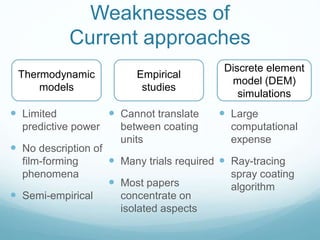
This document proposes a novel approach to modeling tablet film coating processes. It notes that film coating is commonly used to protect drugs, mask tastes, and control drug release profiles, but current batch processes can lead to problems and inconsistency. The proposed approach aims to provide a more holistic model that links macroscale parameters to microstructural processes, addressing weaknesses of prior approaches that considered only isolated aspects or lacked predictive power. This would help manufacturers reduce costly trials and support regulatory compliance by improving process understanding.