development over time. It finds that while drug development times and costs have increased substantially, the number of new drug approvals has not kept pace with rising research and development spending. However, clinical testing pipelines for large pharmaceutical firms have grown in recent years, suggesting more new drugs are now entering development. The potential payoffs could be substantial for improvements that reduce development costs and timelines.
Kaizen draft slides for chris

1) The document discusses the changing regulatory environment in Canada for drug reimbursement, including new PMPRB pricing guidelines and the increasing influence of CADTH and PCPA in determining reimbursement.
2) It notes that PMPRB reforms may require new drugs to be priced at or below existing treatments, weakening the ability to negotiate pricing. HTA submissions need to demonstrate value beyond price to gain reimbursement.
3) Provinces face budget pressures that have led to policies favoring generics and biosimilars over optimized therapies, influencing CADTH and PCPA decisions and lengthening the time for new drugs to be reimbursed.
Summary Demand Models

Demand models analyze the relationships between product characteristics, sales, prices, and consumer preferences. Two common approaches are random utility models that consider consumer preferences and budget constraints, and discrete choice models where consumers value product attributes. Demand models are important in antitrust litigation to statistically illustrate complex consumer demand relationships.
On Innovations in Scholarly Communication: Interoperability of Persistent Ide...

Presentation at ORCID Taiwan Workshop (22 October 2015)
Social Media Marketing 2012-2013

What is social media marketing? Why should companies use it? How does it support CRM? What is the future of social media marketing?
This presentation looks at social media marketing utilizing the latest marketing theories. Blending real-world application with the latest studies and academic rationale for Social Media Marketing, we look at how SMM can bring ROI to your business.
© Copyright Marie Howell 2012
20 Super Actionable Social Media Tips

Use these tips to make your social media marketing EVEN better! We interviewed marketers who are in the weeds of social media every day to find their best tips for everything from building a quality social following to posting creative social content.
From Fans to Advocates: How to Build Community and Grow #BrandLove

What is community? How can making community central to your business help you grow? Beyond likes, +1s, shares and RTs, how can brands engage with their audiences to build lasting relationships that take them from fans to advocates? Hootsuite’s VP Community & Customer Experience, Jeanette Gibson, and Dr. William Ward, Director, Education Strategy share best practices and real-world examples of how a strong community of fans and followers can become a powerful tool in activating others to get involved and fall in love with your brand.
Effects of social media on Youth

The document discusses the effects of social media on youth. It notes that many children have social media profiles and share personal information online from a young age. It also discusses how social media can influence youth lifestyle choices and decision making, but also has some negative impacts like reduced attention spans and increased risk of depression or loneliness. The document also provides a SWOT analysis of social media which notes potential threats like privacy issues but also opportunities like a sense of community and engagement.
The Paradox of Exceptional Marketing

Rand Fishkin's presentation on the Fermi Paradox and how great filters relate to marketing activities. Presented at Conductor C3, Seattle Interactive Conference, and others.
More Related Content
What's hot
Antibody drug conjugate market opportunity analysis

“Antibody Drug Conjugate Market Opportunity Analysis” Report Highlights:
Global ADC Market Insight
ADC Patent Analysis
Orphan Status & ADC
Favorable Market Drivers & Key Issues to be Discussed
ADC Clinical Trial Insight by Phase & Target Indications
ADC Profiles in Report: 201
Majority of ADC in Preclinical Phase: 83
Marketed ADC Clinical Profiles
SOA 2009 - New developments in Predictive Modeling and Risk Adjustment

This document summarizes challenges in selecting a risk adjustment system for a new public health program. It discusses whether to use diagnosis-based or pharmacy-based models and whether to adjust for all members or a subset. It also addresses recalibrating existing models for new programs and ensuring proper weighting for members with partial eligibility periods. The document notes that while risk adjustment is not perfect, the benefits include encouraging market participation and competition to potentially lower costs and improve quality of care. It suggests risk-based contracting between health plans and providers may be a future direction for risk adjustment.
Comparitive Assessment of Medical Devices and Product Choice by Orthodedic Su...

