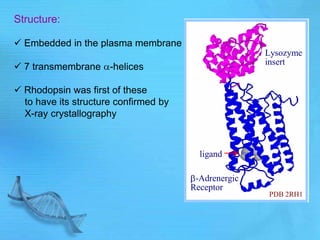
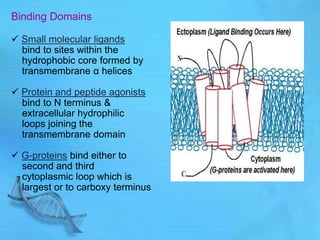
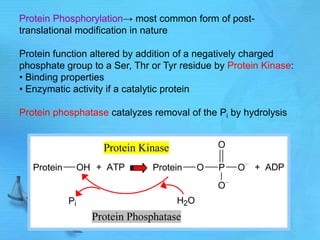
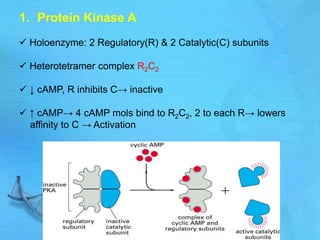
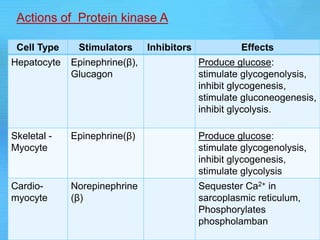
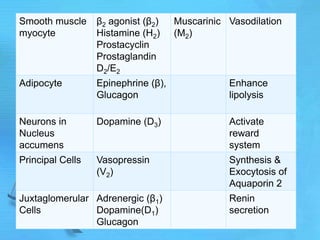
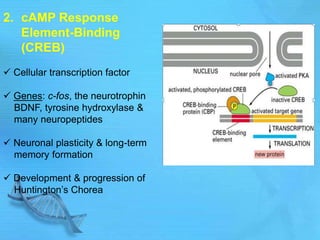
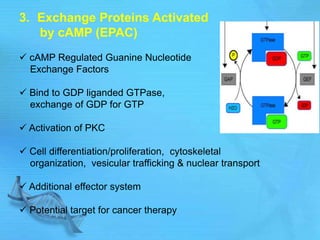
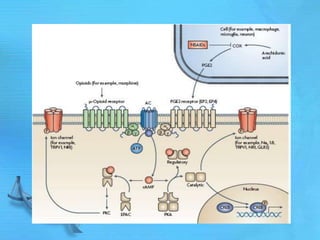
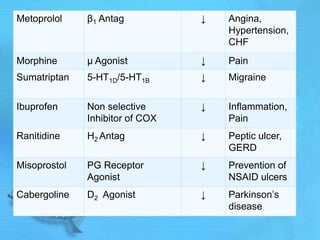
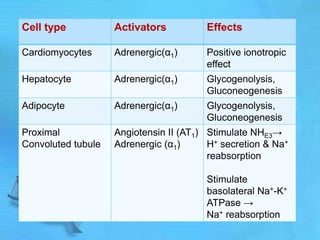
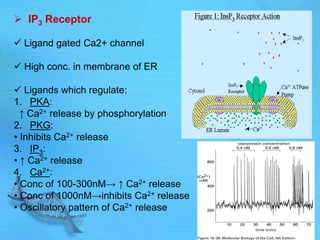
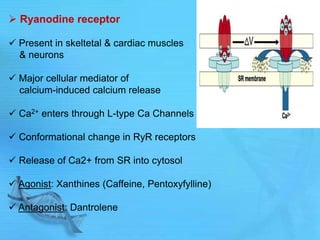
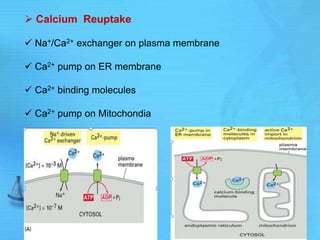
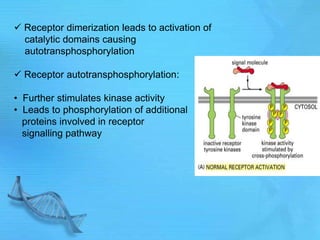
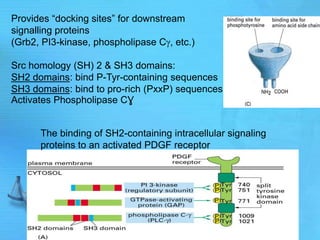
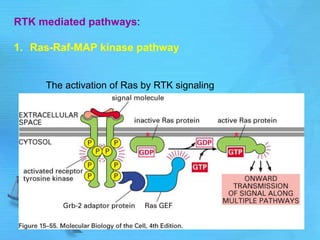
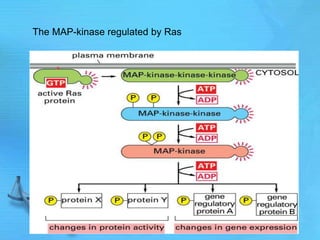
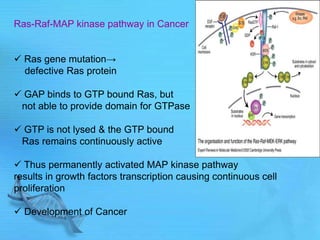
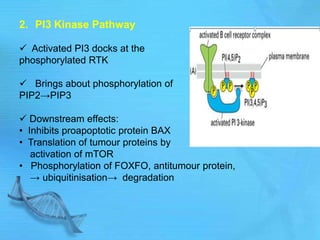
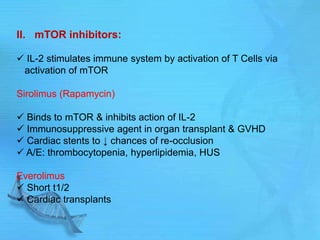
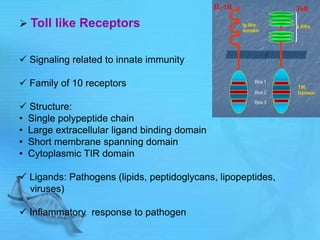
