salt 8 (NH4)3PO4.pdf
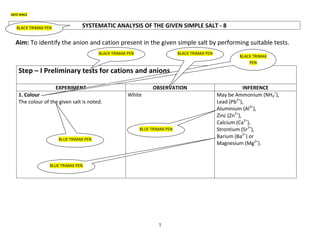
- 1. 1 DATE SPACE SYSTEMATIC ANALYSIS OF THE GIVEN SIMPLE SALT - 8 Aim: To identify the anion and cation present in the given simple salt by performing suitable tests. Step – I Preliminary tests for cations and anions EXPERIMENT OBSERVATION INFERENCE 1. Colour The colour of the given salt is noted. White May be Ammonium (NH4 + ), Lead (Pb2+ ), Aluminium (Al3+ ), Zinc (Zn2+ ), Calcium (Ca2+ ), Strontium (Sr2+ ), Barium (Ba2+ ) or Magnesium (Mg2+ ). BLACK TRIMAX PEN BLACK TRIMAX PEN BLACK TRIMAX PEN BLACK TRIMAX PEN BLUE TRIMAX PEN BLUE TRIMAX PEN BLUE TRIMAX PEN
- 2. 2 2.Solubility test A Pinch of salt is made into solution with Water (*water means distilled water only) Soluble May be chloride (Cl- ), bromide (Br- ), iodide (I- ), sulphate (SO4 2- ) , nitrate (NO3 - ), phosphate (PO4 3- ), oxalate (C2O4 2- ) or acetate (CH3COO- ) or May be Ammonium Carbonate (NH4)2CO3. 3. Action of heat A pinch of salt (dry) is strongly heated in a dry boiling test tube. (*No water/wetness in the salt and test tube) White sublimate formed on cooling the walls of the test tube. May be Ammonium. 4. Flame test A pinch of salt is made into a paste with few drops of Conc. HCI acid in a watch glass and introduced into the non-luminous part of Bunsen flame using loop holder. No characteristic flame. Absence of Cu2+ , Ca2+ , Sr2+ and Ba2+ .
- 3. 3 5. Ash test A small piece of filter paper is soaked into a mixture of given salt solution and Cobalt Nitrate solution and introduced into Bunsen flame and ignited. (*put off the fire completely & dispose in trash bin) No characteristic coloured ash. Absence of Al3+ , Zn2+ and Mg2+ . 6. Action of Sodium Hydroxide To 1 ml of salt solution NaOH is added and heated. A colourless pungent smelling gas is evolved which turns red litmus paper blue and gives dense white fumes with a rod dipped in conc. HCl. Presence of Ammonium.
- 4. 4 Step – II. Identification of Anions (Acid Radical) EXPERIMENT OBSERVATION INFERENCE 1. Action of dilute Sulphuric acid To 1 or 2 ml of dilute H2SO4, a small amount of salt is added. (*Do not use large quantity of dilute acid, *Do not heat the salt with dilute acid, * dil.HCl can also be used) No characteristic changes. Absence of carbonate and sulphide. 2. Action of Conc. sulphuric acid To 1 or 2 ml of Conc. H2SO4, a small amount of salt is added. No characteristic changes Absence of chloride, bromide, iodide, nitrate and oxalate. 3. Action of Manganese dioxide. To 0.1 g of salt, a pinch of manganese dioxide and conc. Sulphuric acid are added and heated. No characteristic changes Absence of chloride. 4. Copper turnings test: A small quantity of the salt is heated with Copper turnings and conc. Sulphuric acid. No reddish brown vapours Absence of Nitrate. 5. Chromyl Chloride test To pinch of salt, Potassium Dichromate salt and conc. Sulphuric acid are added and heated. No characteristic changes Absence of Chloride. 6.Ammonium Molybdate Test To 1 ml of salt solution, conc. Nitric acid is added and heated strongly until fume ceases. Then Ammonium Molybdate salt was added and boiled. Canary yellow precipitate. Presence of Phosphate.
- 5. 5 7. Ethyl Acetate Test To 1 ml of the salt solution, 1 ml of ethanol, few drops of Conc. Sulphuric acid were added and heated over a water bath. Then the mixture is poured into a beaker with excess sodium Carbonate solution. No pleasant fruity smell of ester Absence of Acetate. 8. Barium Chloride Test To a few drops of the salt solution or extract, dil. HCl and Barium Chloride soluble are added. No White Precipitate Absence of sulphate. Ethanol was missed in the previous salts, please include, it’s a mistake from my side. & compulsorily - Heated over water bath
- 6. 6 CONFIMATORY TEST FOR ANION EXPERIMENT OBSERVATION INFERENCE Phosphate – Ammonium molybdate Test To a few drops of the salt solution, conc. Nitric acid is added and heated strongly until fume ceases. Then Ammonium Molybdate salt was added and boiled. Canary yellow precipitate. Presence of Phosphate (PO4 3- ) is confirmed.
- 7. 7 TEST FOR CATIONS Cation Group analysis ( Group Separation) EXPERIMENT OBSERVATION INFERENCE Zero Group: To 1ml of the salt solution, few drops of Nessler's reagent and excess of sodium hydroxide are added and heated slightly. {* Nessler’s Reagent is K2[HgI4] - Potassium tetraiodomercurate(II)} A Reddish brown precipitate was obtained. Presence of Ammonium (NH4 + ) (Zero Group)
- 8. 8 CONFIRMATORY TEST FOR CATION EXPERIMENT OBSERVATION INFERENCE 1. Ammonium To 1ml of the salt solution, few drops of Nessler's reagent and excess of sodium hydroxide are added and heated slightly. {* Nessler’s Reagent is K2[HgI4] - Potassium tetraiodomercurate(II)} A Reddish brown precipitate was obtained. Presence of Ammonium (NH4 + ) Is confirmed. RESULT The given salt contains Basic Radical is Ammonium (NH4 + ) Acid Radical is Phosphate (PO4 3- ) Therefore, the given salt is Ammonium Phosphate (NH4)3PO4