Comparitive Assessment of Medical Devices and Product Choice by Orthodedic Su...Leonard Davis Institute of Health Economics
The document summarizes a study that examined the criteria orthopedic surgeons use when selecting a vendor for hip and knee implants. It found that surgeons reported being most influenced by improved patient outcomes, implant longevity and design, and scientific evidence. They also considered the sales representative's level of service important. While vendor relationships and payments did not impact criteria, surgeons demonstrated loyalty to vendors they had trained with or been exposed to historically. The study concludes that comparative effectiveness research could help influence product choice and that minimal differentiation among vendors may help hospitals in purchasing decisions.David Dranove

This document analyzes market responses to obstetricians' negligence through a study of Florida malpractice and patient data. It finds that "high quality" obstetricians who face malpractice lawsuits lose privately insured patients but gain Medicaid patients, while "low quality" obstetricians lose patients overall. The financial impacts of negligence on obstetricians' earnings can last 5 years or more. While negligence does impose costs, the study questions how much the tort system alone deters negligence given markets also respond and the inefficiency of courts.
David Mitchell: "Runaway Train: America’s Drug Price Problem"

David Mitchell's slides from the Center for Health Journalism webinar, "Runaway Train: America’s Drug Price Problem," 5.15.18
More info: https://www.centerforhealthjournalism.org/content/runaway-train-americas-drug-price-problem
High Drug Costs

Americans pay significantly more for prescription drugs than people in other Western nations. In 2000, Americans paid 60% more on average for brand name drugs compared to seven other Western nations. By 2003, Americans paid 81% more on average. For many patented drugs in 2003, Americans paid 58-118% more than people in Switzerland, Britain, Germany, Canada, Sweden, France, and Italy. High drug costs in the US are driven by the pharmaceutical industry spending billions on marketing to doctors, creating influence over prescribing decisions and loyalty to brand name drugs. Marketing accounts for 30% of drug costs while research and development accounts for only 10-15%. Expensive new drugs are heavily marketed, including for off-label uses, driving up overall
Biosimilars: Regulatory and Clinical Considerations

Other considerations for clinical studies are that the regulations are written with some flexibility, and although clinical studies have been required thus far, they are not mandated by the regulations. Perception from sponsors is that innovators make process changes all the time that impact structure and no clinical study is done because of good analytical characterization so why are biosimilars different? With solid analytical and functional data, we should continue to challenge regulators on the need for clinical studies.
Minnesota Efforts to Promote Nursing Home Quality

Minnesota has tried various strategies to promote quality in nursing homes, including pay-for-performance, quality bonus payments, and nursing home report cards. A pay-for-performance program provided payments based on quality scores, with nursing homes keeping more of their actual costs depending on their quality tier. Minnesota has also developed quality profile elements and quality scores based on factors like staffing and resident satisfaction. A proposed plan tied case mix payments to quality scores but was not passed by the legislature.
PharmaDiplomacy Dialogue

This document summarizes the PharmaDiplomacy Dialogue initiative, which aims to improve relations between health systems and the pharmaceutical industry through diplomatic discussions. It brings together executives from healthcare payers, pharmaceutical companies, patient groups, and experts. The process involves five phases to develop a checklist to guide discussions at key points regarding clinical trials, drug approval, and on-market pricing reviews. The goal is to establish more trust and arrive at mutually acceptable drug prices that improve outcomes while supporting innovation. For this collaborative approach to work requires enabling factors like regulatory adjustments, leadership, policy support, and a neutral convener.
HCF 2019 Panel 3: Elena Montani

The document discusses measuring the performance of the EU chemicals acquis. It assessed effectiveness, efficiency, coherence, relevance, and EU added value. The assessment involved scientific evidence, citizens, stakeholders, and experts from several EU commission services and agencies. Challenges included assessing a broad framework with no baseline, data gaps, attribution issues, and diverging views. Next steps include collecting more data, evidence, and stakeholder input to conduct additional research and develop better indicators for assessing specific legislation.
Increasing the Likelihood of Biosimilar Success with Chemistry, Manufacturing...

A streamlined approach to biosimilar development to reduce timelines using insights gained from work on hundreds of biosimilar programs.
Projecting Medicines Expenditures in the English NHS Mestre-Ferrandiz AES 2013

Projecting Medicines Expenditures in the English NHS Mestre-Ferrandiz AES 2013Office of Health Economics
This document summarizes a study projecting UK National Health Service (NHS) expenditures on medicines from 2011-2015. The study used a bottom-up model incorporating historical trends, product lifecycles, research pipelines, and expert input. Key factors included the impact of new drug launches and increasing generic competition. Projections found total expenditures could grow 3.1-4.1% annually, with brands growing 0.5-1.8% and generics 10-11%. The model accounted for complex factors like varying price and volume erosion for products losing exclusivity in primary and secondary care settings.PharmaDiplomacy: Principles for Collaborative Mutually Acceptable Drug Prici...