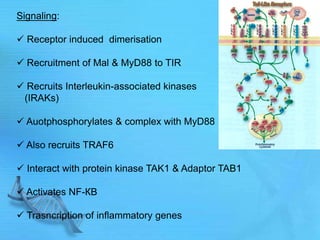
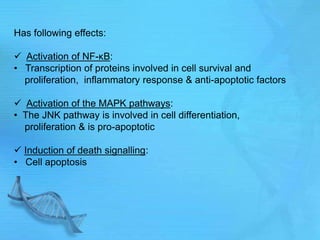
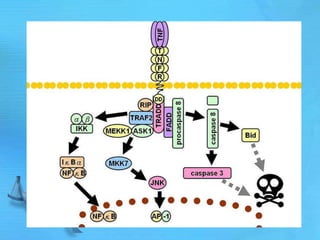
The document discusses second messenger systems. It describes how second messengers relay signals from cell surface receptors to target molecules inside the cell. Some key points discussed include:
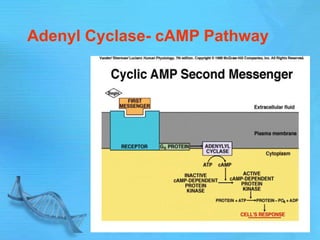
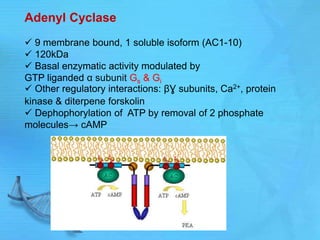
- Earl Sutherland discovered cyclic AMP (cAMP) as the second messenger for epinephrine and won the Nobel Prize for this work.
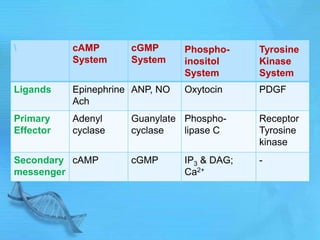
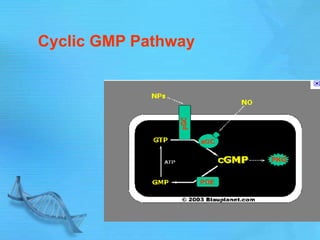
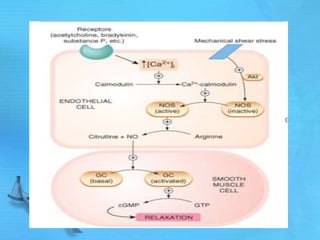
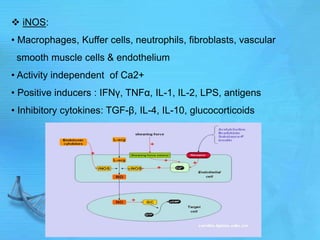
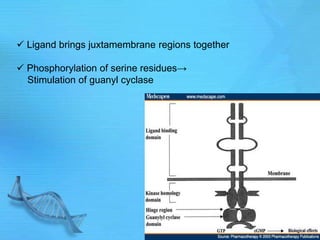
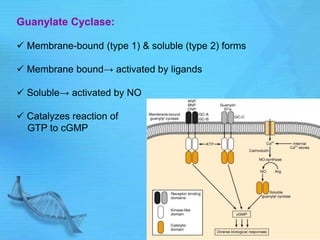
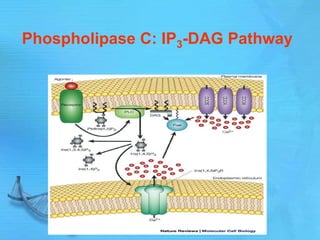
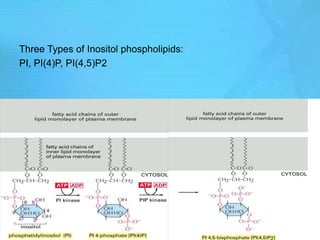
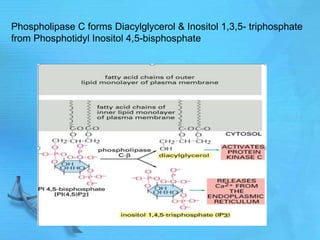
