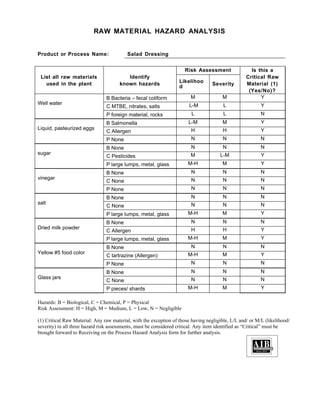
The document provides information about the Sincere Salad Dressing Company's HACCP program. It includes details about the company, products produced, organizational chart, HACCP team members, prerequisite programs in place, and a finished product profile. The company produces salad dressings and adheres to a HACCP plan and various prerequisite programs to control food safety hazards for its ready-to-eat products.