PharmaDiplomacy: Principles for Collaborative Mutually Acceptable Drug Prici...Canadian Organization for Rare Disorders
This document summarizes the work of the PharmaDiplomacy group, which aims to rebuild trust between the pharmaceutical industry and health systems. The group identified pricing as a key source of mistrust. It developed principles for dialogue at different stages - from drug development through approval and ongoing review. The principles emphasize value, accommodate differences between systems, and rank issues by difficulty. Next steps include distributing the principles, applying them to new products, and engaging regulators to help rebuild trust through open communication.Dimasi 2007s

The document discusses trends in new drug development over time. It finds that while drug development times and costs have increased substantially, the number of new drug approvals has not kept pace with rising research and development spending. However, clinical testing pipelines for large pharmaceutical firms have grown in recent years, suggesting more new drugs are now entering development. The potential payoffs could be substantial for improvements that reduce development costs and timelines.
Kaizen draft slides for chris

1) The document discusses the changing regulatory environment in Canada for drug reimbursement, including new PMPRB pricing guidelines and the increasing influence of CADTH and PCPA in determining reimbursement.
2) It notes that PMPRB reforms may require new drugs to be priced at or below existing treatments, weakening the ability to negotiate pricing. HTA submissions need to demonstrate value beyond price to gain reimbursement.
3) Provinces face budget pressures that have led to policies favoring generics and biosimilars over optimized therapies, influencing CADTH and PCPA decisions and lengthening the time for new drugs to be reimbursed.
Summary Demand Models

Demand models analyze the relationships between product characteristics, sales, prices, and consumer preferences. Two common approaches are random utility models that consider consumer preferences and budget constraints, and discrete choice models where consumers value product attributes. Demand models are important in antitrust litigation to statistically illustrate complex consumer demand relationships.
What's hot (16)
Antibody drug conjugate market opportunity analysis

Antibody drug conjugate market opportunity analysis
SOA 2009 - New developments in Predictive Modeling and Risk Adjustment

SOA 2009 - New developments in Predictive Modeling and Risk Adjustment
Comparitive Assessment of Medical Devices and Product Choice by Orthodedic Su...

Comparitive Assessment of Medical Devices and Product Choice by Orthodedic Su...
David Mitchell: "Runaway Train: America’s Drug Price Problem"

David Mitchell: "Runaway Train: America’s Drug Price Problem"
Biosimilars: Regulatory and Clinical Considerations

Biosimilars: Regulatory and Clinical Considerations
Increasing the Likelihood of Biosimilar Success with Chemistry, Manufacturing...

Increasing the Likelihood of Biosimilar Success with Chemistry, Manufacturing...
Projecting Medicines Expenditures in the English NHS Mestre-Ferrandiz AES 2013

Projecting Medicines Expenditures in the English NHS Mestre-Ferrandiz AES 2013
PharmaDiplomacy: Principles for Collaborative Mutually Acceptable Drug Prici...

PharmaDiplomacy: Principles for Collaborative Mutually Acceptable Drug Prici...
Viewers also liked
On Innovations in Scholarly Communication: Interoperability of Persistent Ide...

Presentation at ORCID Taiwan Workshop (22 October 2015)
Social Media Marketing 2012-2013

What is social media marketing? Why should companies use it? How does it support CRM? What is the future of social media marketing?
This presentation looks at social media marketing utilizing the latest marketing theories. Blending real-world application with the latest studies and academic rationale for Social Media Marketing, we look at how SMM can bring ROI to your business.
© Copyright Marie Howell 2012
20 Super Actionable Social Media Tips

Use these tips to make your social media marketing EVEN better! We interviewed marketers who are in the weeds of social media every day to find their best tips for everything from building a quality social following to posting creative social content.
From Fans to Advocates: How to Build Community and Grow #BrandLove

What is community? How can making community central to your business help you grow? Beyond likes, +1s, shares and RTs, how can brands engage with their audiences to build lasting relationships that take them from fans to advocates? Hootsuite’s VP Community & Customer Experience, Jeanette Gibson, and Dr. William Ward, Director, Education Strategy share best practices and real-world examples of how a strong community of fans and followers can become a powerful tool in activating others to get involved and fall in love with your brand.
Effects of social media on Youth