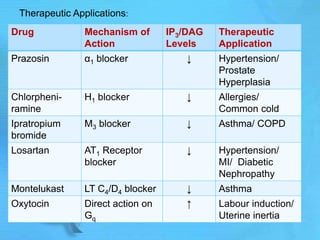
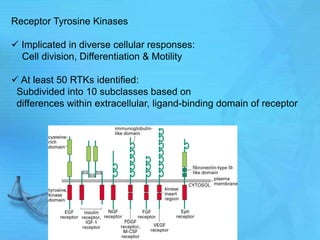
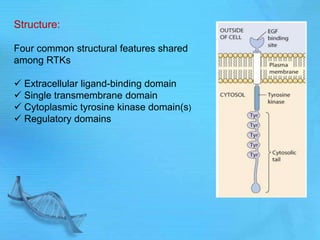
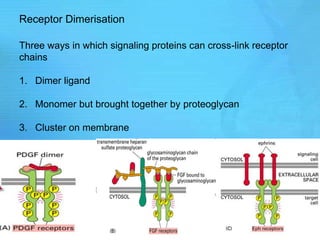
- Common second messenger systems include those using cAMP, cGMP, phosphatidylinositol, and tyrosine kinases as secondary messengers.
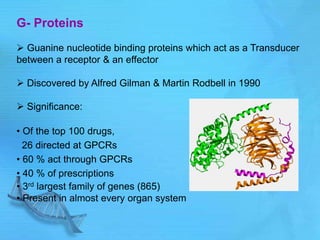
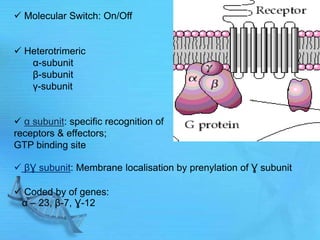
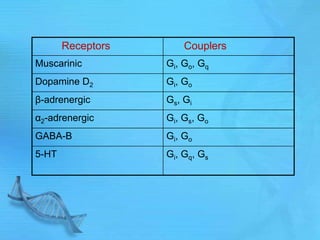
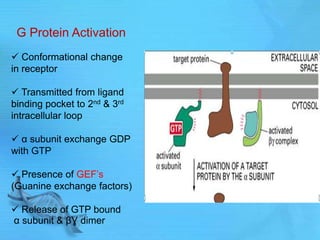
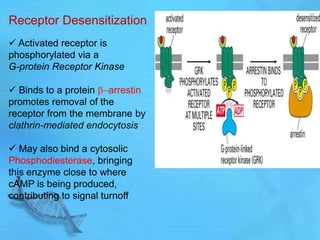
- G proteins act as transducers between receptors and effectors and are important drug targets.
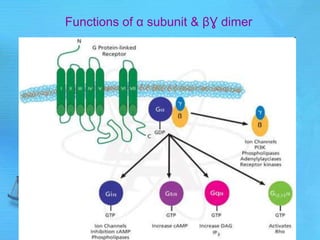
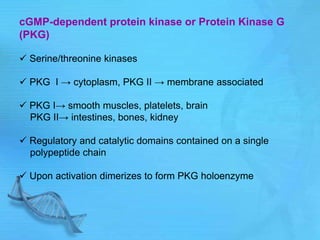
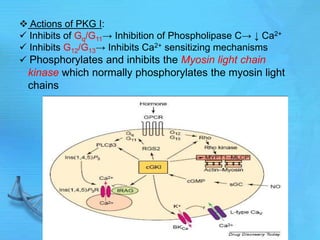
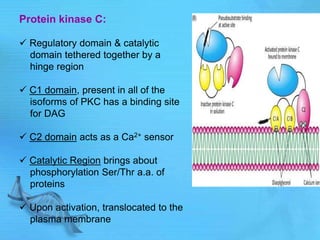
- cAMP and cGMP have several downstream targets including protein kinases that phosphorylate other proteins and regulate various cellular processes.