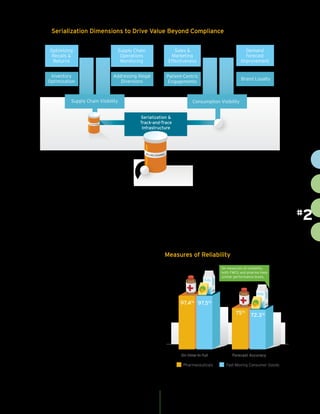
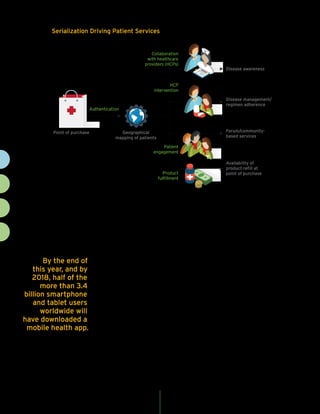
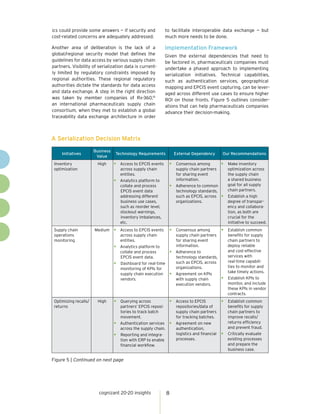
The document discusses the importance of serialization and track-and-trace capabilities in the pharmaceutical industry to combat issues like counterfeiting and improve supply chain efficiency. Regulatory compliance is highlighted as a priority, but the document emphasizes the potential for these tools to enhance patient engagement, operational agility, and sales effectiveness. By leveraging serialization data, pharmaceutical companies can optimize inventory management, streamline returns and recalls, and better target marketing efforts, ultimately driving business value beyond mere compliance.