The document discusses the effects of social media on youth. It notes that many children have social media profiles and share personal information online from a young age. It also discusses how social media can influence youth lifestyle choices and decision making, but also has some negative impacts like reduced attention spans and increased risk of depression or loneliness. The document also provides a SWOT analysis of social media which notes potential threats like privacy issues but also opportunities like a sense of community and engagement.
The Paradox of Exceptional Marketing

Rand Fishkin's presentation on the Fermi Paradox and how great filters relate to marketing activities. Presented at Conductor C3, Seattle Interactive Conference, and others.
The impact of social media

with Melissa Morgan (www.ememdesign.com)
An introduction to gauging the impact of social-media on society in this media saturated, hyper-networked, über-techie, digitally innovative world.
The 4 Most Powerful Facebook Advertising Tools Marketers Aren't Using

This is from a webinar that covers the following features:
Conversion Tracking
Conversion Specs
Custom Audiences
Facebook Ad Reports
How to Pack a Punch With Social Media

This is the slide deck I used at Social Media Marketing World on March 26, 2015. It contains my top tips for using social media.
The Who What Where When And Why Of Social Media Lead Generation

Social Media is the place to be for lead generation. Each platform offers insight and information about thousands of potential prospects, and it’s all right there waiting for you.
But in order for your strategy to be efficient and effective, you have to identify the 5 W’s of your lead generation: Who, What, Where, When, and Why.
This SlideShare will give you a brief breakdown of what these elements are and how to use them to your advantage.
If I Knew Then What I Know Now/Skills That I Think Students Should Have/What ...

The document discusses 10 skills that students should learn: 1) how to continue learning, 2) how to separate correlation and causation, 3) how to pitch, 4) how to write software, 5) how to be brief, 6) how to use graphics, 7) how to make a video, 8) how to work social media, 9) how to reciprocate in advance, and 10) how to suck it up. It also provides information about an initiative called TechHire that aims to expand training opportunities and connect Americans to technology jobs.
Get People to Open your Emails

One of the most important elements of email marketing is writing subject lines that get people to open your emails.
How many times have you received a marketing email from someone and never opened it? Chances are more than once, and more than likely, it’s because the subject line didn’t grab you.
So, as you think about email marketing, how can you write effective email subject lines that get your subscribers to open your email?
Social media marketing ppt

This document discusses using various social media platforms for marketing purposes. It outlines strategies for using Twitter, Facebook, LinkedIn, and other sites like Faves and Delicious to increase traffic, build relationships with customers, promote brands and products, and generate sales. The key advice includes posting regular updates, engaging with followers, sharing links and content, monitoring insights and analytics, creating advertisements and groups, and customizing outreach efforts for each channel.
Building A Content Marketing Strategy

Before you start creating content, you need to build your strategy. We collected such valuable insights from over 40 major brands and thought leaders that we are rereleasing this ebook to help make your content marketing successful.
SOCIAL MEDIA - Connecting the World with Businesses

The document is a presentation about social media statistics and facts. It provides statistics on the number of users and engagement for major social media platforms including Twitter, Facebook, LinkedIn, Pinterest, Google+, Instagram, YouTube and others. Some key facts are that 92% of US companies use social media marketing, Twitter has 500 million users who tweet 58 million tweets per day, and Facebook has over 1.1 billion users and 2.5 billion likes per day. The presentation aims to provide interesting social media facts and statistics to readers.
What the F**K is Social Media: One Year Later

This document provides an overview of social media and why companies should engage with it. In 3 sentences: It discusses how social media usage has grown tremendously, with billions of users worldwide spending significant time on sites like Facebook and YouTube. However, most companies still treat social media as just another marketing channel rather than a way to build relationships through conversations and customer service. The document argues that if companies want to harness the power of social media, they need to define a strategic approach focused on listening, engaging with customers, and measuring the impact on goals like loyalty and word of mouth.
120 Awesome Marketing Stats, Charts and Graphs

http://www.HubSpot.com/charts - Over 120 marketing charts and graphs based on original research and data from a variety of sources, including analysis of our 6,500 business customers, surveys with hundreds of businesses responding, and dozens of top-notch publications like MarketingSherpa, eMarketer, Pew Research, McKinsey, and more. To download a free copy of your own, please visit http://hubspot.com/charts
The World's Biggest Social Media Team

The document describes how Tourism Australia has created the "World's Biggest Social Media Team" by leveraging user-generated content from millions of fans and followers who share their travel experiences and photos from Australia on social media platforms. Tourism Australia focuses on creating platforms where fans can share stories themselves rather than broadcasting messages, and makes fans the heroes by featuring their content. This approach has helped grow Tourism Australia's social media presence and make Australia a more sought-after travel destination through word-of-mouth recommendations.
Introduction to Social Media

Introduction to social media and social marketing. Part one of a two part seminar by Kelsey Ruger on the use of social media in business,
Social Media for Time-Strapped Entrepreneurs

The document discusses how entrepreneurs can effectively use social media with only 10 minutes per day. It recommends focusing on listening to understand audiences, engaging authentically by adding value to conversations, and sharing frequent, valuable content across multiple platforms. The key is to listen more than talk, always provide audience value, and adopt a "frequent snacks" approach to social sharing. With these efficient practices, entrepreneurs can better achieve their social media goals.
Viewers also liked (20)
On Innovations in Scholarly Communication: Interoperability of Persistent Ide...

On Innovations in Scholarly Communication: Interoperability of Persistent Ide...
From Fans to Advocates: How to Build Community and Grow #BrandLove

From Fans to Advocates: How to Build Community and Grow #BrandLove
The 4 Most Powerful Facebook Advertising Tools Marketers Aren't Using

The 4 Most Powerful Facebook Advertising Tools Marketers Aren't Using
The Who What Where When And Why Of Social Media Lead Generation

The Who What Where When And Why Of Social Media Lead Generation
If I Knew Then What I Know Now/Skills That I Think Students Should Have/What ...

If I Knew Then What I Know Now/Skills That I Think Students Should Have/What ...
SOCIAL MEDIA - Connecting the World with Businesses

SOCIAL MEDIA - Connecting the World with Businesses
Similar to SMDM 2015
Session 4 American Healthcare - Meyer

The document discusses the evolution of healthcare quality and the marketplace. It notes that the federal healthcare systems do not operate independently and are increasingly interconnected with civilian healthcare. Trends in civilian healthcare will impact the military health system, and the MHS will be viewed through the lens of civilian oversight. Payment reforms like value-based purchasing and payment for performance are pushing providers to improve quality and efficiency. Various models for alternative payment like bundled payments, global budgets, and accountable care organizations are discussed. Quality measurement and transparency are evolving and being used for different purposes like improvement, selection, and accountability.
Session 4 - American Healthcare

The document discusses the evolution of healthcare quality and the marketplace. It notes that trends in civilian healthcare will increasingly impact the military health system, and that federal health programs are seen as test beds for innovations. Payment reforms like value-based purchasing, pay for performance, and accountable care organizations aim to link payments to quality outcomes and reduce costs. Quality measurement and public reporting are important tools but must be done carefully. Overall the healthcare system is gradually shifting to pay providers based on the value of care delivered rather than volume of services.
Session-3-Status-of-the-Biosimilar-Market-–-Approvals-Challenges-and-Outlook-...

This document discusses the status of the biosimilar market including approvals, challenges, and outlook. It examines key characteristics of biosimilars that create different market dynamics in the US. Some of the major roadblocks to biosimilar approval and adoption that have been identified include reimbursement models, payor preference/rebate traps, provider and patient education needs, and interchangeability requirements. On average, there is a 631 day delay between FDA approval of a biosimilar and its US market launch, representing lost savings opportunities of tens of billions of dollars annually for the healthcare system.
Session-3-Status-of-the-Biosimilar-Market-–-Approvals-Challenges-and-Outlook-...

This document discusses the status of the biosimilar market including approvals, challenges, and outlook. It examines key characteristics of biosimilars that create different market dynamics in the US compared to Europe. Some of the current and historical roadblocks to biosimilar approval and adoption in the US include reimbursement models, payor preference and rebate traps, provider and patient education challenges, and interchangeability requirements. Looking ahead, 2019 is expected to see additional biosimilar competition in oncology with $13.5 billion in oncology spending facing new biosimilar options.
Comparative Effectiveness Research CER: A New Current In Pharmaceutical Bran...

The document discusses the rise of pharmaceutical comparative effectiveness research (CER) in the United States. CER provides insight into the clinical and cost effectiveness of different drug therapies. It is being driven by growing government and private payer interest in justifying healthcare costs. The federal government is a major funder of CER through agencies like AHRQ and NIH. For pharmaceutical companies, demonstrating strong CER performance can help gain preferred formulary placement and market position, while poor performance may disadvantage a drug. The document outlines considerations for a pharmaceutical brand to conduct its own pilot CER study to evaluate its drug against competitors.
Pharmacoeconomics

This document provides an overview of pharmacoeconomics, including its history, definitions, types of evaluations, and limitations. Pharmacoeconomics developed in the 1970s to analyze the costs of drug therapy. It is concerned with the economic impact of pharmaceutical products and services on individuals, health systems, and society. There are several types of pharmacoeconomic evaluations including cost-effectiveness analysis, cost-utility analysis, and cost-benefit analysis, which are used to compare drug programs and therapies. Pharmacoeconomic studies can be conducted during various phases of drug development and are used by industry, government, and private sectors to make decisions about research, pricing, and insurance coverage.
How High Will They Go? Managing Rising Drug Prices in a Changing Healthcare E...

In 2016, spending on prescription drugs is expected to reach $500 billion. So, how high will they really go? That is a common question asked with drug prices as recent headlines have exploited drug pricing schemes and how the pharmaceutical industry is handling rising prices. This webinar discusses how hospitals and health systems can prepare for and manage rising drug costs, ensure patient care and positively impact the bottom line.
Offsetting Effects of Prescription Drug Use on Medicare’s Spending for Medica...

Offsetting Effects of Prescription Drug Use on Medicare’s Spending for Medica...Congressional Budget Office
The Congressional Budget Office analyzed how changes in prescription drug use affect Medicare spending on medical services. It found that higher drug use leads to lower medical costs, as drugs help control diseases and reduce complications. The CBO reviewed studies on this "offset effect" and found that a 1% increase in drug use leads to a 0.2% decrease in other medical spending. As an example, the CBO estimated that closing the Medicare Part D coverage gap would increase drug use by 5% and lower medical costs by 1%, reducing the total cost of closing the gap.Dr. Aaron Kesselheim: "Runaway Train: America’s Drug Price Problem"

Dr. Aaron Kesselheim's slides from the Center for Health Journalism webinar, "Runaway Train: America’s Drug Price Problem," 5.15.18
More info: https://www.centerforhealthjournalism.org/content/runaway-train-americas-drug-price-problem
Incorporating Life-cycle Price Modelling into Pharmaceutical Cost-effectivene...

Incorporating Life-cycle Price Modelling into Pharmaceutical Cost-effectivene...Office of Health Economics
In this presentation, OHE's Pistollato explains why it is important to consider price changes after marketing in CEA analysis and presents an approach for doing so.Enterpreneurial -Pharmaceutical-Marketing (Long Ming).pdf

The document discusses various aspects of pharmaceutical marketing including how companies use sales representatives, drug samples, direct-to-consumer advertising, and pricing to promote their drugs, generate sales, and increase profits, though some argue these practices can negatively impact prescribing decisions and increase healthcare costs. It also outlines the drug development and approval process, and perspectives on the pharmaceutical industry from both advocates and critics.
Global regulatory landscape of biosimilars

This document summarizes the global regulatory landscape for biosimilars. It begins by defining biosimilars and biological drugs. It then discusses the guidelines established by various regulatory bodies including the EMA, FDA, WHO, and agencies in countries like Japan, Korea, Canada, China, and India. The guidelines generally require demonstrating biosimilarity to the reference product through comparative quality, nonclinical and clinical studies. The document also discusses business opportunities for biosimilars in emerging versus established markets and strategies used by originator companies to combat biosimilar competition. It concludes by noting concerns around interchangeability between biosimilars and reference products.
Faculty PowerPoint Amcp Template_032713_final draft_printouts

This document discusses adapting decision analytic models to meet the needs of the health system. It describes tier placement models, facility models, and portfolio models. Tier placement models focus on finding the optimal product placement within a clinical pathway to balance costs and outcomes. Facility models emphasize the facility perspective and can integrate multiple stakeholder views. The case studies demonstrate how these models can be used to evaluate the impact of new technologies and reimbursement levels on costs, resource use, and stakeholder incentives.
New Health Report 2012 - Media Briefing Deck 

The document summarizes the key findings of the 2012 New Health Report survey conducted by Quintiles. The survey gathered responses from over 1,350 stakeholders in the biopharmaceutical industry, managed care organizations, investors, and patients in the US and UK. The report found that stakeholders have differing views on risk tolerance and perceptions of healthcare quality. It also found that current tools for assessing risk are limited and better data is needed. Overall, the report aims to foster collaboration between stakeholders to improve innovation in pharmaceutical development and delivery.
Specialty one pager FINAL

This document discusses specialty drug management solutions from PwC. It notes that specialty drugs are the fastest growing component of pharmacy costs, accounting for 32% of costs while only representing 1% of prescriptions. There are concerns about the high and increasing costs of specialty drugs, variations in their management across pharmacy and medical benefits, and lack of transparency. PwC's approach involves diagnosing spending trends, formulating management strategies like innovative pricing and site of care optimization, and implementing programs to realize annual savings of 5-15% or $186,000-$558,000 for a typical employer.
Pharmacoeconomics5-WPS Office.pdf

Pharmacoeconomics is important to various stakeholders in healthcare. It is relevant to pharmaceutical manufacturers for developing cost-effective drugs, to prescribers for making informed formulary and treatment decisions, to pharmacists for drug evaluation and resource allocation, and especially to Indian patients who bear most out-of-pocket drug costs. The field utilizes economic evaluations like cost-benefit, cost-effectiveness, cost-minimization and cost-utility analyses to compare treatment strategies and outcomes.
Best Practices in Controlling Prescription Drug Costs

An insightful pharmacy benefit cost containment overview co-presented by Jeff Legg, PSG's Consulting Services VP and Labor Practice Leader, at the 2016 IFEBP Conference in Orlando.
Outlook 2011 presentation

The document provides an outlook on the global bio-pharmaceutical sector in 2011. It discusses factors that are positive for the sector such as increased R&D spending and novel drug approvals. Challenges mentioned include regulatory hurdles, biosimilars, and patent expiries. The document analyzes investment opportunities in areas like HCV, autoimmune diseases, and rare genetic disorders. It also examines issues facing mature biotechs and provides a valuation methodology.
Impact of Payer Policies on Pharmaceutical R&D

This comprehensive presentation examines the most important incentives and disincentives for innovation in the pharmaceutical and biotech industries, discussing their effect on decisions about R&D direction/targets.
20061020 mayo presentation

1. The document discusses moving healthcare competition away from lowering costs and towards increasing patient value. It proposes focusing on medical conditions over full care cycles and competing based on quality outcomes.
2. Key principles for value-based competition include defining value as health outcomes rather than just costs; basing competition on standardized outcome measures; focusing competition at the medical condition level; and that high quality care can be less expensive in the long run.
3. Experience, scale, and learning at the medical condition level drive value. Competition should also be regional or national rather than just local to allow for growth in condition-specific expertise. Public reporting of standardized outcome and cost data is necessary to enable value-based competition.
Similar to SMDM 2015 (20)
Session-3-Status-of-the-Biosimilar-Market-–-Approvals-Challenges-and-Outlook-...

Session-3-Status-of-the-Biosimilar-Market-–-Approvals-Challenges-and-Outlook-...
Session-3-Status-of-the-Biosimilar-Market-–-Approvals-Challenges-and-Outlook-...

Session-3-Status-of-the-Biosimilar-Market-–-Approvals-Challenges-and-Outlook-...
Comparative Effectiveness Research CER: A New Current In Pharmaceutical Bran...

Comparative Effectiveness Research CER: A New Current In Pharmaceutical Bran...
How High Will They Go? Managing Rising Drug Prices in a Changing Healthcare E...

How High Will They Go? Managing Rising Drug Prices in a Changing Healthcare E...
Offsetting Effects of Prescription Drug Use on Medicare’s Spending for Medica...

Offsetting Effects of Prescription Drug Use on Medicare’s Spending for Medica...
Dr. Aaron Kesselheim: "Runaway Train: America’s Drug Price Problem"

Dr. Aaron Kesselheim: "Runaway Train: America’s Drug Price Problem"
Incorporating Life-cycle Price Modelling into Pharmaceutical Cost-effectivene...

Incorporating Life-cycle Price Modelling into Pharmaceutical Cost-effectivene...
Enterpreneurial -Pharmaceutical-Marketing (Long Ming).pdf

Enterpreneurial -Pharmaceutical-Marketing (Long Ming).pdf
Faculty PowerPoint Amcp Template_032713_final draft_printouts

Faculty PowerPoint Amcp Template_032713_final draft_printouts
Best Practices in Controlling Prescription Drug Costs

Best Practices in Controlling Prescription Drug Costs
SMDM 2015
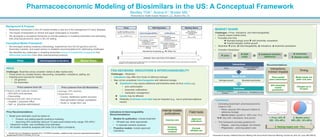
- 1. Recommendations: MARKET SHARE Challenges – Price, indications, and interchangeability: • Directly impact market share • Fluctuate over time è Branded biologic price ê with biosimilar competition è Overall biologics market growth • Biosimilar ê price, é interchangeability, é indications, é biosimilar penetration • Complex interactions: Pharmacoeconomic Modeling of Biosimilars in the US: A Conceptual Framework Bentley TGK1; Anene A1; Broder MS.1 1Partnership for Health Analytic Research, LLC, Beverly Hills, CA. Conceptual Model Framework • We leveraged existing modeling methodology, experiences from the US generics and EU biosimilars markets, and expert opinion to establish recommendations for addressing challenges. • We identified key challenges in modeling biosimilars around 3 fundamental components that differentiate biosimilars from other pharmaceutical products: Background & Purpose • Biosimilars’ introduction in the US market heralds a new era in the management of many diseases. • The impact of biosimilars on clinical and payer landscapes is uncertain. • We developed a conceptual framework to provide guidance in modeling biosimilars and estimating their pharmacoeconomic value in the US setting. Price Lessons from US § Generic small molecule market: – 50%-80% price declines – Discounting § Varying prices with sites of service: – Hospital v. physician office – Self- vs. physician-administered Price Lessons from EU Biosimilars § Average: 25% declines § Dependent on: – Country, healthcare system structure – Next-generation biologic competition – Acute vs. longer-term use 1. Mulcahy AW, et al. Perspective. November 2014. The RAND Corporation. Available at: http://www.rand.org/content/dam/rand/pubs/ perspectives/PE100/PE127/RAND_PE127.pdf. Presented at Society of Medical Decision Making 37th Annual North American Meeting, October 20, 2015, St. Louis, MO (PS3-9). Outcomes é Treatment initiation, duration é Adherence ê Healthcare costs Market share Biologics growth Biosimilar penetration Interactions Price Interchangeability/ indications 1. Price: 35% ! (SD: 10%-40%) 2. Market share: 60% (SD: 10%-90%) 3. Varying impact over ~10 y Price Interchangeability & Indications Market Share Conclusions • Estimating biosimilars’ pharmacoeconomic impact in US: • Price: assume 35% discount relative to reference biologic • Market share: growth to ~60% over 10 yr èè Vary with: indications, time period • This framework provides guidance for: • Payers – planning budgets, formularies • Physicians – planning patient care FDA DECISIONS: INDICATIONS & INTERCHANGEABILITY Challenges – Biosimilar: • Indications may differ from those of reference biologic • May not be considered interchangeable with reference biologic è Substitution may require additional administrative tasks (& thus costs), e.g.: • prior authorization • prescriber notifications • medication management è Uptake may be affected è Reliability of adverse event data may be impacted (e.g., due to pharmacovigilance issues) Indications & Interchangeability Recommendations: • Models for publication, include biosimilar: • Off-label use, when appropriate • In separate scenario analyses • Proactive models: include approved indications PRICE Challenges: Biosimilar prices uncertain: before & after market entry • Prices driven by complex factors: discounting, competition, indications, setting, etc. • Potential price sources for models: • US generics • EU biosimilars Price Recommendations: • Model price estimates could be based on: • Product- and setting-specific predictive modeling • Assuming 35% biosimilar discount 10 years post-market entry (range 10%-40%)1 • Interactive models: user-modifiable price estimates • All models: sensitivity analyses with wide CIs to reflect uncertainty Internal models, publications Case-by-case Scenario, sensitivity analyses Field tools Approved indications Exclude off-label use Interactions, Indirect Impacts Price-market share interactions 60% biosimilar market penetration1 Timing Model inputs not static over time Changes up to 10 years ê price ê OOP costs é treatment duration, adherence é outcomes, ê disease costs